Advanced Synthesis of Cyclopropyl-Imidazo Pyridazine Derivatives for Commercial Scale-Up
Advanced Synthesis of Cyclopropyl-Imidazo Pyridazine Derivatives for Commercial Scale-Up
The pharmaceutical industry is constantly seeking novel scaffolds that offer potent biological activity combined with synthetic accessibility. Patent CN101648953B introduces a groundbreaking class of imidazo[1,2-b]pyrrolo[1,2-f]pyridazine derivatives, specifically engineered with a cyclopropyl moiety to enhance anticancer properties. This technology represents a significant leap forward in the design of small molecule inhibitors targeting hepatocellular carcinoma (HCC). By integrating a cyclopropyl group into the indolizine-fused pyridazine system, researchers have unlocked a new avenue for developing high-purity API intermediates with superior efficacy profiles. The structural versatility of this core allows for extensive modification at the R1 and R2 positions, enabling medicinal chemists to fine-tune pharmacokinetic properties while maintaining robust antiproliferative activity against Hep-G2 cell lines.
![General chemical structure of imidazo[1,2-b]pyrrolo[1,2-f]pyridazine derivatives showing variable substituents R1 and R2](/insights/img/imidazo-pyridazine-synthesis-pharma-supplier-20260305041146-01.png)
For procurement managers and supply chain directors, the implications of this patent extend beyond mere biological potential. The synthetic route described eliminates the need for complex transition metal catalysis, which often poses significant challenges in terms of residual metal removal and regulatory compliance. Instead, the process relies on an efficient organocatalytic system using TPCD (a specialized catalyst) in a polar aprotic solvent environment. This shift not only streamlines the purification workflow but also drastically reduces the environmental footprint associated with heavy metal waste disposal. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advantages is crucial for evaluating the long-term viability and cost-effectiveness of incorporating such scaffolds into your drug development pipeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of fused heterocyclic systems often involves multi-step sequences requiring harsh reaction conditions, such as high temperatures and strong acidic or basic environments, which can lead to decomposition of sensitive functional groups. Furthermore, many classical approaches rely on stoichiometric amounts of expensive reagents or toxic heavy metal catalysts that necessitate rigorous downstream processing to meet stringent purity specifications required by global regulatory bodies. The presence of residual metals like palladium or copper can be a major bottleneck in the commercial scale-up of complex pharmaceutical intermediates, often requiring additional scavenging steps that increase both production time and overall manufacturing costs. Additionally, conventional methods may suffer from poor regioselectivity, resulting in difficult-to-separate isomeric mixtures that lower the overall yield and complicate the quality control process.
The Novel Approach
In contrast, the methodology disclosed in CN101648953B offers a streamlined, one-pot strategy that operates under relatively mild thermal conditions ranging from 70°C to 120°C. By utilizing a specific onium salt precursor and an activated alkene (such as acrylonitrile or diethyl maleate) in the presence of TPCD and triethylamine, the reaction proceeds efficiently to form the desired tricyclic core. This approach minimizes the generation of hazardous byproducts and simplifies the workup procedure to a straightforward precipitation and filtration step followed by standard column chromatography. The ability to directly introduce the biologically active cyclopropyl group during the cyclization event rather than in a separate post-functionalization step represents a significant process intensification. This novel route not only enhances the atom economy of the synthesis but also ensures a cleaner impurity profile, which is critical for accelerating the timeline from preclinical research to clinical trials.
Mechanistic Insights into TPCD-Catalyzed Cyclization
The core of this innovation lies in the unique reactivity of the onium salt intermediate, which acts as a 1,3-dipole in a cycloaddition reaction with the electron-deficient alkene. The TPCD catalyst plays a pivotal role in facilitating the formation of the reactive ylide species under mild conditions, thereby lowering the activation energy barrier for the ring-closing step. This mechanism allows for the construction of the pyrrolo[1,2-f]pyridazine framework with high regioselectivity, ensuring that the cyclopropyl carbonyl group is positioned correctly to exert its biological effect. The reaction kinetics are optimized by the choice of DMF as the solvent, which stabilizes the charged intermediates and promotes the nucleophilic attack necessary for ring closure. Understanding this mechanistic pathway is essential for R&D directors aiming to replicate or modify the synthesis for analog generation, as it highlights the critical parameters controlling product distribution and stereochemistry.
