Advanced Copper-Catalyzed Synthesis of Beta-Thioindolone Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of Beta-Thioindolone Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic methodologies that balance high purity with operational safety, particularly when constructing complex heterocyclic scaffolds essential for anticancer and anti-inflammatory agents. Patent CN110256496B introduces a groundbreaking approach for the preparation of beta-thioindolone derivatives and related phosphonate intermediates, addressing critical limitations found in legacy synthetic routes. This technology leverages a copper-catalyzed radical addition strategy, utilizing readily available alkynes, phosphorus reagents, and trimethylsilyl isothiocyanate (TMSNCS) under remarkably mild thermal conditions ranging from 30°C to 70°C. By shifting away from hazardous reagents like carbon disulfide, this methodology not only enhances the safety profile of the manufacturing process but also streamlines the purification workflow, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines for high-value bioactive compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfur-containing heterocycles and diphosphonate compounds with therapeutic potential has relied heavily on precursors such as benzothiazine-2,4-dithione. These traditional pathways are fraught with significant operational hazards and inefficiencies that hinder scalable manufacturing. A primary concern is the generation and use of carbon disulfide (CS2), a highly volatile and toxic liquid that poses severe risks to human health, including damage to the nervous system and cardiovascular complications upon inhalation or skin contact. Furthermore, conventional routes often involve harsh reaction conditions, lengthy synthetic sequences, and poor selectivity, leading to complex impurity profiles that require rigorous and costly purification efforts. The difficulty in sourcing specialized starting materials combined with the environmental burden of handling toxic byproducts creates a substantial bottleneck for procurement managers and supply chain heads seeking sustainable and cost-effective production strategies for these critical drug intermediates.
The Novel Approach
In stark contrast to these archaic methods, the novel approach detailed in the patent utilizes a modular and atom-economical strategy centered on the functionalization of terminal alkynes. By employing a copper catalyst alongside peroxides, the process facilitates the efficient addition of phosphorus and thiocyanate groups across the alkyne triple bond to form beta-thiocyanato alkenyl phosphono derivatives. This transformation proceeds with high regioselectivity and yield under mild temperatures, eliminating the need for cryogenic conditions or extreme heating. The subsequent conversion of these intermediates into beta-thioindolone derivatives involves a streamlined sequence of zinc-mediated reduction and oxidative cyclization, avoiding the release of toxic gases entirely. 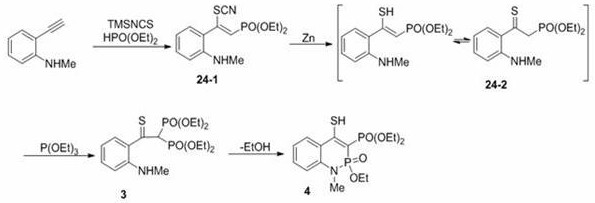 This shift not only simplifies the reaction operation but also drastically reduces the environmental footprint, aligning perfectly with modern green chemistry principles while ensuring a consistent supply of high-purity intermediates for downstream drug development.
This shift not only simplifies the reaction operation but also drastically reduces the environmental footprint, aligning perfectly with modern green chemistry principles while ensuring a consistent supply of high-purity intermediates for downstream drug development.
Mechanistic Insights into Copper-Catalyzed Radical Addition
The core of this technological advancement lies in the intricate radical mechanism driven by the copper catalyst and peroxide oxidant. The reaction initiates with the generation of phosphorus-centered radicals or thiocyanate radicals through the interaction of the copper species with the peroxide. These reactive intermediates then add across the electron-rich triple bond of the alkyne substrate, forming a vinyl radical species that is subsequently trapped by the complementary reagent to yield the beta-thiocyanato alkenyl phosphono structure. This radical pathway is highly tolerant of various functional groups, allowing for the use of diverse aryl and alkyl alkynes without compromising the integrity of sensitive moieties such as esters, halogens, or nitro groups. The versatility of this mechanism ensures that the process can be adapted to synthesize a wide library of derivatives, providing R&D directors with the flexibility needed to explore structure-activity relationships efficiently.
Furthermore, the control over impurity formation is inherent in the mildness of the reaction conditions. Unlike high-temperature processes that often promote polymerization or decomposition of sensitive intermediates, the 30-70°C range minimizes side reactions. The use of trimethylsilyl isothiocyanate as a stable source of the thiocyanate group prevents the formation of polysulfides or other sulfur-based impurities common in sulfuration reactions.  The subsequent steps, involving zinc reduction and oxidative cyclization with iodosobenzene, are equally selective, ensuring that the final beta-thioindolone products possess the stringent purity specifications required for pharmaceutical applications. This mechanistic precision translates directly into reduced downstream processing costs and higher overall process efficiency.
