Advanced Synthesis of 3,4,5-Tribenzyloxyaniline: A Commercial Scale-Up Strategy for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of 3,4,5-Tribenzyloxyaniline: A Commercial Scale-Up Strategy for High-Purity Pharmaceutical Intermediates
The chemical landscape for producing critical aromatic amines is undergoing a significant transformation, driven by the urgent need for cleaner, more efficient synthetic routes that align with modern environmental standards. Patent CN100500643C introduces a robust and economically viable methodology for the synthesis of 3,4,5-tribenzyloxyaniline, a pivotal intermediate widely utilized in the manufacture of anti-anginal medications such as Khellin. This innovative approach departs from conventional nitro-reduction pathways, instead leveraging a sophisticated three-step sequence comprising Williamson ether synthesis, ammonolysis, and a highly selective Hofmann rearrangement. By meticulously optimizing reaction conditions and reagent ratios, this technology delivers a product with exceptional purity levels exceeding 98%, effectively addressing the persistent challenges of impurity profiles and waste generation that have long plagued the fine chemical industry. For global procurement leaders and R&D directors, this patent represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality while adhering to stringent regulatory frameworks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aromatic amines like 3,4,5-tribenzyloxyaniline has relied heavily on the reduction of corresponding nitro compounds, a process fraught with significant technical and environmental drawbacks. Traditional methods often employ metal reduction in acidic or alkaline systems, which generates substantial quantities of hazardous waste sludge and wastewater, complicating post-processing and disposal logistics considerably. Furthermore, chemical reduction techniques utilizing agents such as sodium borohydride or hydrazine hydrate frequently suffer from low reduction efficiency and exorbitant reagent costs, rendering them economically unfeasible for large-scale operations. Catalytic hydrogenation, while cleaner in theory, demands expensive catalyst preparation, high-pressure equipment investments, and is notoriously susceptible to catalyst poisoning by sulfides present in hydrogen sources. These legacy processes not only inflate production costs but also compromise product quality, often yielding deeply colored products that require extensive and costly purification steps to meet pharmaceutical grade specifications.
The Novel Approach
In stark contrast to these archaic methodologies, the novel approach detailed in the patent utilizes a Hofmann rearrangement strategy that fundamentally alters the economic and environmental equation of aniline synthesis. By starting from methyl 3,4,5-trihydroxybenzoate and proceeding through a stable amide intermediate, the process bypasses the formation of nitro groups entirely, thereby eliminating the need for hazardous reduction steps. The use of sodium hypochlorite as the oxidizing agent in the rearrangement step provides a mild yet highly effective means of converting the amide to the amine, operating under conditions that are far less aggressive than traditional catalytic hydrogenation. This shift not only simplifies the operational workflow by removing the requirement for high-pressure reactors but also ensures that the final product emerges with superior clarity and minimal impurity burden. Consequently, this method facilitates cost reduction in pharma intermediates manufacturing by streamlining the purification process and drastically reducing the volume of toxic effluent requiring treatment.
Mechanistic Insights into Hofmann Rearrangement and Selective Oxidation
The core chemical innovation of this synthesis lies in the precise execution of the Hofmann rearrangement, a reaction that converts a primary amide into a primary amine with one fewer carbon atom, although in this specific context, it serves to transform the functional group while maintaining the aromatic core integrity through an isocyanate intermediate. The mechanism initiates with the deprotonation of the amide nitrogen by sodium hydroxide, followed by N-chlorination using sodium hypochlorite to form an N-chloroamide species. Under basic conditions, this intermediate undergoes a concerted rearrangement where the aryl group migrates from the carbonyl carbon to the nitrogen atom, expelling a chloride ion and forming a highly reactive isocyanate. This isocyanate is subsequently hydrolyzed in the presence of excess base to yield the unstable carbamic acid, which spontaneously decarboxylates to release the final 3,4,5-tribenzyloxyaniline product. The elegance of this mechanism is its inherent selectivity; unlike non-specific reducing agents that might attack other sensitive functional groups on the molecule, the Hofmann rearrangement targets the amide moiety exclusively, preserving the delicate benzyl ether protections intact throughout the transformation.
Controlling the reaction parameters during this rearrangement is paramount to suppressing the formation of urea by-products, which can occur if the isocyanate reacts with the amine product before hydrolysis is complete. The patent specifies maintaining a low temperature range of 0-5°C during the initial addition of sodium hypochlorite to control the exothermic nature of the chlorination and prevent side reactions. Following the formation of the carbamate intermediate, the protocol dictates a subsequent hydrolysis step at elevated temperatures, typically around 80°C, to ensure complete conversion without isolating the intermediate. This telescoped approach minimizes material handling losses and exposure to potentially unstable intermediates, thereby enhancing the overall mass balance of the process. The result is a highly pure crystalline product that requires minimal downstream processing, validating the method's suitability for producing high-purity pharmaceutical intermediates destined for sensitive therapeutic applications.
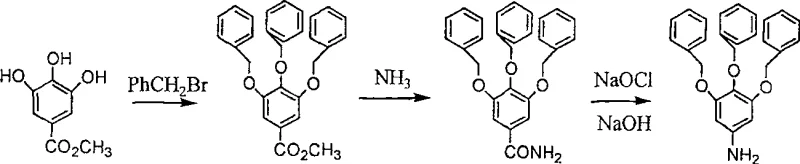
How to Synthesize 3,4,5-Tribenzyloxyaniline Efficiently
Implementing this synthesis route on a commercial scale requires strict adherence to the optimized stoichiometry and thermal profiles established in the patent data to ensure reproducibility and safety. The process begins with the protection of the phenolic hydroxyl groups via Williamson ether synthesis, utilizing benzyl bromide and a base like potassium carbonate in a polar aprotic solvent, followed by the conversion of the ester to an amide using ammonia gas in ethylene glycol. The final and most critical stage involves the careful addition of sodium hypochlorite to the amide solution under controlled cooling, followed by a prolonged hydrolysis period to drive the rearrangement to completion. Operators must monitor the disappearance of the carbamate intermediate closely, typically via TLC or HPLC, to determine the precise endpoint for hydrolysis, ensuring that no starting material remains before proceeding to isolation. Detailed standardized operating procedures regarding reagent addition rates, stirring speeds, and crystallization protocols are essential for maintaining batch-to-batch consistency.
