Revolutionizing Macitentan Production: A Novel Late-Stage Sulfonamide Coupling Strategy
Revolutionizing Macitentan Production: A Novel Late-Stage Sulfonamide Coupling Strategy
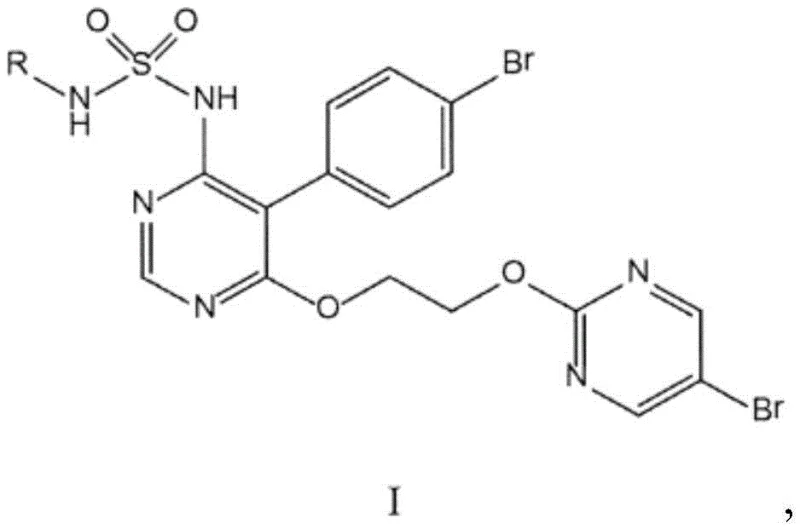
The pharmaceutical landscape for pulmonary hypertension treatment relies heavily on the efficient supply of high-purity active pharmaceutical ingredients (APIs), with Macitentan standing out as a critical endothelin receptor antagonist. The manufacturing of such complex molecules often faces significant bottlenecks during the final functionalization steps, particularly regarding purity profiles and the removal of hazardous reagents. Patent CN107162988B discloses a groundbreaking process for the manufacture of pyrimidine sulfonamide derivatives of Formula I, specifically addressing the challenges associated with introducing the sulfonamide moiety. This innovation shifts the paradigm from early-stage functionalization to a robust late-stage nucleophilic substitution, offering a pathway that significantly enhances process viability. By leveraging specific fluoride-mediated activation, this method overcomes electronic deactivation barriers that previously rendered certain synthetic routes unfeasible, providing a reliable foundation for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Macitentan and related analogues, as described in prior art such as WO 02/053557, relied on reacting a chloro-pyrimidine intermediate with a large excess of ethylene glycol to install the critical ether side chain. This approach presents severe downstream processing challenges. Ethylene glycol possesses a high boiling point, making its complete removal via distillation energetically expensive and technically difficult. Consequently, residual ethylene glycol often persists into subsequent steps, acting as a persistent impurity that complicates the final isolation of the API. To achieve the requisite purity levels, manufacturers were forced to employ labor-intensive purification techniques, such as column chromatography, which are notoriously difficult to translate from laboratory benchtop to multi-ton commercial production. Furthermore, the use of strong bases like sodium hydride in tetrahydrofuran (THF) for the subsequent coupling steps introduces safety hazards and solvent exchange complexities that inflate operational expenditures.
