Advanced Solvent-Free Synthesis of Tenofovir Alafenamide Impurity TAF-Y for Global Pharma Supply Chains
Advanced Solvent-Free Synthesis of Tenofovir Alafenamide Impurity TAF-Y for Global Pharma Supply Chains
The pharmaceutical industry's relentless pursuit of higher safety standards and regulatory compliance has placed a premium on the availability of high-purity reference standards and impurities for critical antiviral medications. Patent CN109265480B, published in August 2021, introduces a groundbreaking preparation method for Tenofovir Alafenamide (TAF) impurity TAF-Y, addressing significant bottlenecks in the current supply chain for quality control materials. Tenofovir Alafenamide, a prodrug of tenofovir, has become a cornerstone in the treatment of chronic hepatitis B and HIV due to its superior plasma stability and reduced renal toxicity compared to its predecessor, Tenofovir Disoproxil Fumarate. As global demand for TAF continues to surge, the ability to accurately quantify and control related substances, such as the diphenyl phosphate impurity TAF-Y, is paramount for meeting stringent ICH guidelines. This technical insight report analyzes the novel synthetic pathway disclosed in the patent, highlighting its potential to revolutionize the manufacturing of this critical reference standard through enhanced efficiency and purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 9-[(R)-2-[[(S)-[di-(phenoxy) phosphinyl] methoxy] propyl] adenine, commonly known as TAF-Y, was plagued by inefficiencies that hindered scalable production. Existing literature, such as patent CN105330700A, describes routes that suffer from critically low yields, necessitating complex and costly purification techniques like column chromatography to achieve acceptable purity levels. Furthermore, alternative methods reported in documents like WO2015/161781A1 rely heavily on the use of dichloromethane as a reaction solvent and require the addition of acid-binding agents such as triethylamine. These conventional approaches not only introduce significant environmental and safety hazards associated with volatile organic compounds but also result in low reaction conversion rates and the formation of numerous side products. The reliance on column chromatography is particularly detrimental in a commercial setting, as it creates a massive bottleneck in throughput, generates substantial silica gel waste, and consumes excessive amounts of elution solvents, thereby driving up the cost of goods sold and extending lead times for procurement teams.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN109265480B offers a streamlined, high-efficiency alternative that fundamentally reimagines the esterification step. The core innovation lies in the direct esterification of the chlorophosphinyl intermediate with phenol under solvent-free conditions, eliminating the need for any external acid-binding agent. By leveraging the reactants themselves to drive the equilibrium, this method achieves a remarkably high reaction conversion rate, effectively bypassing the formation of complex impurity profiles associated with traditional solvent-based systems. The resulting crude product is sufficiently pure to undergo a simple, single-step recrystallization operation using a specific mixed solvent system, yielding the final TAF-Y product with an HPLC purity exceeding 98%. This drastic simplification of the workflow not only accelerates the production cycle but also aligns perfectly with green chemistry principles by minimizing solvent usage and waste generation.
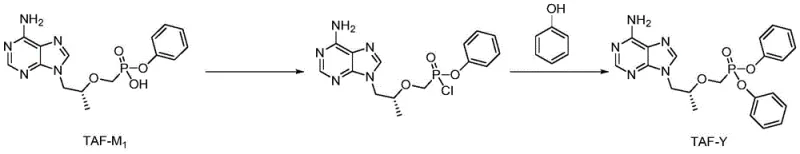
Mechanistic Insights into Solvent-Free Phosphonylation and Esterification
The chemical elegance of this new route is rooted in the precise control of the phosphorus center's reactivity. The synthesis initiates with the chlorination of the starting material, 9-[(R)-2-[[(phenoxy phosphinyl) methoxy] propyl] adenine (TAF-M1), using thionyl chloride. This step converts the P-OH group into a highly reactive P-Cl species, specifically 9-[(R)-2-[[(phenoxy chlorophosphinyl) methoxy] propyl] adenine. The use of thionyl chloride is strategic, as the byproducts (SO2 and HCl) are gaseous and can be easily removed, driving the reaction to completion and preventing the accumulation of acidic residues that could degrade the sensitive adenine moiety. Following concentration, the reactive chlorophosphinyl intermediate is immediately subjected to esterification. In a departure from standard protocols, the reaction is conducted by adding the concentrate directly into preheated phenol at 80°C. The absence of a diluting solvent increases the effective concentration of the nucleophile (phenol), facilitating a rapid nucleophilic attack on the phosphorus atom and displacing the chloride ion. This high-concentration environment suppresses competitive hydrolysis reactions that typically occur in dilute solutions, thereby maximizing the yield of the desired diphenyl phosphate ester.
Following the reaction, the workup procedure is designed to maximize recovery while ensuring high purity. The reaction mixture is quenched and extracted, but the true purification power lies in the recrystallization step. The patent specifies the use of a mixed solvent system, preferably toluene and acetonitrile in a volume ratio of 4:1. This specific solvent pairing is critical for differential solubility; it allows the target impurity TAF-Y to dissolve completely at elevated temperatures (60°C) while remaining insoluble at low temperatures (-5°C). This thermal gradient crystallization effectively excludes structurally similar byproducts and unreacted starting materials from the crystal lattice. The result is a white solid with exceptional homogeneity, as confirmed by mass spectrometry and NMR data, demonstrating that the process controls not just chemical purity but also physical form, which is essential for the stability of reference standards during long-term storage.
