Advanced Synthesis of Hexamethyl Terphenyl Dimethyl Ester for High-Performance Fuel Cell Membranes
Advanced Synthesis of Hexamethyl Terphenyl Dimethyl Ester for High-Performance Fuel Cell Membranes
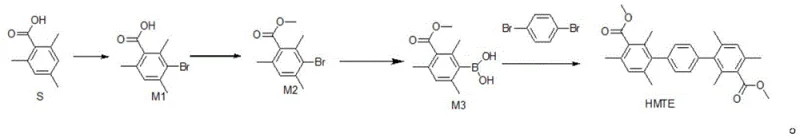
The development of efficient synthetic routes for advanced polymer monomers is critical for the next generation of energy storage technologies, particularly Alkaline Anion Exchange Membranes (AAEM). Patent CN109180483B discloses a novel and economically superior method for synthesizing hexamethyl terphenyl dimethyl ester (HMTE), a pivotal intermediate monomer used in the production of HMT-PMBI conductive polymers. This technical breakthrough addresses the longstanding cost barriers associated with traditional synthesis pathways by strategically reversing the coupling partners in the final Suzuki-Miyaura cross-coupling step. Instead of relying on expensive diboronic acid derivatives, this innovative process utilizes inexpensive p-dihalobenzenes, thereby fundamentally altering the cost structure of the supply chain. For R&D directors and procurement managers in the electronic materials sector, understanding this shift is essential for securing a reliable hexamethyl terphenyl dimethyl ester supplier capable of delivering high-purity intermediates at a competitive price point.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hexamethyl terphenyl dimethyl ester has been hindered by the reliance on costly starting materials that severely impact the overall profitability of the final polymer product. As illustrated in prior art such as Chinese patent application CN106255715A, the conventional route typically involves the reaction of methyl 3-bromo-2,4,6-trimethylbenzoate with 1,4-phenylene diboronic acid. While chemically feasible, this approach suffers from significant economic drawbacks because 1,4-phenylene diboronic acid is a high-value reagent with a complex supply chain and elevated market price. Furthermore, the yield of the final coupling step in these traditional methods often hovers around 60%, which necessitates larger batch sizes and increased solvent consumption to meet production targets. These inefficiencies create a bottleneck for the commercial scale-up of complex electronic chemical manufacturing, making the final AAEM membranes less competitive against established proton exchange membrane technologies.
The Novel Approach
The methodology described in patent CN109180483B introduces a strategic inversion of the synthetic logic that dramatically improves cost efficiency and raw material availability. By converting the bromo-intermediate into a boronate species first, the process enables the use of p-dibromobenzene, p-diiodobenzene, or p-dichlorobenzene as the coupling partner in the final step. These dihalobenzenes are commodity chemicals produced on a massive industrial scale, offering a stark contrast to the niche availability of diboronic acids. This switch not only reduces the direct material costs but also simplifies the purification process, as the byproducts generated from the halide coupling are often easier to remove than those from boronic acid couplings. The result is a streamlined workflow that maintains high yields while drastically lowering the entry barrier for mass production.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling Optimization
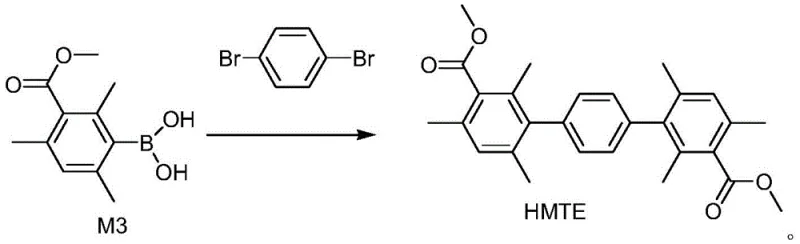
The core of this technological advancement lies in the precise execution of a two-step sequence involving organolithium chemistry followed by palladium-catalyzed cross-coupling. In the first stage, methyl 3-bromo-2,4,6-trimethylbenzoate undergoes lithium-halogen exchange at cryogenic temperatures ranging from -40°C to -50°C in an inert atmosphere. This low-temperature control is absolutely critical to prevent nucleophilic attack on the ester group and to ensure the exclusive formation of the aryl lithium species, which is subsequently trapped by a trialkyl borate to form the stable methyl 3-borato-2,4,6-trimethylbenzoate intermediate. The second stage involves the reaction of this newly formed boronate with the dihalobenzene in the presence of a palladium catalyst, such as tetrakis(triphenylphosphine)palladium, and an inorganic base like potassium carbonate. This Suzuki coupling proceeds under reflux conditions in a solvent system such as dioxane, facilitating the formation of the terphenyl backbone with high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages over the reverse coupling strategy. The use of excess boronate intermediate relative to the dihalobenzene (molar ratio of 2 to 2.5 : 1) ensures that the dihalobenzene is fully consumed, minimizing the presence of mono-coupled byproducts that are difficult to separate. Additionally, the workup procedure described involves pouring the reaction mixture into an aqueous ethanol solution, which induces precipitation of the crude product while leaving soluble impurities in the mother liquor. Subsequent recrystallization from ethyl acetate further refines the crystal lattice, removing trace palladium residues and unreacted starting materials. This rigorous purification protocol is essential for achieving the stringent purity specifications required for high-purity OLED material and fuel cell membrane precursors, ensuring consistent performance in the final electrochemical device.
