Advanced Manufacturing of Tropicamide: A Strategic Shift from Tropic Acid to Malonate Precursors
Advanced Manufacturing of Tropicamide: A Strategic Shift from Tropic Acid to Malonate Precursors
The global demand for high-quality ophthalmic agents continues to drive innovation in the synthesis of key active pharmaceutical ingredients (APIs) and their intermediates. Among these, Tropicamide (CAS: 1508-75-4) remains a cornerstone drug for mydriasis and cycloplegia in diagnostic eye examinations. However, traditional manufacturing pathways have long been plagued by inefficiencies that hinder cost-effective mass production. A pivotal breakthrough in this domain is documented in patent CN103159672A, which outlines a robust preparation method utilizing phenyl ethyl malonate as the primary raw material. This technical insight report analyzes the strategic advantages of this novel synthetic route, specifically designed for R&D directors and procurement leaders seeking a reliable ophthalmic API intermediate supplier. By shifting the synthetic entry point from unstable tropic acid derivatives to stable malonate esters, the industry can achieve significant improvements in yield consistency and impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tropicamide has relied heavily on routes originating from tropic acid, as exemplified by the method reported by Silvia Dei et al. This conventional pathway necessitates a complex sequence involving the overprotection of the hydroxyl group, followed by acylation, condensation, and eventual hydrolysis. The fundamental flaw in this approach lies in the chemical instability of the protected intermediates generated early in the sequence. Specifically, the intermediate designated as compound 2 in the traditional scheme is notoriously unstable, leading to uncontrolled side reactions and the generation of a complex impurity spectrum. This instability creates a bottleneck in downstream processing, making purification extremely difficult and resulting in a disappointingly low total recovery rate. For procurement managers, this translates to higher waste disposal costs and inconsistent batch-to-batch quality, undermining the economic viability of large-scale production.
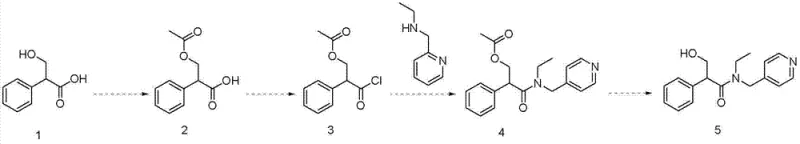
The Novel Approach
In stark contrast, the methodology disclosed in CN103159672A introduces a paradigm shift by employing diethyl phenylmalonate (phenyl ethyl malonate) as the starting scaffold. This strategic change bypasses the need for hydroxyl protection entirely, as the hydroxyl functionality is introduced late in the synthesis via a controlled reduction step. The new route proceeds through a logical sequence of selective hydrolysis, acylation, condensation, and reduction. By avoiding the fragile protected intermediates of the legacy route, the process ensures that each step operates under mild and controllable conditions. This not only simplifies the post-reaction workup procedures but also drastically improves the yield coefficient. For supply chain heads, this represents a transition from a fragile, low-yield process to a robust, industrialization-ready platform that guarantees commercial scale-up of complex amides with greater predictability.
Mechanistic Insights into Malonate-Based Synthesis and Reduction
The core of this technological advancement lies in the precise manipulation of the malonate ester functionality. The first critical step involves the selective hydrolysis of the diester to a mono-ester under alkaline conditions. By carefully controlling the molar ratio of mineral alkali (such as potassium hydroxide or sodium hydroxide) to the substrate between 1:1 and 1:5, and maintaining temperatures between -5°C and 40°C, the reaction selectively cleaves one ester bond while leaving the other intact. This selectivity is paramount; over-hydrolysis would lead to the di-acid, a dead-end byproduct, while under-hydrolysis leaves unreacted starting material. The subsequent acylation with thionyl chloride converts the carboxylic acid into a highly reactive acid chloride intermediate without affecting the remaining ester group. This activated species then undergoes nucleophilic attack by N-ethyl-4-picoline amine to form the amide backbone. Finally, the reduction of the remaining ester group to a primary alcohol using sodium borohydride completes the molecular architecture. This sequence demonstrates a masterful application of chemoselectivity, ensuring that functional groups are transformed in the exact order required to maintain structural integrity.
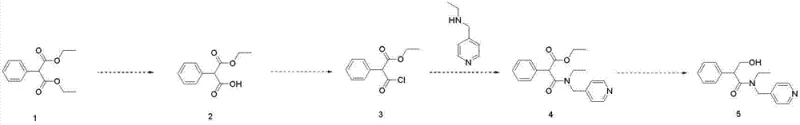
From an impurity control perspective, this mechanism offers distinct advantages over the traditional tropic acid route. In the legacy method, the instability of the protected intermediate often leads to racemization or elimination byproducts that are structurally similar to the final API, making them difficult to remove. In the malonate route, the intermediates are chemically stable solids or oils that can be easily characterized and purified if necessary. Furthermore, the final reduction step using sodium borohydride in alcoholic solvents (methanol or ethanol) is highly specific for esters in the presence of amides, minimizing the risk of over-reduction or amide cleavage. This inherent selectivity results in a crude product with a much cleaner impurity profile, reducing the burden on final crystallization steps. For R&D directors focused on high-purity tropicamide specifications, this mechanistic clarity provides the confidence needed to validate the process for regulatory filings.
