Advanced Synthesis of Spirocyclic Dienone Derivatives for High-Purity Anti-Tumor Drug Development
Advanced Synthesis of Spirocyclic Dienone Derivatives for High-Purity Anti-Tumor Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved therapeutic indices and distinct mechanisms of action for oncology treatments. Patent CN102285934B introduces a groundbreaking class of spirocyclic dienone derivatives that demonstrate remarkable anti-tumor activity through a sophisticated transition metal-catalyzed synthesis. This technology represents a significant leap forward in the construction of complex bicyclic quinone structures, which are known pharmacophores in various bioactive molecules. By leveraging high regioselective nitrene transfer reactions, this method overcomes traditional synthetic bottlenecks, providing a robust pathway for generating diverse libraries of potential drug candidates. For R&D teams focused on oncology, understanding the nuances of this synthetic route is crucial for developing next-generation therapeutics.
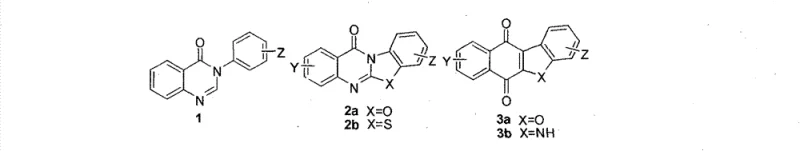
As illustrated in the general structural formula, the versatility of this scaffold allows for extensive modification at multiple positions, enabling fine-tuning of physicochemical properties and biological activity. The core structure features a spiro-fused system incorporating a dienone moiety, which is critical for its interaction with biological targets. The ability to vary substituents such as alkyl, halogen, aromatic rings, and sulfonyl groups provides medicinal chemists with a powerful toolkit for structure-activity relationship (SAR) studies. This flexibility is essential for optimizing potency while minimizing off-target effects, a common challenge in anti-cancer drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing quinone-based heterocycles often suffer from poor regioselectivity and harsh reaction conditions that can degrade sensitive functional groups. Conventional cyclization strategies may require high temperatures or strong acids, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, many existing routes rely on multi-step sequences with low overall yields, making them economically unviable for large-scale production. The lack of efficient methods to construct the specific spirocyclic architecture found in these derivatives has historically limited the exploration of this chemical space. These inefficiencies translate directly into higher costs and longer lead times for bringing new drug candidates to clinical evaluation.
The Novel Approach
The methodology described in the patent utilizes a transition metal-catalyzed nitrene transfer reaction to construct the spirocyclic core with high precision. This approach allows for the direct formation of the carbon-nitrogen or carbon-carbon bonds necessary for the spiro junction under relatively mild conditions. By employing hypervalent iodine compounds as oxidants in conjunction with metal catalysts like rhodium or copper, the reaction achieves excellent conversion rates. This novel strategy not only simplifies the synthetic route but also enhances the purity of the final product by minimizing side reactions. The result is a more streamlined process that is better suited for the rigorous demands of pharmaceutical manufacturing.
Mechanistic Insights into Transition Metal-Catalyzed Nitrene Transfer
The core of this synthetic innovation lies in the generation and subsequent reaction of a metal-nitrenoid species. When a precursor such as N-β-chloroethyl benzenesulfonamide is treated with an oxidant like iodobenzene diacetate in the presence of a catalyst, a reactive nitrene intermediate is formed. This electrophilic species then undergoes an intramolecular insertion or addition reaction with the adjacent aromatic or olefinic system. The choice of metal catalyst is critical; rhodium acetate, for instance, is highly effective at facilitating this transformation with high stereocontrol. The mechanism ensures that the spiro center is formed selectively, avoiding the formation of linear or alternative cyclic byproducts that often plague similar reactions.