The subsequent steps, involving zinc reduction and oxidative cyclization with iodosobenzene, are equally selective, ensuring that the final beta-thioindolone products possess the stringent purity specifications required for pharmaceutical applications. This mechanistic precision translates directly into reduced downstream processing costs and higher overall process efficiency.
How to Synthesize Beta-Thioindolone Derivatives Efficiently
The synthesis of these valuable heterocyclic compounds follows a logical, multi-step sequence that is amenable to standard laboratory and pilot plant equipment. The process begins with the copper-catalyzed coupling of the alkyne and phosphorus reagent, followed by a reduction step to convert the thiocyanate group into a thiol or thione functionality. Finally, an oxidative cyclization closes the ring to form the indolone core. Each step has been optimized to maximize yield and minimize waste, with detailed protocols available for various substrates including substituted phenylacetylenes and alkyl alkynes. For a comprehensive understanding of the specific reaction parameters and workup procedures, please refer to the standardized guide below.
- React alkyne, phosphorus reagent, and trimethylsilyl isothiocyanate with a copper catalyst and peroxide at 30-70°C to form beta-thiocyanato alkenyl phosphono derivatives.
- Reduce the thiocyanato intermediate using a zinc catalyst and acetic acid to generate beta-thiocarbonylphosphinyl derivatives.
- Perform oxidative cyclization using iodosobenzene and tetra-n-butyl ammonium iodide to finalize the beta-thioindolone structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative advantages that directly impact the bottom line and supply chain resilience. By replacing toxic and difficult-to-handle reagents with stable, commercially available alternatives, the process significantly lowers the barrier to entry for manufacturing these complex intermediates. The elimination of carbon disulfide not only reduces regulatory compliance costs associated with hazardous waste disposal but also mitigates the risk of production shutdowns due to safety incidents. Moreover, the high yields reported in the patent examples, often exceeding 80% for key steps, indicate a robust process that maximizes raw material utilization, thereby driving down the cost of goods sold (COGS) for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic benefits of this route are substantial, primarily driven by the use of inexpensive starting materials like terminal alkynes and diethyl phosphite, which are produced on a massive industrial scale. The avoidance of exotic catalysts or ligands further reduces material costs, while the mild reaction conditions lower energy consumption compared to high-temperature or high-pressure alternatives. Additionally, the simplified post-treatment process, which often requires only standard column chromatography or extraction, reduces solvent usage and labor hours, contributing to significant overall cost savings in pharmaceutical intermediate manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the broad availability of the requisite raw materials. Alkynes and phosphorus reagents are commodity chemicals with established global supply networks, reducing the risk of shortages that can plague specialized reagents. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch reproducibility. This reliability is crucial for maintaining steady production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the simplicity of the unit operations involved. The reaction does not require specialized pressure vessels or cryogenic cooling systems, allowing for easy scale-up in standard stainless steel reactors. Furthermore, the green chemistry attributes of the process, such as the absence of toxic gas evolution and the use of less hazardous solvents, ensure compliance with increasingly stringent environmental regulations. This alignment with sustainability goals not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their own production needs.
Q: How does this new method improve safety compared to traditional synthesis?
A: Traditional methods often utilize toxic carbon disulfide (CS2), which poses severe respiratory and neurological risks. This patented process replaces CS2 with trimethylsilyl isothiocyanate (TMSNCS) and operates under mild conditions (30-70°C), significantly reducing operator exposure to hazardous volatiles.
Q: What represents the primary cost driver in this synthesis route?
A: The primary cost drivers are the alkyne starting materials and the phosphorus reagents. However, the process eliminates the need for expensive heavy metal removal steps associated with other catalytic systems and utilizes commercially available, low-cost reagents like zinc powder and copper salts, optimizing the overall cost structure.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights suitability for industrial production due to simple reaction operations, mild temperature requirements, and straightforward post-treatment processes like column chromatography or crystallization, which are easily adaptable to kilogram or ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Thioindolone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering beta-thioindolone derivatives and related phosphonate intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite procurement leaders and R&D directors to collaborate with us to leverage this innovative copper-catalyzed technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
