Advanced Manufacturing of High-Purity HCA Flame Retardant Intermediates via Optimized Solvent Systems
Advanced Manufacturing of High-Purity HCA Flame Retardant Intermediates via Optimized Solvent Systems
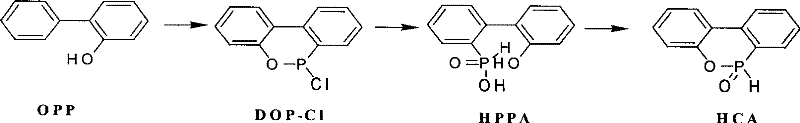
The landscape of flame retardant intermediate manufacturing is undergoing a significant transformation driven by the need for higher purity and more efficient processing protocols. Patent CN101089005B introduces a groundbreaking methodology for the preparation of cyclic phosphonate compounds, specifically targeting the synthesis of 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (HCA). This innovation addresses critical bottlenecks in the traditional production of HCA, a key precursor for high-performance halogen-free flame retardants used in epoxy resin curing agents and semiconductor encapsulation materials. By shifting from conventional non-polar solvent systems or direct thermal drying to a controlled intramolecular dehydration process utilizing polar protic solvents, this technology offers a robust pathway to achieving superior product quality. The technical breakthrough lies in the strategic selection of solvents such as methanol, ethanol, or water, which not only enhance heat transfer efficiency but also accelerate the removal of reaction by-products. For R&D directors and process engineers, this represents a pivotal opportunity to optimize impurity profiles and streamline the synthesis of complex organophosphorus structures without compromising on yield or safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of HCA has been plagued by inefficiencies inherent in older dehydration technologies. Traditional methods, such as those described in earlier patents like US6,291,626, often rely on the direct heating of wet 2-hydroxybiphenyl-2-phosphinic acid (HPPA) under reduced pressure or the use of non-polar solvents like toluene for azeotropic distillation. These approaches suffer from significant thermal inefficiencies, where uneven local heating frequently leads to the formation of undesirable oxide and organic phosphate impurities. Furthermore, alternative methods involving hot air drying in electrical furnace dryers are excessively time-consuming, often requiring dehydration cycles extending over several days to ensure complete conversion. This prolonged exposure to heat not only degrades the thermal stability of the intermediate but also results in higher residual levels of starting materials like o-phenylphenol (OPP) and elevated chloride ion content. Such impurities are detrimental when the final HCA product is intended for high-end applications such as semiconductor sealing, where ionic contamination must be minimized to prevent device failure. The reliance on specialized drying equipment further complicates the supply chain, increasing capital expenditure and limiting the flexibility of manufacturing sites to scale production rapidly.
The Novel Approach
The methodology disclosed in CN101089005B fundamentally reengineers the dehydration step by introducing a polar protic solvent system that acts as both a dispersing medium and a reaction accelerator. Instead of struggling with the poor thermal conductivity of wet HPPA cakes, the new process disperses the material in solvents like methanol, ethanol, or water, typically in amounts ranging from 20% to 60% of the HPPA weight. This modification ensures uniform heat distribution throughout the reaction mass, effectively eliminating hot spots that cause oxidative degradation. The presence of the polar solvent facilitates the rapid removal of generated water through atmospheric distillation at moderate temperatures between 60°C and 100°C, followed by a swift vacuum distillation phase at 110°C to 130°C. This dual-stage distillation strategy drastically shortens the production cycle, reducing what was once a multi-day operation into a matter of hours. Additionally, the inclusion of phenolic antioxidants, such as 2,6-di-tert-butyl-4-methylphenol (BHT), at molar ratios of 0.05% to 1% provides an extra layer of protection against oxidation, ensuring the final crystalline product maintains exceptional whiteness and chemical stability. This approach allows for the use of standard reaction kettles rather than specialized dryers, significantly lowering the barrier to entry for high-quality production.
Mechanistic Insights into Polar Protic Solvent-Assisted Cyclization
The efficacy of this novel synthesis route is rooted in the physicochemical interactions between the polar protic solvent and the phosphinic acid moiety of the HPPA intermediate. In the context of intramolecular dehydration, the choice of solvent is critical for lowering the activation energy required for the P-OH and Ar-OH groups to condense and form the stable P-O-C cyclic structure. Polar protic solvents possess high dielectric constants and the ability to form hydrogen bonds, which helps in solvating the transition state and stabilizing the charged intermediates that may form during the acid-catalyzed or thermal cyclization process. By maintaining the HPPA in a semi-dissolved or well-dispersed state, the solvent prevents the aggregation of particles that typically leads to mass transfer limitations in solid-state dehydration. As the temperature is raised to the 60-100°C range, the solvent forms an azeotrope or simply co-distills with the water produced by the condensation reaction, continuously shifting the equilibrium towards the product side according to Le Chatelier's principle. This continuous removal of water is far more efficient in a liquid phase mediated by a volatile polar solvent than in a viscous melt or solid cake, thereby accelerating the reaction kinetics significantly. The subsequent vacuum distillation step at 110-130°C ensures the removal of any remaining trace solvent and drives the reaction to completion, yielding the thermodynamically stable HCA ring system with minimal side reactions.
