Scalable Synthesis of Alpha-Murine Cholic Acid: A Technical Breakthrough for Pharmaceutical Intermediates
Scalable Synthesis of Alpha-Murine Cholic Acid: A Technical Breakthrough for Pharmaceutical Intermediates
The pharmaceutical and biochemical research sectors are increasingly demanding high-purity bile acid derivatives for metabolic studies and therapeutic applications. A significant advancement in this domain is detailed in Chinese Patent CN113336818A, which discloses a robust preparation method for alpha-murine cholic acid (α-MCA). This patent outlines a sophisticated synthetic route that transforms 3α, 6α, 7α-trihydroxy-5β-cholanic acid, commonly known as hyocholic acid, into the target molecule through a series of precise chemical transformations. The process encompasses carboxyl esterification, selective acetylation of hydroxyl groups, oxidation of the C6 position, stereoselective reduction, and final hydrolysis. This technical evolution represents a pivotal shift from traditional extraction methods, offering a reliable alpha-murine cholic acid supplier pathway that ensures consistent quality and supply continuity for global research institutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of murine cholic acid has been fraught with significant logistical and chemical challenges that hinder large-scale application. The primary conventional method involves direct extraction from animal bile, specifically from rats; however, this approach is severely constrained by the limited biological availability of the raw material. The extraction process is not only labor-intensive and troublesome but also yields inconsistent quantities that cannot meet the growing demands of modern pharmacological research. Alternatively, artificial synthesis routes starting from chenodeoxycholic acid or hyodeoxycholic acid have been employed, yet these pathways suffer from inherent chemical inefficiencies. Introducing hydroxyl groups specifically at the 6-position or 7-position carbon atoms on the steroid nucleus is chemically arduous, often requiring harsh conditions that lead to multiple side reactions. Consequently, these older synthetic methods typically exhibit low conversion rates and poor selectivity, rendering them unsuitable for mass production and resulting in prohibitively high costs for the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to the limitations of extraction and de novo synthesis, the novel approach presented in the patent leverages the structural proximity of hyocholic acid to the target molecule to streamline the manufacturing process. By utilizing hyocholic acid as the starting material, the synthesis bypasses the most difficult step of hydroxyl introduction, as the starting material already possesses the necessary trihydroxy framework. The innovation lies in the strategic manipulation of the C6 stereochemistry rather than building the skeleton from scratch. This method employs a sequence of protection, oxidation, and reduction that is both chemically elegant and operationally simple. The reaction conditions are mild, side reactions are minimized, and the conversion rate is exceptionally high, making this route ideally suited for the commercial scale-up of complex pharmaceutical intermediates. This shift allows for cost reduction in bile acid intermediate manufacturing by maximizing atom economy and minimizing waste generation associated with failed synthetic attempts.
Mechanistic Insights into Stereoselective Oxidation and Reduction
The core chemical ingenuity of this process resides in the precise control of stereochemistry at the C6 position of the cholanic acid backbone. The transformation begins with the protection of the carboxylic acid and the 3,7-dihydroxy groups to prevent unwanted side reactions during the critical oxidation step. The subsequent oxidation of the 6-alpha hydroxyl group to a ketone using Jones reagent (chromium trioxide in sulfuric acid) is a pivotal moment in the synthesis. This step effectively erases the original stereochemical information at C6, creating a planar carbonyl intermediate that is primed for stereoselective reconstruction. The choice of Jones reagent is critical here, as it provides the necessary oxidative power to convert the secondary alcohol to a ketone without degrading the sensitive steroid ring system, ensuring that the intermediate retains its structural integrity for the subsequent reduction phase.
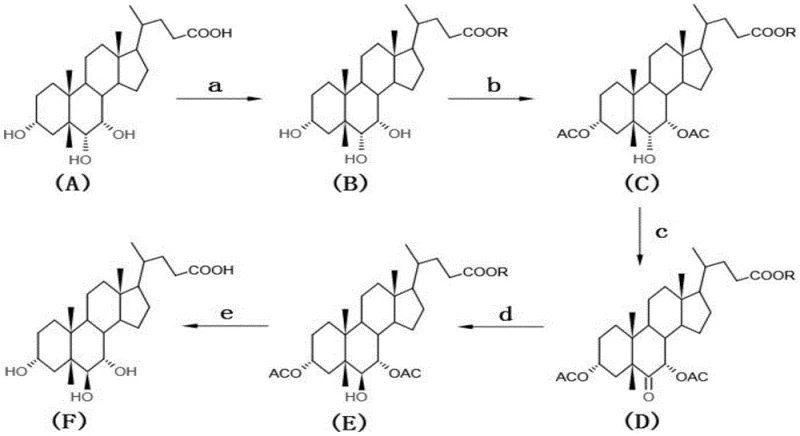
Following the oxidation, the stereoselective reduction of the C6 ketone is executed using sodium borohydride in methanol at controlled low temperatures, typically below 0°C. This step is the determinant of the final product's biological identity, as the hydride attack must occur from the alpha face to yield the desired 6-beta hydroxyl configuration found in alpha-murine cholic acid. The low temperature is essential to kinetically favor the formation of the equatorial 6-beta alcohol over the axial 6-alpha isomer, leveraging the steric environment of the steroid nucleus. Following this inversion, the protecting groups are removed via alkaline hydrolysis, restoring the free hydroxyl and carboxylic acid functionalities. The entire mechanism is designed to minimize impurity profiles, ensuring that the final crude product requires only simple recrystallization to achieve high-purity alpha-murine cholic acid specifications suitable for sensitive biological assays.