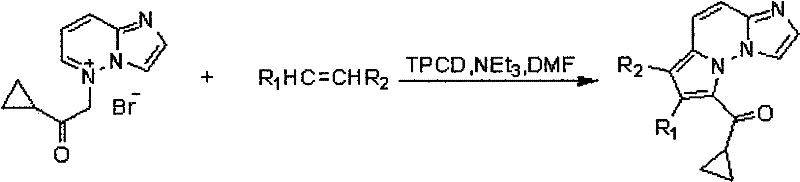
Furthermore, the impurity control mechanism inherent in this process is driven by the specificity of the cycloaddition. Unlike radical-based processes that can lead to polymerization or non-specific coupling, this ionic pathway is highly directed. The use of triethylamine as a base ensures the rapid deprotonation of the onium salt to generate the dipole, while the subsequent reaction with the alkene is fast and irreversible under the specified conditions. This kinetic control minimizes the formation of side products such as oligomers or hydrolysis byproducts, which are common pitfalls in heterocyclic synthesis. For quality assurance teams, this means that the crude product obtained after acid precipitation is already of high purity, reducing the burden on the final purification steps and ensuring consistent batch-to-batch reproducibility which is vital for GMP manufacturing.
How to Synthesize Imidazo[1,2-b]pyrrolo[1,2-f]pyridazine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable intermediates. The process begins with the dissolution of the specific onium salt in dry DMF, followed by the sequential addition of triethylamine, the alkene component, and the TPCD catalyst. The reaction mixture is then heated to an optimal temperature of 90°C, where it is maintained for a period of 4 to 15 hours depending on the specific substituents involved. Upon completion, the reaction is quenched by pouring into dilute hydrochloric acid, causing the product to precipitate out of solution. This solid is then collected by filtration and purified using silica gel chromatography with a petroleum ether and ethyl acetate eluent system. Detailed standardized synthesis steps are provided in the guide below to ensure successful replication in your laboratory.
- Dissolve the onium salt precursor in DMF and add triethylamine, acrylonitrile (or diethyl maleate), and TPCD catalyst.
- Heat the reaction mixture to 70-120°C (optimally 90°C) and maintain stirring for 4-15 hours to ensure complete conversion.
- Cool the mixture, pour into dilute hydrochloric acid to precipitate the product, filter, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains. The elimination of precious metal catalysts removes a significant cost driver and simplifies the regulatory filing process by avoiding the need for extensive heavy metal testing and clearance validation. Moreover, the starting materials, including the onium salt precursors and simple alkenes like acrylonitrile, are commodity chemicals that are readily available from multiple global suppliers, ensuring supply continuity and mitigating the risk of raw material shortages. The simplicity of the workup procedure, which relies on precipitation rather than complex extractions or distillations, translates to reduced solvent consumption and lower energy requirements during the isolation phase.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive organocatalysts instead of costly transition metals, effectively removing the need for expensive metal scavengers and reducing the overall cost of goods sold (COGS). The high atom economy of the cycloaddition reaction ensures that a greater proportion of the starting mass is converted into the final product, minimizing waste disposal fees. Additionally, the ability to perform the reaction in a single pot without intermediate isolation steps reduces labor costs and equipment occupancy time, leading to a more efficient utilization of manufacturing assets.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals rather than specialized custom-synthesized reagents, the supply chain becomes more resilient to market fluctuations and geopolitical disruptions. The robustness of the reaction conditions allows for flexibility in sourcing, as the process is tolerant to minor variations in reagent quality without compromising the final product specification. This reliability is crucial for maintaining consistent production schedules and meeting the demanding delivery timelines required by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The absence of toxic heavy metals and the use of standard organic solvents make this process highly scalable from kilogram to multi-ton production scales without significant engineering hurdles. The simplified waste stream, primarily consisting of aqueous acid and organic solvents that can be recycled, aligns well with green chemistry principles and facilitates easier compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces the risk of regulatory penalties but also enhances the corporate sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these imidazo pyridazine derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing R&D and manufacturing workflows.
Q: What is the primary biological activity of these derivatives?
A: These imidazo[1,2-b]pyrrolo[1,2-f]pyridazine derivatives exhibit significant antiproliferative activity against hepatoma cell lines (Hep-G2), making them valuable candidates for anti-liver cancer drug development.
Q: Why is the cyclopropyl group significant in this structure?
A: The cyclopropyl group is known to enhance biological activity in various drug classes, such as quinolones. In this scaffold, it contributes to unique metabolic stability and improved binding affinity against tumor cells.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the process utilizes readily available solvents like DMF and avoids expensive transition metal catalysts, simplifying purification and making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazo[1,2-b]pyrrolo[1,2-f]pyridazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the imidazo[1,2-b]pyrrolo[1,2-f]pyridazine scaffold in the fight against liver cancer. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from the bench to the market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards for safety and efficacy. We are committed to supporting your drug discovery efforts with reliable supply and technical expertise.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall development budget. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a successful partnership and accelerated time-to-market for your novel therapeutics.