- Perform Williamson ether synthesis by reacting methyl 3,4,5-trihydroxybenzoate with benzyl bromide in an inert solvent like DMF or acetone using potassium carbonate as a base.
- Convert the resulting methyl ester to the corresponding amide by passing ammonia gas into an ethylene glycol solution of the ester at elevated temperatures.
- Execute Hofmann rearrangement by treating the amide with sodium hypochlorite and sodium hydroxide in methanol, followed by hydrolysis to yield the final aniline product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthesis methodology offers profound advantages in terms of raw material availability, operational safety, and logistical simplicity. The reagents employed, such as sodium hypochlorite, sodium hydroxide, and benzyl bromide, are commodity chemicals available globally in bulk quantities, mitigating the risk of supply disruptions often associated with specialized catalysts or exotic reducing agents. Furthermore, the elimination of heavy metal catalysts removes the necessity for complex and costly metal scavenging steps, which are typically required to meet strict residual metal limits in pharmaceutical ingredients. This simplification of the downstream processing train directly translates to shorter production cycles and reduced utility consumption, as there is no need for high-pressure hydrogenation equipment or extensive filtration systems for metal removal. For procurement managers, this means securing a more resilient supply chain with lower exposure to volatile pricing of noble metals and reduced dependency on specialized waste disposal vendors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalytic systems and complex reduction protocols with inexpensive, readily available oxidants and bases. By avoiding the use of precious metal catalysts like palladium or platinum, which are subject to significant market price fluctuations, manufacturers can stabilize their cost of goods sold and improve margin predictability. Additionally, the high selectivity of the Hofmann rearrangement minimizes the formation of difficult-to-remove impurities, thereby reducing the consumption of solvents and adsorbents during the purification phase. The ability to telescope the final rearrangement and hydrolysis steps without isolating the intermediate further reduces labor costs and equipment occupancy time, leading to substantial overall cost efficiencies in the production of complex aromatic amines.
- Enhanced Supply Chain Reliability: The reliance on common industrial chemicals rather than bespoke reagents ensures that production schedules are not held hostage by the lead times of niche suppliers. Sodium hypochlorite and caustic soda are produced at massive scales for various industries, guaranteeing a steady flow of materials even during periods of global supply chain stress. Moreover, the robustness of the reaction conditions, which do not require stringent exclusion of moisture or oxygen to the same degree as organometallic catalysis, allows for more flexible manufacturing windows. This operational flexibility enables suppliers to respond more rapidly to fluctuating demand signals from downstream pharmaceutical clients, effectively reducing lead time for high-purity pharmaceutical intermediates and strengthening the overall reliability of the supply network.
- Scalability and Environmental Compliance: Scaling this process from laboratory to multi-ton production is facilitated by the absence of exothermic hazards associated with catalytic hydrogenation and the use of aqueous workups that are easier to manage than organic extractions. The process generates significantly less hazardous waste, particularly avoiding the heavy metal sludge that characterizes traditional nitro reductions, which simplifies compliance with increasingly stringent environmental regulations. This 'green' profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity, a factor of growing importance to multinational corporations auditing their supply chains. The straightforward isolation of the product via filtration and washing further supports seamless commercial scale-up of complex pharmaceutical intermediates without the need for specialized containment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 3,4,5-tribenzyloxyaniline, based on the specific technological advantages outlined in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own drug substance manufacturing workflows. The answers provided reflect the consensus on best practices for handling the specific reagents and conditions required for the Hofmann rearrangement pathway.
Q: What are the primary advantages of using Hofmann rearrangement over nitro reduction for synthesizing 3,4,5-tribenzyloxyaniline?
A: The Hofmann rearrangement method described in patent CN100500643C offers significantly higher selectivity and avoids the generation of heavy metal waste and colored impurities often associated with traditional nitro reduction methods, resulting in a final product purity exceeding 98%.
Q: Which solvents are recommended for the initial etherification step in this synthesis route?
A: The patent specifies the use of inert organic solvents such as N,N-dimethylformamide (DMF) or acetone for the Williamson ether synthesis step, ensuring efficient reaction kinetics and ease of subsequent workup.
Q: How does this synthesis method impact environmental compliance and waste management?
A: By utilizing sodium hypochlorite instead of heavy metal catalysts or harsh reducing agents, this process eliminates the production of toxic waste slag and废气 (waste gas), thereby simplifying wastewater treatment and enhancing overall environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Tribenzyloxyaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the Hofmann rearrangement requires a partner with deep technical expertise and proven manufacturing capabilities. Our facility is equipped to handle the commercial scale-up of complex pharmaceutical intermediates, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our state-of-the-art rigorous QC labs, ensuring that every batch of 3,4,5-tribenzyloxyaniline meets the exacting standards required for API synthesis. Our commitment to process optimization allows us to deliver this critical intermediate with the consistency and reliability that global pharmaceutical supply chains demand.
We invite you to engage with our technical team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this cleaner, more efficient manufacturing method. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production needs, ensuring a seamless integration of this high-value intermediate into your supply chain.
