Advanced Manufacturing of Zaltoprofen Intermediates via Novel Iodine-Mediated Rearrangement Technology
Advanced Manufacturing of Zaltoprofen Intermediates via Novel Iodine-Mediated Rearrangement Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for non-steroidal anti-inflammatory drugs (NSAIDs) that balance efficacy with manufacturing feasibility. Zaltoprofen, chemically known as 2-(10-oxo-10,11-dihydrodibenz[b,f]thiepin-2-yl)propionic acid, represents a high-value target due to its potent analgesic properties and selective action on inflammation sites with minimal gastric side effects. A pivotal advancement in this domain is detailed in patent CN100462362C, which discloses an improved preparation process that fundamentally alters the construction of the dibenzothiepin core. This technology replaces hazardous and costly reagents with a streamlined iodine-mediated rearrangement strategy, offering a compelling value proposition for large-scale API production. The structural integrity and therapeutic potential of the final molecule rely heavily on the precision of these intermediate steps.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
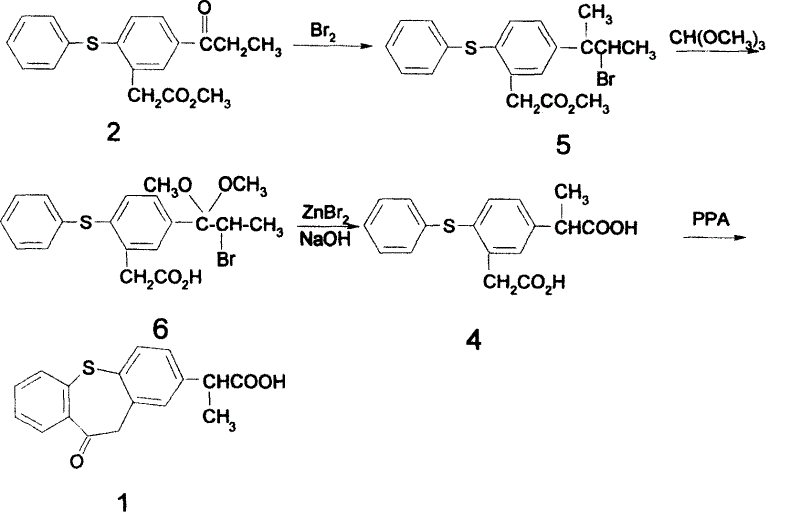
Historically, the synthesis of zaltoprofen intermediates has been plagued by significant operational and economic hurdles that hinder industrial scalability. Early methodologies, such as those disclosed in Japanese patent literature, relied heavily on the use of iodobenzene diacetate for key rearrangement reactions. This reagent is not only prohibitively expensive but also difficult to recover and recycle, leading to inflated production costs and substantial chemical waste. Furthermore, alternative routes involving bromination followed by ketalization and cyanation introduced severe safety and environmental liabilities. The use of elemental bromine and potassium cyanide necessitates rigorous containment protocols and complex purification steps to remove toxic residues, often resulting in low overall yields due to product degradation during harsh workup procedures. These legacy processes create bottlenecks in the supply chain, making the reliable sourcing of high-purity intermediates a persistent challenge for procurement teams.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach outlined in the patent utilizes elemental iodine and trialkyl orthoformate to drive the critical rearrangement of the phenylacetate backbone. This strategic substitution dramatically simplifies the reaction profile by eliminating the need for hypervalent iodine oxidants or toxic cyanide sources. The process flow is optimized to proceed through a direct rearrangement of 5-propionyl-2-thiophenyl phenylacetate esters, facilitated by the unique reactivity of iodine in the presence of orthoformates. This method not only shortens the synthetic sequence by removing intermediate protection and deprotection steps but also enhances the purity profile of the crude product. By leveraging common industrial chemicals, this route transforms a complex multi-step synthesis into a more linear and manageable operation, directly addressing the cost and efficiency pain points associated with traditional NSAID manufacturing.

Mechanistic Insights into Iodine-Mediated Rearrangement
The core innovation of this synthesis lies in the mechanistic pathway of the iodine-mediated rearrangement, which converts the propionyl-substituted precursor into the requisite propionic acid side chain configuration. In this transformation, elemental iodine acts as an electrophilic activator, interacting with the enolizable ketone moiety of the propionyl group. The presence of trialkyl orthoformate serves a dual purpose: it acts as a water scavenger to drive the equilibrium forward and potentially stabilizes reactive intermediates through transient ketal formation. This interaction facilitates a 1,2-shift or similar rearrangement mechanism that effectively migrates the carbon skeleton to the desired position. The reaction can be further optimized through the addition of catalytic amounts of Lewis acids or copper salts, which enhance the electrophilicity of the iodine species and allow for reduced reagent loading. This catalytic enhancement is crucial for minimizing halogenated waste and improving the atom economy of the overall process.