Impurity control is another critical aspect where the mechanistic advantages of this process shine, particularly for applications demanding ultra-low ionic content. The traditional direct heating methods often promote the oxidation of the phosphorus center, leading to phosphate by-products that are difficult to separate. By incorporating antioxidants like BHT directly into the reaction mixture, the process actively scavenges free radicals that could initiate oxidative degradation pathways. Furthermore, the washing and crystallization steps facilitated by the solvent system allow for the effective leaching of chloride ions, which originate from the initial phosphorus trichloride reactant. The patent data indicates that this method consistently achieves chloride ion levels below 50ppm, a specification that is challenging to meet with older technologies without extensive post-treatment. The residual OPP content is also tightly controlled to below 0.05%, ensuring that the final flame retardant does not introduce plasticizing impurities that could compromise the mechanical properties of the cured epoxy resin. This level of purity is essential for maintaining the reliability of electronic components where HCA derivatives are utilized as curing agents or reactive flame retardants.
How to Synthesize HCA Efficiently
The synthesis of high-purity HCA via this optimized route involves a sequential process starting from readily available raw materials, designed to maximize throughput while minimizing operational complexity. The procedure begins with the preparation of the HPPA wet product, which serves as the direct precursor for the cyclization step. Operators must ensure that the HPPA is adequately separated from the hydrolysis mother liquor to minimize the introduction of extraneous acids or salts that could interfere with the dehydration equilibrium. Once the wet cake is prepared, it is immediately transferred to the reaction vessel containing the pre-selected polar protic solvent and the antioxidant charge. The detailed standardized synthesis steps, including precise stirring rates, heating ramps, and vacuum thresholds, are outlined in the technical guide below to ensure reproducibility across different manufacturing scales.
- Prepare the HPPA wet product by hydrolyzing DOP-Cl in an organic solvent like toluene, followed by filtration and separation.
- Disperse the HPPA wet product in a polar protic solvent (methanol, ethanol, or water) comprising 20-60% of the HPPA weight, adding 0.05%-1% antioxidant (BHT).
- Perform atmospheric distillation at 60-100°C to remove solvent and water, followed by vacuum distillation at 110-130°C under -0.095Mpa to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this solvent-mediated dehydration technology translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the production infrastructure; by eliminating the need for specialized electrical furnace dryers and long-cycle hot air ovens, manufacturers can utilize standard stainless steel reactors equipped with distillation columns. This compatibility with existing general-purpose chemical equipment significantly reduces capital expenditure (CAPEX) and allows for greater flexibility in production scheduling. The reduction in cycle time from several days to merely a few hours enhances the asset turnover rate, enabling facilities to respond more agilely to market demand fluctuations without the need for massive inventory buffers. Furthermore, the use of common solvents like methanol, ethanol, or water ensures a stable and cost-effective supply chain for raw materials, avoiding the volatility associated with specialized or hazardous reagents. The ability to recover and recycle these solvents further contributes to a circular economy model within the plant, reducing waste disposal costs and environmental compliance burdens.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of energy-intensive long-duration drying cycles and the reduction of labor hours associated with monitoring extended batch times. By switching to a liquid-phase dehydration that completes in a fraction of the time, energy consumption per kilogram of product is significantly lowered, directly impacting the variable cost of goods sold. Additionally, the high selectivity of the reaction minimizes the formation of by-products, which reduces the load on downstream purification units and increases the overall yield of saleable material from the same amount of raw input. The avoidance of expensive transition metal catalysts or complex purification resins further streamlines the cost structure, making the final HCA product more competitive in the global flame retardant market. These cumulative efficiencies allow for a more robust margin structure even in the face of fluctuating raw material prices for phosphorus chemicals.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures consistent delivery schedules and reduces the risk of production bottlenecks. Traditional methods that rely on specific drying equipment often become single points of failure; if a dryer goes down, the entire production line halts. In contrast, this process can be run in multiple standard reactors in parallel, providing redundancy and ensuring business continuity. The use of widely available solvents means that procurement teams are not locked into single-source suppliers for niche reagents, mitigating the risk of supply disruptions. Moreover, the improved purity profile reduces the likelihood of customer rejections or returns due to specification failures, fostering stronger long-term relationships with downstream polymer manufacturers who rely on consistent quality for their own production lines.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the reliance on unit operations that are well-understood in the chemical industry, such as distillation and crystallization. The process generates less hazardous waste compared to methods using chlorinated solvents or producing heavy metal contaminated residues, aligning with increasingly stringent global environmental regulations. The closed-system nature of the reactor setup minimizes VOC emissions, and the potential for solvent recovery loops supports sustainability goals. This environmental friendliness not only simplifies permitting processes for new facilities but also enhances the brand value of the supplier as a responsible partner in the green chemistry initiative, which is a growing priority for multinational corporations sourcing electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced HCA synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy processes. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why are polar protic solvents preferred over toluene for HCA synthesis?
A: Polar protic solvents like methanol or water facilitate better heat dispersion and faster water removal during the dehydration step, significantly reducing reaction time and preventing local overheating that leads to oxidation impurities.
Q: What purity levels can be achieved with this novel dehydration method?
A: This method consistently achieves product purity exceeding 99%, with residual OPP content below 0.05% and chloride ion content less than 50ppm, meeting stringent requirements for semiconductor sealing materials.
Q: Does this process require specialized drying equipment?
A: No, unlike traditional methods requiring electrical furnace dryers, this process utilizes standard reaction kettles capable of atmospheric and vacuum distillation, simplifying equipment investment and maintenance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable HCA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to high-purity flame retardant intermediates requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101089005B are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify parameters such as chloride ion content and residual monomer levels. Our commitment to quality assurance means that every batch of HCA delivered meets the exacting standards required for semiconductor and high-performance polymer applications, providing our clients with the confidence to innovate without supply chain constraints.
We invite you to collaborate with us to leverage these technological advancements for your specific product needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this solvent-based synthesis route for your operations. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-purity HCA and the technical support necessary to drive your next generation of flame retardant solutions forward.
