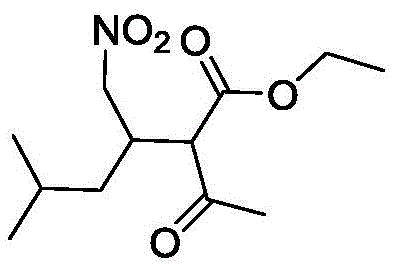
Advanced Green Synthesis of Pregabalin Intermediate Ethyl 2-Acetyl-3-Nitromethylene-5-Methyl-Hexanoate
Introduction to Next-Generation Pregabalin Intermediate Manufacturing
The pharmaceutical industry is currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of cost reduction. A pivotal development in this arena is documented in patent CN110862322B, which details a groundbreaking synthetic method for producing ethyl 2-acetyl-3-nitromethylene-5-methyl-hexanoate, a critical intermediate in the supply chain for Pregabalin. This molecule serves as a foundational building block for one of the most widely prescribed medications for neuropathic pain and epilepsy. The innovation lies not merely in the chemical transformation but in the complete reimagining of the reaction medium, replacing hazardous volatile organic solvents with a biodegradable Deep Eutectic Solvent (DES). For R&D directors and procurement strategists, this represents a dual opportunity: enhancing the purity profile of the final API while simultaneously securing a more resilient and environmentally compliant supply chain. As we analyze the technical specifics, it becomes clear that this methodology offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitro-containing intermediates like ethyl 2-acetyl-3-nitromethylene-5-methyl-hexanoate has been plagued by significant operational and environmental challenges. Traditional protocols typically rely on volatile organic solvents such as dichloromethane, tetrahydrofuran, or various alcohols, which pose severe health risks to operators and require complex, energy-intensive distillation processes for recovery. Furthermore, these conventional routes often necessitate the use of strong bases or expensive transition metal catalysts to drive the Henry reaction and subsequent Michael addition. These harsh conditions frequently lead to the formation of difficult-to-remove impurities, complicating the purification process and lowering the overall atom economy. The inability to recycle these solvents effectively results in substantial chemical waste, creating a heavy burden on waste treatment facilities and inflating the cost of goods sold (COGS). For supply chain heads, these factors translate into longer lead times and higher vulnerability to regulatory changes regarding solvent emissions.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a transformative approach utilizing a Deep Eutectic Solvent (DES) formed from choline chloride and urea. This eutectic mixture acts as both the reaction medium and the catalyst, creating a unique hydrogen-bonding network that activates the substrates without the need for additional hazardous reagents. The process operates under mild thermal conditions, specifically at 80°C, which significantly reduces energy consumption compared to high-temperature reflux methods. A standout feature of this novel approach is the simplified work-up procedure; upon completion of the reaction, the addition of a small amount of water causes the product to precipitate as a solid, allowing for immediate isolation via simple filtration. This eliminates the need for extensive extraction and chromatography steps. Moreover, the aqueous phase containing the DES can be evaporated to recover the solvent for reuse, establishing a closed-loop system that aligns perfectly with green chemistry principles and offers substantial cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into DES-Catalyzed Cascade Reaction
The success of this synthetic route relies on the unique physicochemical properties of the Choline Chloride-Urea DES. In the first stage, the DES facilitates a Henry reaction (nitroaldol condensation) between isovaleraldehyde and nitromethane. The urea component of the DES acts as a hydrogen bond donor, activating the carbonyl group of the aldehyde, while the basic nature of the eutectic mixture assists in the deprotonation of nitromethane to form the reactive nitronate anion. This synergistic effect accelerates the formation of 1-nitro-4-methyl-1-pentene with high stereoselectivity and yield. In the second stage, this nitroalkene undergoes a Michael addition with ethyl acetoacetate. The DES environment stabilizes the transition state of this conjugate addition, ensuring that the nucleophilic attack occurs efficiently at the beta-carbon of the nitroalkene. This cascade sequence is crucial for constructing the carbon skeleton required for Pregabalin, and the absence of competing side reactions ensures a clean impurity profile, which is a primary concern for R&D directors focusing on downstream processing.
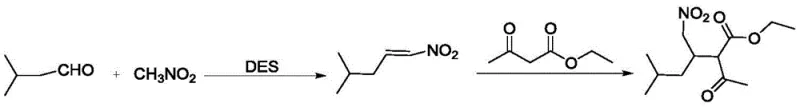
Controlling impurities in nitro-compound synthesis is notoriously difficult due to the potential for polymerization or over-reaction. However, the specific molar ratios employed in this patent—specifically a ratio of choline chloride to urea to isovaleraldehyde to nitromethane of roughly 1:2:1:1.2—create an optimized microenvironment that suppresses these unwanted pathways. The viscosity and polarity of the DES likely restrict the mobility of reactive intermediates just enough to favor the desired bimolecular collisions while preventing uncontrolled radical reactions. Furthermore, the mild acidity/basicity balance of the DES prevents the hydrolysis of the ester group in ethyl acetoacetate, a common degradation pathway in aqueous or strongly basic traditional systems. This precise control over the reaction mechanism translates directly to higher purity crude products, reducing the load on purification units and increasing the overall throughput of the manufacturing facility.