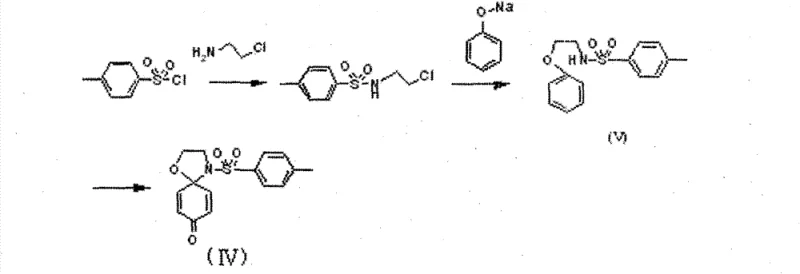
Furthermore, the reaction conditions allow for a broad scope of substrates, accommodating various electron-donating and electron-withdrawing groups on the aromatic rings. The use of solvents like dichloromethane at temperatures ranging from 10°C to 100°C provides the necessary thermal energy to drive the reaction without compromising the stability of the intermediates. This mechanistic robustness is key to achieving consistent quality across different batches. For process chemists, understanding these parameters is vital for troubleshooting and optimizing the reaction during scale-up activities. The ability to control the oxidation state and the coordination environment of the metal center ensures reproducible outcomes.
How to Synthesize Spirocyclic Dienone Derivatives Efficiently
The synthesis protocol outlined in the patent involves a logical sequence of steps designed to build complexity efficiently. It begins with the preparation of the sulfonamide precursor, followed by an etherification step to introduce the phenolic component, and culminates in the key cyclization reaction. Each step has been optimized to maximize yield and minimize purification challenges. The use of standard workup procedures, such as washing with saturated copper sulfate solution and purification via silica gel chromatography, ensures that the intermediates are of sufficient quality for the subsequent transformations. This systematic approach reduces the risk of carrying impurities through the synthesis, which is critical for maintaining high final product purity.
- Synthesize N-β-chloroethyl substituted benzenesulfonamide by reacting sulfonyl chloride with 2-chloroethylamine hydrochloride in dichloromethane with pyridine.
- Perform etherification by reacting the sulfonamide intermediate with phenol derivatives using NaH in THF or via microwave irradiation in DMF.
- Execute the key cyclization step using hypervalent iodine oxidants and transition metal catalysts (e.g., Rhodium acetate) to form the spirocyclic dienone core.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement and supply chain management within the pharmaceutical sector. The reliance on readily available starting materials, such as sulfonyl chlorides and phenols, mitigates the risk of raw material shortages. Additionally, the elimination of exotic or highly specialized reagents simplifies the sourcing process and reduces dependency on single-source suppliers. This resilience in the supply chain is increasingly important in the current global market environment. By adopting this methodology, manufacturers can secure a more stable and predictable flow of critical intermediates.
- Cost Reduction in Manufacturing: The high regioselectivity of the nitrene transfer reaction significantly reduces the formation of byproducts, thereby lowering the costs associated with purification and waste disposal. Eliminating the need for complex separation techniques translates directly into lower operational expenditures. Furthermore, the use of catalytic amounts of transition metals, rather than stoichiometric quantities of expensive reagents, contributes to overall cost efficiency. These factors combined make the production of these high-value intermediates more economically sustainable.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes common organic solvents and reagents that are widely produced and distributed globally. This ubiquity ensures that supply disruptions are less likely to impact production schedules. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further enhancing reliability. For supply chain heads, this translates to reduced lead times and greater confidence in meeting delivery commitments to downstream partners.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, allowing for the transition from gram-scale laboratory synthesis to kilogram or ton-scale commercial production without fundamental changes to the chemistry. Moreover, the process generates less hazardous waste compared to traditional methods that might use heavy metal oxidants in large excess. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this technology. They are derived from the detailed specifications and experimental data provided in the patent documentation. Understanding these points helps stakeholders make informed decisions about integrating this chemistry into their development pipelines. The answers reflect the current state of the art as described in the intellectual property.
Q: What is the primary catalytic mechanism used in this synthesis?
A: The process utilizes transition metal-catalyzed nitrene transfer reactions, typically employing rhodium or copper catalysts alongside hypervalent iodine oxidants to achieve high regioselectivity.
Q: What are the biological applications of these spirocyclic derivatives?
A: These compounds exhibit significant anti-tumor activity against various cell lines, including cervical cancer (Hela), breast cancer (MCF-7), and lung cancer (A549) cells.
Q: Can this synthesis be scaled for commercial production?
A: Yes, the reaction conditions utilize standard organic solvents and commercially available reagents, making the pathway amenable to scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirocyclic Dienone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of spirocyclic dienone derivatives in the fight against cancer. As a leading CDMO partner, we possess the technical expertise and infrastructure to bring this complex chemistry from the lab bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to explore the full potential of this technology for your drug discovery programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, high-quality intermediates.