How to Synthesize Alpha-Murine Cholic Acid Efficiently
The synthesis protocol described in the patent offers a clear, step-by-step methodology for producing high-quality alpha-murine cholic acid suitable for laboratory and pilot-scale operations. The process is designed to be robust, utilizing standard organic synthesis techniques that can be easily implemented in equipped facilities. The following guide summarizes the operational flow derived from the patent examples, highlighting the critical parameters for success. For detailed standardized synthesis steps and specific safety protocols, please refer to the guide below.
- Perform carboxyl esterification of hyocholic acid using methanol or ethanol with an acid catalyst at 60-80°C to protect the carboxylic acid group.
- Execute selective acetylation of the 3 and 7-position hydroxyl groups using acetic anhydride and DMAP at temperatures below 0°C.
- Oxidize the 6-position hydroxyl group to a ketone using Jones reagent, followed by stereoselective reduction with sodium borohydride to invert the stereochemistry to the 6-beta configuration.
- Complete the synthesis via alkaline hydrolysis to remove protecting groups, followed by recrystallization from methanol to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages over traditional sourcing models. The shift from animal extraction to a defined chemical synthesis fundamentally alters the risk profile of the supply chain, moving it from a biologically dependent variable to a controllable industrial process. This transition ensures that supply continuity is no longer held hostage by the seasonal or biological limitations of raw bile collection. Furthermore, the simplicity of the reaction sequence means that the manufacturing process can be scaled up rapidly to meet surging demand without the need for specialized or exotic equipment, thereby enhancing the overall resilience of the supply network for critical research materials.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the efficiency of the raw material utilization and the high yield of the reaction steps. By starting with hyocholic acid, which is more readily available and less expensive than attempting to synthesize the steroid nucleus de novo, the input costs are significantly optimized. Additionally, the elimination of complex purification steps required to separate numerous side products—common in older synthesis methods—drastically reduces solvent consumption and waste disposal costs. The high conversion rates reported in the patent examples imply that less raw material is wasted, leading to substantial cost savings in the overall production budget without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: Reliability in the supply of specialized intermediates is paramount for uninterrupted research and development timelines. This synthetic method utilizes common, commercially available reagents such as methanol, acetic anhydride, and sodium borohydride, which are not subject to the same supply bottlenecks as rare biological extracts. The robustness of the chemistry means that production batches are highly reproducible, reducing the likelihood of batch failures that could delay downstream projects. This predictability allows procurement teams to forecast inventory needs with greater accuracy and secure long-term contracts with confidence, knowing that the manufacturing technology is stable and scalable.
- Scalability and Environmental Compliance: From an environmental and operational perspective, the process is designed with scalability in mind. The reactions are conducted in standard solvents like dichloromethane and methanol, which can be efficiently recovered and recycled in a modern chemical plant, aligning with green chemistry principles. The avoidance of heavy metal catalysts or extremely hazardous reagents simplifies the waste treatment process, ensuring easier compliance with stringent environmental regulations. This ease of handling and waste management facilitates a smoother path to commercial scale-up, allowing manufacturers to increase production volumes from kilograms to tons without encountering significant regulatory or technical barriers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of alpha-murine cholic acid based on the patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers.
Q: What is the primary advantage of using hyocholic acid as a starting material for alpha-murine cholic acid?
A: Using hyocholic acid eliminates the difficult and low-yield step of introducing hydroxyl groups at the 6 or 7 positions from scratch. Since hyocholic acid already possesses the required trihydroxy framework, the process only requires stereochemical inversion at the C6 position, significantly simplifying the synthetic route and improving overall yield.
Q: How is the stereochemistry at the C6 position controlled in this synthesis?
A: The process utilizes a two-step oxidation-reduction sequence. First, the 6-alpha hydroxyl group of hyocholic acid is oxidized to a ketone using Jones reagent. Subsequently, sodium borohydride reduction is performed under controlled low-temperature conditions (below 0°C), which favors the formation of the thermodynamically stable 6-beta hydroxyl configuration required for alpha-murine cholic acid.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the patent explicitly states the method is suitable for mass preparation. The reactions utilize common industrial reagents such as methanol, acetic anhydride, and sodium borohydride, and avoid exotic catalysts. The purification relies on standard recrystallization techniques, making the process highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Murine Cholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in advancing pharmaceutical research and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to implement complex synthetic routes, such as the stereoselective oxidation-reduction sequence described in CN113336818A, positions us as a strategic partner capable of solving your most challenging supply chain needs.
We invite you to collaborate with us to optimize your sourcing strategy for bile acid derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your development timeline and reduce your overall operational costs.
