Advanced Metal-Free Synthesis of Selenium Heterocycles for Commercial Scale-Up
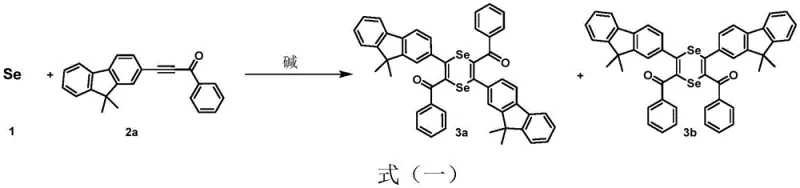
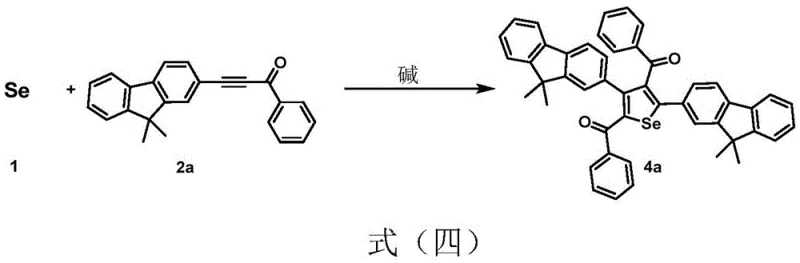
The landscape of organic synthesis for functional materials is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective methodologies. Patent CN113173909A introduces a groundbreaking approach to the preparation of selenium and tellurium-containing heterocyclic compounds, specifically targeting 1,4-diselenenes and selenophenes. These compounds are critical building blocks in the development of advanced optoelectronic materials, pharmaceutical intermediates, and bioactive molecules. The core innovation lies in the direct utilization of elemental selenium or tellurium, activated simply by a base, to react with activated internal alkynes. This represents a paradigm shift away from the reliance on toxic, unstable, and expensive selenium sources such as potassium selenocyanate or selenium halides, which have historically plagued the industry with safety hazards and complex waste disposal issues. By leveraging temperature regulation, this technology allows for the selective generation of either six-membered 1,4-diselenene rings or five-membered selenophene rings, offering unprecedented control over molecular architecture without the need for transition metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of selenium-containing heterocycles has been fraught with significant technical and economic challenges that hinder large-scale commercial adoption. Traditional protocols often necessitate the use of highly toxic and unstable inorganic salts like potassium selenocyanate or volatile organic selenium ethers, which pose severe safety risks to personnel and require specialized containment infrastructure. Furthermore, these conventional routes frequently depend on the presence of transition metal catalysts, such as palladium or copper complexes, to facilitate the cyclization reactions. The reliance on these precious metals not only inflates the raw material costs but also introduces a critical downstream bottleneck: the rigorous removal of trace metal residues to meet the stringent purity specifications required for electronic grade materials or pharmaceutical applications. Additionally, many prior art methods involve harsh reaction conditions, including the use of strong Grignard reagents or alkali metals, which demand strictly anhydrous environments and cryogenic temperatures, thereby escalating energy consumption and operational complexity in a production setting.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN113173909A offers a streamlined, metal-free pathway that fundamentally simplifies the manufacturing process. By employing elemental selenium, a stable and economically abundant industrial raw material, the process eliminates the volatility and toxicity associated with traditional selenium sources. The reaction is induced solely by the addition of a base, such as potassium hydroxide or cesium carbonate, in common polar aprotic solvents like dimethyl sulfoxide. This base-induced activation allows for a direct [2+2] cycloaddition with activated alkynes, proceeding under remarkably mild conditions. The true brilliance of this approach is its tunability; by merely adjusting the oil bath temperature, operators can selectively steer the reaction towards the formation of 1,4-diselenenes at lower temperatures or selenophenes at elevated temperatures. This level of control, achieved without exotic ligands or catalysts, drastically reduces the chemical footprint and simplifies the workup procedure, making it an ideal candidate for reliable optoelectronic material supplier operations seeking to optimize their supply chains.