How to Synthesize Tropicamide Efficiently
The implementation of this synthesis route requires strict adherence to the optimized reaction parameters defined in the patent to maximize efficiency. The process begins with the hydrolysis of phenyl ethyl malonate in a mixed solvent system of alcohol and water, followed by pH adjustment to isolate the mono-ester. Subsequent steps involve the activation of the acid and coupling with the amine component, culminating in the borohydride reduction. Each stage has been engineered to operate at near-ambient temperatures, reducing energy consumption and safety risks associated with exothermic runaways. While the general flow is straightforward, the specific stoichiometry and temperature controls are critical for success. The detailed standardized synthesis steps, including exact reagent grades and workup procedures, are outlined in the technical guide below.
- Selective hydrolysis of phenyl ethyl malonate under alkaline conditions to form the mono-ester intermediate.
- Conversion of the mono-ester to an acid chloride using thionyl chloride followed by condensation with N-ethyl-4-picoline amine.
- Final reduction of the ester group to a hydroxyl group using sodium borohydride to yield the final tropicamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of the malonate-based synthesis route offers compelling economic and operational benefits that extend beyond simple yield improvements. The primary advantage is the elimination of the protection-deprotection cycle, which traditionally adds at least two synthetic steps and consumes significant amounts of reagents and solvents. By removing these non-value-added steps, the overall material throughput is increased, and the consumption of auxiliary chemicals is drastically reduced. This streamlining directly contributes to cost reduction in pharmaceutical manufacturing by lowering the variable cost per kilogram of the final API. Additionally, the use of commodity chemicals like phenyl ethyl malonate, thionyl chloride, and sodium borohydride ensures that raw material sourcing is stable and less susceptible to market volatility compared to specialized chiral protecting groups.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the synthetic tree. By avoiding the use of expensive protecting group reagents and the associated labor for installation and removal, the direct manufacturing costs are significantly lowered. Furthermore, the higher yield coefficient means that less raw material is required to produce the same amount of finished goods, effectively amplifying the purchasing power of the procurement budget. The simplified workup procedures also reduce solvent recovery costs and waste treatment expenses, creating a leaner and more cost-efficient production model that enhances overall margin potential without compromising quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that rely on unstable intermediates which cannot be stockpiled. The intermediates in this new route, particularly the mono-ester and the amide-ester, are stable enough to be isolated and stored if necessary, providing flexibility in production scheduling. This stability allows manufacturers to build buffer stocks of key intermediates, thereby reducing lead time for high-purity pharmaceutical intermediates during periods of surge demand. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility supplies (such as cooling water temperature), ensuring consistent output even in varying operational environments.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant often reveals hidden hazards, but this route is inherently safe due to its mild thermal profile. The reactions proceed efficiently at temperatures ranging from 0°C to 40°C, eliminating the need for energy-intensive heating or deep cryogenic cooling. From an environmental standpoint, the reduction in step count directly correlates to a lower E-factor (mass of waste per mass of product). The avoidance of heavy metal catalysts or toxic protecting group byproducts simplifies effluent treatment, making it easier for facilities to meet stringent environmental regulations. This alignment with green chemistry principles future-proofs the supply chain against tightening regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced tropicamide synthesis method. These answers are derived directly from the experimental data and beneficial effects described in patent CN103159672A, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion.
Q: Why is the phenyl ethyl malonate route superior to the traditional tropic acid method?
A: The traditional method relies on unstable protected intermediates derived from tropic acid, leading to significant impurity formation and purification difficulties. The malonate route avoids overprotection strategies, utilizing stable starting materials and mild reaction conditions that significantly enhance overall yield and product purity.
Q: What are the critical control points in the hydrolysis step of this new process?
A: The hydrolysis of diethyl phenylmalonate requires precise control of pH and temperature (-5 to 40°C) to ensure selective formation of the mono-ester without over-hydrolysis to the di-acid. Using mineral alkalis like potassium hydroxide in alcohol-water solvents allows for tunable reactivity that maximizes the yield of the key intermediate.
Q: How does this synthesis method impact commercial scalability?
A: By eliminating complex protection and deprotection sequences, the process reduces the total number of unit operations and solvent usage. The use of common reagents like thionyl chloride and sodium borohydride, combined with ambient or mild temperature requirements, makes the process highly adaptable for large-scale commercial production without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tropicamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively analyzed the malonate-based pathway and possesses the technical capability to execute this process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in actual manufacturing output. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required by international pharmacopoeias.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and discuss route feasibility assessments tailored to your project timelines. Let us help you secure a sustainable and cost-effective supply of high-quality tropicamide.
