Advanced Synthesis of Rivaroxaban Intermediate via Efficient Isonitrification and Epoxidation
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing critical intermediates, particularly for high-volume anticoagulants like Rivaroxaban. A recent breakthrough detailed in patent CN114560853A introduces a superior preparation method for 5-chloro-N-[(2S)-epoxy-2-yl-methyl]thiophene-2-formamide, a pivotal building block in the manufacturing of Factor Xa inhibitors. This innovative approach fundamentally shifts the synthetic strategy by utilizing 1-amino-3,3-diethoxypropane as a cost-effective starting material, bypassing the traditional reliance on expensive chiral pool reagents. By streamlining the synthesis into three highly efficient steps—isocyanation, coupling, and epoxidation—this technology offers a compelling value proposition for global supply chains seeking to optimize both cost and environmental footprint. For R&D directors and procurement specialists, understanding the mechanistic nuances of this route is essential for evaluating its potential to enhance the reliability of API intermediate sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of this key thiophene-based intermediate has been plagued by significant economic and environmental inefficiencies inherent in legacy synthetic routes. As illustrated in prior art such as US2010/184767, traditional methods typically initiate with 2-chlorothiophene-5-carboxylic acid, necessitating a cumbersome sequence of acyl chlorination followed by amidation with costly chiral amines. 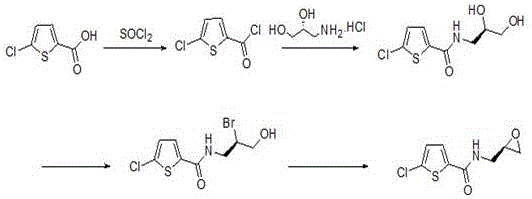 The reliance on (S)-3-amino-1,2-propanediol hydrochloride as a chiral starter imposes a heavy financial burden due to the high market price of this specialized reagent. Furthermore, subsequent steps often involve hazardous bromination reactions to install the leaving group required for epoxide formation, generating substantial amounts of toxic waste and requiring complex purification protocols. Alternative routes, such as those disclosed in EP2354128, attempt to mitigate some issues by using sulfonic acid esterification, yet they still suffer from high raw material costs and generate significant three-waste pollution, complicating regulatory compliance and increasing the total cost of ownership for manufacturers.
The reliance on (S)-3-amino-1,2-propanediol hydrochloride as a chiral starter imposes a heavy financial burden due to the high market price of this specialized reagent. Furthermore, subsequent steps often involve hazardous bromination reactions to install the leaving group required for epoxide formation, generating substantial amounts of toxic waste and requiring complex purification protocols. Alternative routes, such as those disclosed in EP2354128, attempt to mitigate some issues by using sulfonic acid esterification, yet they still suffer from high raw material costs and generate significant three-waste pollution, complicating regulatory compliance and increasing the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast to these convoluted legacy processes, the methodology disclosed in CN114560853A presents a paradigm shift by decoupling the chirality introduction from the initial starting material selection. Instead of purchasing expensive chiral amines, this novel route begins with the achiral and commercially abundant 1-amino-3,3-diethoxypropane. The process cleverly constructs the chiral center in situ through a sophisticated sequence involving isonitrification and organocatalytic functionalization. 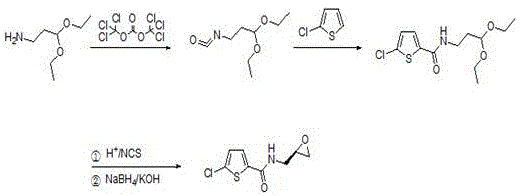 This strategic pivot not only drastically reduces the direct material costs but also simplifies the overall process flow by eliminating the need for separate chiral resolution steps early in the synthesis. By integrating the formation of the amide bond with the subsequent construction of the epoxide ring in a telescoped manner, the new method achieves a higher overall yield while minimizing the generation of hazardous byproducts, thereby offering a cleaner and more sustainable manufacturing profile.
This strategic pivot not only drastically reduces the direct material costs but also simplifies the overall process flow by eliminating the need for separate chiral resolution steps early in the synthesis. By integrating the formation of the amide bond with the subsequent construction of the epoxide ring in a telescoped manner, the new method achieves a higher overall yield while minimizing the generation of hazardous byproducts, thereby offering a cleaner and more sustainable manufacturing profile.
Mechanistic Insights into Isonitrification and Organocatalytic Epoxidation
The core innovation of this synthesis lies in the precise execution of the isonitrification step followed by a highly selective organocatalytic transformation. Initially, 1-amino-3,3-diethoxypropane reacts with a phosgene equivalent, such as triphosgene or diphosgene, under mild basic conditions to form a reactive isocyanate intermediate. This species is then coupled with the thiophene moiety to establish the critical amide linkage, preserving the acetal protection group which serves as a masked aldehyde for downstream functionalization. The subsequent hydrolysis of the acetal reveals the aldehyde functionality, setting the stage for the stereoselective construction of the epoxide ring. This sequence demonstrates remarkable chemical elegance, converting simple feedstock chemicals into a complex chiral scaffold without the need for transition metal catalysts.