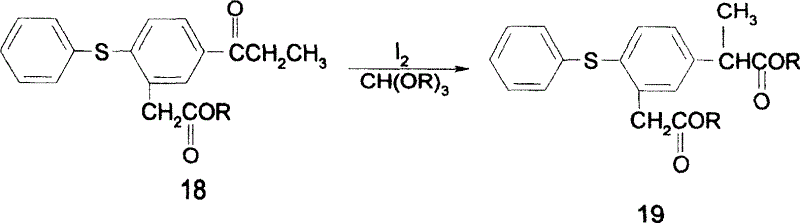
Impurity control is another critical aspect where this mechanism offers distinct advantages over bromination routes. In traditional bromination, the presence of multiple active hydrogen atoms often leads to poly-brominated byproducts that are structurally similar to the target and difficult to separate. The iodine-mediated pathway, however, exhibits higher regioselectivity, largely avoiding the formation of complex halogenated impurities. Furthermore, the protocol includes a post-reaction treatment with sodium thiosulfate, which effectively quenches any unreacted iodine. This step prevents iodine carryover into subsequent stages, ensuring that the final hydrolysis and cyclization steps proceed without interference from oxidative side reactions. The resulting intermediate, 5-(1-carboxy ethyl)-2-thiophenyl toluylic acid, is obtained with high purity, facilitating a cleaner final cyclization to zaltoprofen and reducing the burden on downstream purification units.
How to Synthesize Zaltoprofen Efficiently
The synthesis of zaltoprofen via this improved route involves a logical sequence of unit operations designed for maximum throughput and minimal waste generation. The process begins with the esterification of o-chlorophenylacetic acid, followed by a Friedel-Crafts acylation to install the propionyl group. Subsequent Ullmann condensation with thiophenol constructs the diphenyl sulfide framework, which is then subjected to the pivotal iodine-mediated rearrangement. Following rearrangement, hydrolysis reveals the free acid, which undergoes intramolecular cyclization in the presence of polyphosphoric acid (PPA) to close the seven-membered ring. Each step has been optimized for solvent recovery and reagent efficiency, making the entire sequence highly amenable to kilogram-to-ton scale production. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Perform esterification of o-chlorophenylacetic acid followed by Friedel-Crafts acylation to generate the propionyl-chloro precursor.
- Execute Ullmann condensation with thiophenol to form the diphenyl sulfide backbone, followed by esterification.
- Conduct the critical rearrangement using iodine and trialkyl orthoformate, followed by hydrolysis and PPA cyclization to yield zaltoprofen.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iodine-mediated synthesis route offers transformative benefits for procurement managers and supply chain directors focused on cost optimization and risk mitigation. The primary driver of value is the substitution of high-cost specialty reagents with commodity chemicals. Elemental iodine and orthoformates are widely available in the global chemical market with stable pricing structures, unlike hypervalent iodine compounds which are subject to supply volatility and premium pricing. This shift significantly reduces the raw material cost base for the active pharmaceutical ingredient (API). Additionally, the elimination of toxic cyanide and bromine reagents lowers the regulatory burden and waste disposal costs associated with hazardous material handling. Facilities can operate with greater flexibility and reduced insurance liabilities, translating into tangible long-term savings for the manufacturing organization.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic reduction in reagent costs and the simplification of the purification workflow. By avoiding expensive oxidants like iodobenzene diacetate and eliminating the need for complex chromatographic separations required to remove brominated impurities, the overall cost of goods sold (COGS) is substantially lowered. The ability to recycle sodium iodide byproducts further enhances the economic efficiency of the process. Moreover, the shorter reaction sequence reduces utility consumption, including energy for heating and cooling, as well as solvent usage, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on robust, commercially available starting materials. O-chlorophenylacetic acid and thiophenol are produced at scale by multiple global suppliers, reducing the risk of single-source dependency. The simplified process flow also means fewer unit operations and less equipment occupancy time, allowing for faster batch turnover and increased production capacity. This agility enables manufacturers to respond more rapidly to fluctuations in market demand for zaltoprofen, ensuring consistent delivery schedules to downstream pharmaceutical partners without the delays often caused by complex, multi-step legacy syntheses.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry principles and stringent regulatory standards. The absence of heavy metal catalysts and highly toxic reagents like cyanide simplifies effluent treatment and reduces the environmental footprint of the facility. The process generates aqueous waste streams that are easier to treat compared to the halogenated organic waste from bromination routes. This compliance advantage facilitates smoother regulatory approvals and site audits. Furthermore, the robustness of the reaction conditions allows for seamless scale-up from pilot plant to commercial production volumes, ensuring that quality and yield remain consistent regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zaltoprofen synthesis technology. These insights are derived directly from the patent specifications and practical considerations for industrial application. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement teams assessing supplier capabilities. The answers provided reflect the specific advantages of the iodine-mediated route over conventional methods, highlighting its suitability for modern pharmaceutical manufacturing environments.
Q: What are the primary advantages of the iodine-mediated rearrangement over traditional bromination methods?
A: The iodine-mediated rearrangement eliminates the need for toxic bromine and cyanide reagents, significantly reducing environmental hazards and purification complexity while utilizing lower-cost elemental iodine compared to expensive hypervalent iodine reagents.
Q: How does this process improve supply chain stability for zaltoprofen production?
A: By relying on readily available commodity chemicals like o-chlorophenylacetic acid and elemental iodine, the process reduces dependency on specialized, high-cost reagents like iodobenzene diacetate, ensuring more consistent raw material availability.
Q: Is the sodium iodide byproduct manageable in this synthesis route?
A: Yes, the process generates sodium iodide as a byproduct during the hydrolysis stage, which is easily separable and can be recycled or disposed of with standard waste treatment protocols, unlike the complex organic waste from previous methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zaltoprofen Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity zaltoprofen intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific reagent requirements of the iodine-mediated process, guaranteeing a consistent and reliable supply of this vital NSAID building block for your drug development programs.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your project's bottom line while ensuring the highest standards of quality and regulatory compliance.