How to Synthesize Hexamethyl Terphenyl Dimethyl Ester Efficiently
The synthesis of this critical monomer requires strict adherence to anhydrous conditions and precise temperature management to maximize yield and safety. The process begins with the preparation of the boronate intermediate via lithiation, followed immediately by the cross-coupling reaction without isolating unstable organometallic species. Operators must ensure that the inert atmosphere is maintained throughout to prevent oxidation of the sensitive intermediates. The detailed standardized synthesis steps, including specific reagent quantities, addition rates, and quenching protocols, are outlined in the structured guide below to facilitate technology transfer and process validation.
- Prepare methyl 3-borato-2,4,6-trimethylbenzoate by reacting methyl 3-bromo-2,4,6-trimethylbenzoate with trialkyl borate and a nucleophile at low temperature.
- Perform Suzuki coupling between the boronate intermediate and p-dihalobenzene using a palladium catalyst and inorganic base under reflux.
- Isolate the crude product via filtration and purify through recrystallization in ethyl acetate to obtain high-purity HMTE.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond simple unit price reductions. By shifting the dependency from specialized boronic acids to ubiquitous dihalobenzenes, manufacturers can mitigate supply chain risks associated with single-source suppliers of niche reagents. This diversification of the raw material base enhances supply continuity and provides greater leverage in vendor negotiations, ultimately leading to substantial cost savings in electronic chemical manufacturing. Furthermore, the simplified downstream processing reduces the burden on waste treatment facilities, aligning with increasingly strict environmental compliance standards.
- Cost Reduction in Manufacturing: The substitution of expensive 1,4-phenylene diboronic acid with low-cost p-dibromobenzene eliminates a major cost driver from the bill of materials. Since the dihalobenzene is used as the limiting reagent in a favorable stoichiometric ratio, the overall atom economy is improved, and the cost per kilogram of the final API intermediate is significantly lowered. This economic efficiency allows for more competitive pricing strategies in the global market for fuel cell components.
- Enhanced Supply Chain Reliability: p-Dibromobenzene and related dihalobenzenes are produced by numerous chemical manufacturers worldwide, ensuring a robust and redundant supply network. This abundance means that lead times for high-purity intermediates can be drastically reduced compared to sourcing specialized boronic acids, which often have longer production cycles. Consequently, inventory holding costs can be minimized, and just-in-time delivery models become more feasible for large-scale polymer production.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in dioxane with standard inorganic bases, are highly amenable to scale-up from laboratory benchtop to multi-ton industrial reactors. The absence of exotic catalysts or extreme pressure requirements simplifies equipment specifications and reduces capital expenditure. Additionally, the aqueous workup and recrystallization steps generate waste streams that are easier to treat and dispose of, supporting sustainable manufacturing practices and reducing the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process feasibility and product quality. Understanding these details is crucial for stakeholders evaluating the potential for licensing or contracting the production of this material.
Q: Why is p-dibromobenzene preferred over 1,4-phenylene diboronic acid in this synthesis?
A: p-Dibromobenzene is significantly cheaper and more readily available as a commodity chemical compared to 1,4-phenylene diboronic acid, leading to substantial raw material cost reductions without compromising the final yield.
Q: What are the critical temperature controls required for the lithiation step?
A: The lithiation and boration reaction must be strictly maintained between -40°C and -50°C to ensure selective formation of the boronate intermediate and prevent side reactions or decomposition of the organolithium species.
Q: How does this method impact the purity profile of the final polymer monomer?
A: By utilizing a robust Suzuki coupling protocol with optimized workup procedures including ethanol precipitation and ethyl acetate recrystallization, the method consistently achieves purity levels exceeding 98%, which is critical for high-performance membrane applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexamethyl Terphenyl Dimethyl Ester Supplier
As the demand for high-efficiency fuel cells and advanced ion-exchange membranes continues to grow, securing a stable supply of high-quality monomers like HMTE is paramount for maintaining production schedules and product performance. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of hexamethyl terphenyl dimethyl ester delivered meets the exacting standards required for next-generation electronic materials.
We invite you to engage with our technical team to discuss how this optimized synthetic route can be integrated into your supply chain to drive down costs and improve efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us partner with you to accelerate the deployment of advanced energy solutions.