Crucially, the stereochemical outcome is controlled during the chlorination and reduction phases using L-prolinamide as an organocatalyst in conjunction with N-chlorosuccinimide (NCS). This organocatalytic system facilitates the alpha-chlorination of the aldehyde intermediate with high enantioselectivity, establishing the necessary chiral center before the ring closure. Following chlorination, sodium borohydride reduction converts the chloro-aldehyde into a chlorohydrin, which spontaneously cyclizes under basic conditions to form the desired epoxide. This mechanism ensures that the final product achieves an enantiomeric excess (ee) of greater than 98%, meeting the stringent purity specifications required for pharmaceutical applications. The avoidance of heavy metal catalysts in this key step further simplifies the purification process, reducing the risk of metal contamination in the final API.
How to Synthesize 5-Chloro-N-[(2S)-Epoxypropyl]thiophene-2-formamide Efficiently
Implementing this advanced synthetic route requires careful attention to reaction conditions, particularly temperature control and reagent stoichiometry, to maximize yield and optical purity. The process is designed to be robust, utilizing common organic solvents such as dichloromethane, ethanol, and tetrahydrofuran, which facilitates easy adoption in existing multipurpose chemical plants. The following guide outlines the critical operational parameters derived from the patent examples, ensuring that technical teams can replicate the high efficiency reported in the literature.
- Perform isonitrification of 1-amino-3,3-diethoxypropane using triphosgene to generate the reactive isocyanate intermediate.
- Couple the isocyanate intermediate with the chlorinated thiophene moiety to form the protected amide precursor.
- Execute hydrolysis followed by organocatalytic chlorination and base-mediated cyclization to form the final chiral epoxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple unit price reductions. By shifting the raw material base from specialized chiral amines to commodity chemicals like 1-amino-3,3-diethoxypropane, manufacturers can significantly insulate their supply chains from the volatility associated with niche fine chemical markets. This diversification of the supply base enhances continuity and reduces the risk of production stoppages caused by shortages of expensive starting materials. Furthermore, the reduction in synthetic steps from four or more down to three directly correlates with lower operational expenditures, as fewer unit operations mean reduced labor, energy, and solvent consumption per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials represents a major driver for cost optimization in API intermediate manufacturing. Traditional routes rely heavily on (S)-3-amino-1,2-propanediol derivatives, which command a premium price due to their complex synthesis; replacing these with achiral precursors allows for substantial savings in direct material costs. Additionally, the removal of hazardous bromination and sulfonic acid esterification steps reduces the need for specialized corrosion-resistant equipment and expensive waste disposal services. The overall simplification of the process flow leads to a leaner manufacturing operation with improved throughput and lower overheads.
- Enhanced Supply Chain Reliability: Sourcing stability is markedly improved as the new route depends on widely available bulk chemicals rather than limited-supply specialty reagents. 1-amino-3,3-diethoxypropane and standard thiophene derivatives are produced at scale by multiple global suppliers, ensuring a competitive marketplace and consistent availability. This redundancy in the supply base mitigates the risk of single-source dependency, allowing procurement teams to negotiate better terms and secure long-term contracts with greater confidence. The robustness of the chemistry also means that production schedules are less susceptible to delays caused by reagent quality fluctuations.
- Scalability and Environmental Compliance: The streamlined nature of this three-step process inherently supports easier scale-up from pilot plant to commercial tonnage production. With fewer isolation and purification stages, the cumulative yield loss is minimized, and the physical footprint required for manufacturing is reduced. From an environmental perspective, the avoidance of heavy metals and the reduction in halogenated waste streams align perfectly with increasingly stringent global environmental regulations. This green chemistry advantage not only lowers compliance costs but also enhances the corporate sustainability profile of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The novel route eliminates the need for expensive chiral starting materials like (S)-3-amino-1,2-propanediol hydrochloride, significantly reducing raw material costs. Additionally, it avoids hazardous bromination and sulfonic acid esterification steps, resulting in lower environmental impact and simplified waste treatment.
Q: How does the process ensure high enantiomeric excess (ee) for the final product?
A: The process utilizes L-prolinamide as an organocatalyst during the critical functionalization step, combined with precise temperature control and specific reduction conditions using sodium borohydride. This ensures the stereochemical integrity is maintained, achieving ee values exceeding 98%.
Q: Is this manufacturing process scalable for commercial API production?
A: Yes, the synthesis relies on robust, commodity-grade reagents such as 1-amino-3,3-diethoxypropane and standard solvents like dichloromethane and ethanol. The reduction in step count from four or more to just three main stages enhances operational efficiency and facilitates seamless scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-N-[(2S)-Epoxypropyl]thiophene-2-formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory technologies like the one described in CN114560853A can be successfully translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-chloro-N-[(2S)-epoxy-2-yl-methyl]thiophene-2-formamide meets the exacting standards required for downstream API synthesis. Our commitment to quality assurance ensures that our partners receive intermediates that facilitate smooth and efficient final drug substance manufacturing.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced technology for their Rivaroxaban supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your operations.