Mechanistic Insights into Base-Induced Selenium Activation and Cyclization
The mechanistic elegance of this synthesis lies in the base-mediated activation of elemental selenium, which generates reactive selenium species capable of attacking the electron-deficient triple bond of the activated alkyne. Unlike metal-catalyzed cycles that rely on oxidative addition and reductive elimination steps, this process likely proceeds through a nucleophilic attack mechanism where the base facilitates the cleavage of Se-Se bonds in the elemental selenium lattice. Once activated, the selenium species undergoes a concerted addition across the alkyne moiety. At moderate temperatures, typically ranging from 0°C to 110°C, the kinetic product is favored, resulting in the formation of the unique six-membered 1,4-diselenene ring system. This structure possesses a non-planar, ship-shaped conformation that is distinct from typical aromatic systems, offering unique electronic properties that are valuable for tuning the bandgap in organic semiconductors. The absence of metal coordination spheres means that the reaction trajectory is governed purely by electronic and steric factors of the substrates and the thermal energy provided, allowing for a cleaner reaction profile with fewer side products derived from catalyst decomposition.
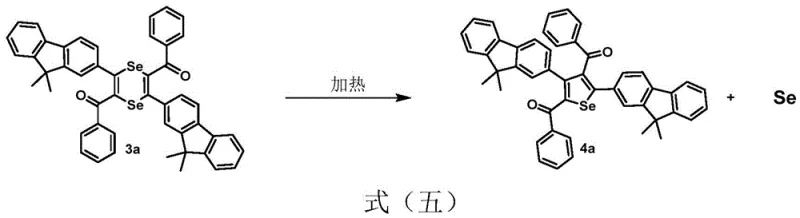
Furthermore, the system demonstrates remarkable versatility in its ability to convert the initial 1,4-diselenene intermediates into the thermodynamically more stable five-membered selenophene derivatives. This transformation can be triggered either by increasing the reaction temperature to the 111-160°C range or by introducing mild oxidizing agents. Under high-temperature conditions, the six-membered ring undergoes a rearrangement and extrusion of selenium to form the aromatic selenophene core, a process that is highly exothermic and self-sustaining once initiated. Alternatively, oxidative conditions promote the dehydrogenation and cyclization necessary to aromatize the ring system. This dual-pathway capability provides manufacturers with flexible process options; one can choose to isolate the novel 1,4-diselenene for specific applications requiring non-planar geometries or push the reaction to completion to yield high-purity selenophenes for standard OLED emissive layers. The ability to control impurity profiles through simple thermal or oxidative adjustments ensures that the final product meets the rigorous quality standards demanded by the display and pharmaceutical industries.
How to Synthesize Selenium Heterocycles Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory or pilot plant equipment without the need for specialized high-pressure reactors or inert gas manifolds beyond standard nitrogen protection. The process begins with the sequential addition of the activated alkyne substrate, elemental selenium powder, and a stoichiometric amount of base into a reaction vessel containing a solvent such as DMSO or DMF. The mixture is then heated to the target temperature, which serves as the primary control knob for product selectivity. Following the reaction period, which typically spans several hours depending on the scale and temperature, the workup involves a straightforward aqueous quench and extraction with dichloromethane. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification parameters for various substrate scopes, are outlined in the guide below.
- Mix activated alkyne, elemental selenium, and a base (e.g., KOH) in a solvent like DMSO.
- Heat the mixture to 60-110°C for 1,4-diselenenes or 111-160°C for selenophenes.