How to Synthesize Tenofovir Alafenamide Impurity TAF-Y Efficiently
The synthesis of TAF-Y via this novel route is operationally straightforward yet requires precise adherence to temperature and stoichiometric parameters to ensure reproducibility at scale. The process begins with the activation of the phosphonate precursor, followed by a solvent-free coupling that demands careful thermal management to prevent degradation. The final purification relies on a specific solvent ratio that must be maintained to achieve the target purity specifications. For laboratory and pilot-scale operations, strict control over the cooling rate during crystallization is essential to obtain the desired crystal habit and particle size distribution. The detailed standardized synthetic steps, including exact reagent quantities, addition rates, and safety precautions for handling thionyl chloride and phenol, are outlined in the comprehensive guide below.
- Chlorinate 9-[(R)-2-[[(phenoxy phosphinyl) methoxy] propyl]] adenine (TAF-M1) using thionyl chloride to form the chlorophosphinyl intermediate.
- Perform direct esterification by reacting the intermediate with excess phenol at 80°C without additional solvents or acid-binding agents.
- Purify the crude product via recrystallization using a mixed solvent system of toluene and acetonitrile to achieve >98% HPLC purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic advantages that extend far beyond simple technical metrics. The elimination of column chromatography represents a paradigm shift in operational efficiency, removing one of the most time-consuming and resource-intensive unit operations in fine chemical manufacturing. This reduction in processing steps directly correlates to a significant decrease in production lead times, allowing suppliers to respond more agilely to fluctuating market demands for reference standards. Furthermore, the solvent-free nature of the key esterification step drastically reduces the volume of organic waste generated, simplifying waste disposal logistics and lowering the environmental compliance burden. These factors combine to create a more resilient supply chain capable of delivering high-quality materials with greater consistency and reliability.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the removal of expensive and labor-intensive purification stages. By eliminating the need for large quantities of silica gel and elution solvents required for column chromatography, the direct variable costs associated with materials are substantially reduced. Additionally, the solvent-free esterification step minimizes the energy consumption required for solvent recovery and distillation, further driving down utility costs. The high reaction conversion rate ensures that raw material utilization is maximized, reducing the cost per gram of the final active pharmaceutical ingredient intermediate. This lean manufacturing approach allows for a more competitive pricing structure without compromising on the stringent quality requirements demanded by the pharmaceutical sector.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of synthesis routes that rely on specialized reagents or difficult-to-source solvents. This novel method utilizes commodity chemicals such as phenol, thionyl chloride, toluene, and acetonitrile, all of which are readily available in the global chemical market with stable pricing and supply. The robustness of the solvent-free reaction also reduces the risk of batch failures due to solvent quality issues or moisture contamination, which are common pitfalls in traditional phosphonylation reactions. Consequently, manufacturers can maintain higher inventory levels of finished goods with greater confidence, ensuring that downstream pharmaceutical clients never face shortages of critical quality control references.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often exposes hidden inefficiencies, particularly regarding heat transfer and mixing in large vessels. The simplified nature of this synthesis, characterized by fewer unit operations and the absence of viscous silica slurries, makes it inherently easier to scale up to multi-kilogram or tonne quantities. The reduction in solvent usage aligns perfectly with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. By generating less hazardous waste and consuming fewer resources, this process positions the manufacturer as a sustainable partner, mitigating regulatory risks and enhancing the corporate social responsibility profile of the entire supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Tenofovir Alafenamide impurity TAF-Y. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for quality assurance teams and technical buyers who need to validate the suitability of this material for their specific analytical methods.
Q: How does the novel solvent-free method improve upon conventional TAF-Y synthesis?
A: Conventional methods often rely on dichloromethane solvents and acid-binding agents like triethylamine, leading to low conversion rates and complex purification via column chromatography. The novel approach eliminates solvents during esterification, significantly increasing reaction conversion and simplifying downstream processing to a single recrystallization step.
Q: What purity levels can be achieved with this optimized synthetic route?
A: By utilizing a specific recrystallization protocol with a toluene and acetonitrile mixed solvent system, the process consistently yields TAF-Y with an HPLC purity exceeding 98%, making it highly suitable for use as a certified reference standard in quality control laboratories.
Q: Is this process suitable for large-scale commercial production of reference materials?
A: Yes, the elimination of column chromatography and the use of a solvent-free esterification step drastically reduce operational complexity and waste generation. This streamlined workflow enhances scalability, allowing for efficient production of kilogram quantities required for global supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TAF-Y Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your drug development pipeline depends on the quality of the reference standards you utilize. Our technical team has extensively analyzed the synthetic pathways described in CN109265480B and possesses the expertise to implement this advanced, solvent-free methodology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for method validation or kilograms for stability studies, our supply remains uninterrupted. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of TAF-Y meets the >98% HPLC purity benchmark required for global regulatory submissions.
We invite pharmaceutical companies and contract research organizations to collaborate with us to secure a stable supply of this critical impurity. By leveraging our optimized manufacturing processes, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, helping you reduce the overall cost of quality control without sacrificing data integrity. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and discover how our commitment to innovation can support your long-term strategic goals in antiviral drug development.