How to Synthesize Ethyl 2-Acetyl-3-Nitromethylene-5-Methyl-Hexanoate Efficiently
Implementing this synthesis requires strict adherence to the preparation of the eutectic solvent prior to substrate addition. The process begins with the formation of the liquid DES by heating choline chloride and urea, followed by the sequential addition of reactants. Temperature control is vital, with the reaction maintained at 80°C to ensure optimal kinetics without thermal degradation. The simplicity of the work-up, involving water precipitation and filtration, makes this protocol highly attractive for pilot plant operations. For detailed standard operating procedures and specific stoichiometric calculations, please refer to the technical guide below.
- Preparation of DES and Nitroalkene: Mix choline chloride and urea (1:2 molar ratio) at 80°C to form DES. React isovaleraldehyde with nitromethane in DES at 80°C to obtain 1-nitro-4-methyl-1-pentene.
- Michael Addition Reaction: In the recovered DES, react the obtained 1-nitro-4-methyl-1-pentene with ethyl acetoacetate at 80°C.
- Isolation and Recovery: Cool the mixture, add water to precipitate the solid product, filter, and wash. Evaporate the aqueous phase to recover DES for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DES-based technology offers compelling strategic advantages beyond mere technical feasibility. The elimination of volatile organic solvents removes a significant cost center associated with solvent purchase, storage, and hazardous waste disposal. By shifting to a reusable, non-toxic medium, manufacturers can drastically simplify their environmental compliance reporting and reduce the risk of production stoppages due to regulatory audits. The raw materials—isovaleraldehyde, nitromethane, and ethyl acetoacetate—are commodity chemicals with stable global supply chains, ensuring that production is not bottlenecked by exotic reagent availability. This reliability is essential for maintaining continuous supply to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the recyclability of the reaction medium. Since the DES can be recovered and reused multiple times without significant loss of efficiency, the recurring cost of solvents is virtually eliminated. Additionally, the simplified isolation process (precipitation and filtration) reduces the requirement for expensive separation equipment and lowers energy consumption associated with distillation. These factors combine to lower the overall production cost per kilogram, providing a competitive edge in pricing negotiations for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing a robust catalytic system that tolerates minor variations in feedstock quality enhances the resilience of the supply chain. The mild reaction conditions (80°C) reduce the risk of thermal runaway incidents, ensuring safer operations and minimizing unplanned downtime. Furthermore, the high yields reported (consistently above 90%) mean that less raw material is required to produce the same amount of product, optimizing inventory turnover rates. This efficiency allows suppliers to respond more rapidly to fluctuations in market demand, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of sensitive catalysts or extreme pressure requirements. The non-toxic nature of choline chloride and urea aligns with increasingly strict global environmental standards, future-proofing the manufacturing asset against tightening regulations on chemical emissions. The ability to treat the aqueous waste stream easily, given the biodegradability of the components, further reduces the environmental footprint, making this an ideal candidate for sustainable chemical manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply networks. The answers are derived directly from the experimental data and beneficial effects described in the patent literature.
Q: What are the primary advantages of using DES over traditional organic solvents for this synthesis?
A: The use of Deep Eutectic Solvents (DES) composed of choline chloride and urea eliminates the need for toxic volatile organic compounds (VOCs). This significantly simplifies post-treatment, as the product precipitates upon water addition, allowing for easy filtration. Furthermore, the DES can be recovered from the aqueous phase and reused multiple times without significant loss in catalytic activity, drastically reducing waste disposal costs and environmental impact.
Q: What yields can be expected from this novel synthetic route?
A: According to the patent data, this method achieves exceptionally high yields. The first step, synthesizing 1-nitro-4-methyl-1-pentene, reaches yields of approximately 97%. The subsequent Michael addition step to form the final pregabalin intermediate consistently achieves yields between 93% and 97%, depending on the stoichiometric ratio of ethyl acetoacetate used, demonstrating superior efficiency compared to conventional methods.
Q: Is the DES catalyst stable enough for industrial scale-up?
A: Yes, the stability of the DES system is a key feature of this technology. Experimental data indicates that the DES can be recycled and reused for at least five consecutive cycles while maintaining product yields above 95%. This robustness ensures consistent production quality and minimizes the consumption of fresh catalyst materials, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 2-Acetyl-3-Nitromethylene-5-Methyl-Hexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the long-term viability of the pharmaceutical sector. Our team of expert chemists has extensively evaluated the DES-catalyzed pathway described in patent CN110862322B and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the laboratory can be faithfully reproduced on an industrial scale. Our rigorous QC labs and stringent purity specifications guarantee that every batch of ethyl 2-acetyl-3-nitromethylene-5-methyl-hexanoate meets the exacting standards required for GMP API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative technology can optimize your supply chain. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and sustainability in your manufacturing operations.