- Purify the crude product via column chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology presents a compelling value proposition centered on cost stability and operational resilience. The primary driver of cost reduction in electronic chemical manufacturing here is the complete elimination of precious metal catalysts. In traditional processes, the cost of palladium or platinum catalysts, along with the expensive ligands required to stabilize them, constitutes a significant portion of the bill of materials. Moreover, the downstream processing required to strip these metals from the final product to parts-per-million levels often involves costly scavenger resins or multiple recrystallization steps, which erode overall yield. By removing this requirement entirely, the new process not only lowers direct material costs but also significantly simplifies the purification train, leading to higher throughput and reduced solvent consumption. This efficiency translates directly into improved margins and a more competitive pricing structure for high-purity selenium heterocycles in the global market.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just the catalyst savings; the use of elemental selenium as the primary chalcogen source offers substantial stability in raw material pricing. Unlike specialized organoselenium reagents that may be subject to supply volatility and high markups from niche chemical suppliers, elemental selenium is a commodity chemical produced in vast quantities for other industries, ensuring a robust and predictable supply chain. Additionally, the mild reaction conditions reduce energy expenditures associated with maintaining cryogenic temperatures or high-pressure environments, further driving down the operational expenditure (OPEX) per kilogram of product. The simplified workup procedure also minimizes waste generation, reducing the costs associated with hazardous waste disposal and environmental compliance, which are increasingly significant financial burdens in modern chemical manufacturing facilities.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemistry mitigates the risk of production delays caused by reagent shortages. Since the method relies on widely available bases and solvents alongside elemental selenium, it is less susceptible to the geopolitical or logistical disruptions that often affect the supply of complex organometallic catalysts. The tolerance of the reaction to various functional groups on the alkyne substrate also means that a single production line can be easily adapted to synthesize a diverse library of derivatives simply by swapping the starting alkyne, enhancing the agility of the manufacturing unit to respond to changing customer demands. This flexibility is crucial for maintaining continuity of supply for key clients in the OLED and pharmaceutical sectors who require rapid iteration of material structures during their own R&D phases.
- Scalability and Environmental Compliance: The scalability of this process is inherently superior due to the absence of exothermic risks associated with metal-catalyzed cross-couplings that often require careful heat management on a large scale. The reaction can be safely scaled from gram to multi-ton quantities using standard batch reactors, facilitating the commercial scale-up of complex organic intermediates without the need for extensive process re-engineering. Furthermore, the green chemistry credentials of this method align perfectly with the increasingly strict environmental regulations governing chemical production. By avoiding toxic selenium halides and heavy metals, the facility reduces its environmental footprint, simplifying the permitting process and enhancing the company's sustainability profile, which is a key factor for multinational corporations when selecting long-term partners for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium heterocycle synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using elemental selenium over traditional reagents?
A: Elemental selenium is low toxicity, stable, and economical compared to toxic and unstable reagents like potassium selenocyanate or selenium halides used in conventional methods.
Q: How is selectivity between 1,4-diselenene and selenophene controlled?
A: Selectivity is achieved through precise temperature regulation; lower temperatures (0-110°C) favor 1,4-diselenenes, while higher temperatures (111-160°C) favor selenophenes.
Q: Is metal catalyst removal required in this process?
A: No, the process is completely metal-free, utilizing only base induction, which eliminates the need for expensive and complex heavy metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selenium Heterocycles Supplier
As the demand for high-performance organic electronic materials continues to surge, the ability to produce specialized intermediates like 1,4-diselenenes and selenophenes efficiently is a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this innovative metal-free synthesis to the market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, free from the metal contaminants that plague conventionally produced materials. We understand the critical nature of supply continuity for our partners in the display and pharmaceutical industries, and our robust supply chain management ensures that we can deliver consistent quality at scale, supporting your R&D and commercialization timelines effectively.
We invite you to explore how this advanced synthesis route can optimize your material costs and enhance your product performance. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact our technical procurement team to request specific COA data for our selenium heterocycle portfolio and to discuss route feasibility assessments for your custom projects. By collaborating with NINGBO INNO PHARMCHEM, you gain access to cutting-edge chemistry backed by a commitment to quality, sustainability, and commercial reliability.