The Novel Approach
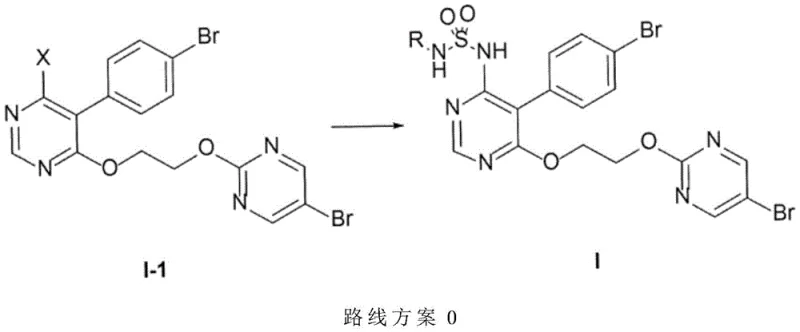
The process disclosed in CN107162988B fundamentally reengineers the synthetic sequence by deferring the introduction of the sulfonamide function to the very last step. Instead of building the side chain onto a sulfonamide-bearing core, the inventors utilize a pre-functionalized pyrimidine intermediate (Formula I-1) that already contains the 2-(5-bromopyrimidin-2-yloxy)ethoxy side chain. This strategic pivot eliminates the need for excess ethylene glycol in the final stages, thereby removing the primary source of difficult-to-remove impurities. The core innovation lies in the successful execution of a nucleophilic aromatic substitution on this complex intermediate using a sulfonamide nucleophile. Contrary to established chemical intuition which suggested such a reaction would fail due to electronic effects, the new method employs specific reaction conditions—including polar aprotic solvents and fluoride additives—to drive the reaction to completion with high yields, enabling direct crystallization of the product.
Mechanistic Insights into Fluoride-Mediated Nucleophilic Substitution
From a mechanistic perspective, the success of this process is counterintuitive and represents a significant technical achievement. The pyrimidine ring in the intermediate Formula I-1 is substituted with an electron-donating alkoxy side chain at the 6-position. In classical organic chemistry, such electron-donating groups deactivate the aromatic ring towards nucleophilic attack, typically rendering the displacement of a halogen leaving group (such as chlorine or bromine) at the 4-position extremely sluggish or impossible under standard conditions. Standard fluorinating agents like KF or NaF were observed to fail in producing fluorinated intermediates with sufficient purity, and reactions with sulfonamides in DMSO at moderate temperatures initially showed no conversion. The inventors discovered that the addition of specific fluoride sources, namely tetra-n-butylammonium fluoride (TBAF) hydrate or cesium fluoride (CsF), acts as a crucial promoter. These additives likely function by enhancing the nucleophilicity of the sulfonamide or by activating the carbon-halogen bond through complexation, effectively overcoming the electronic deactivation imposed by the alkoxy substituent.
This mechanistic breakthrough allows for the use of milder bases, such as potassium carbonate (K2CO3) or cesium carbonate (Cs2CO3), in polar aprotic solvents like dimethyl sulfoxide (DMSO). The reaction proceeds efficiently at temperatures ranging from 60°C to 80°C, which is significantly milder than the harsh conditions often required for unactivated substrates. The ability to control the reaction kinetics through the stoichiometry of the fluoride additive (typically 1.1 to 4 equivalents) provides a robust handle for process optimization. This level of control minimizes the formation of side products and ensures that the sulfonamide coupling occurs selectively at the desired position on the pyrimidine ring, preserving the integrity of the sensitive bromo-pyrimidine moiety on the side chain. Such precision is vital for maintaining the impurity profile within strict regulatory limits required for pharmaceutical intermediates.
How to Synthesize Macitentan Intermediates Efficiently
The implementation of this novel synthetic route requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The process begins with the preparation of the key halogenated intermediate, Formula I-1, which serves as the universal precursor for various R-group derivatives. Detailed standardized synthesis steps for preparing the starting materials and executing the final coupling are outlined below, reflecting the optimized conditions found in the patent examples. This guide focuses on the critical fluoride-mediated substitution step that defines the novelty of this manufacturing route.
- Prepare the key intermediate Formula I-1 (wherein X is Cl or Br) containing the pre-functionalized ethoxy side chain.
- React Formula I-1 with the corresponding sulfonamide (Formula I-2) in a polar aprotic solvent like DMSO.
- Utilize a fluoride source such as TBAF hydrate or Cesium Fluoride alongside a base like Potassium Carbonate to facilitate the substitution at 60-80°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel manufacturing process offers tangible economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the drastic simplification of the purification workflow. By eliminating the need for column chromatography—a technique that is cost-prohibitive and slow at commercial scale—the process enables isolation via crystallization. Crystallization is inherently more scalable, consumes less solvent, and generates less waste, directly translating to lower manufacturing costs and a reduced environmental footprint. Furthermore, the removal of ethylene glycol from the late-stage process inventory mitigates the risk of batch failures due to persistent solvent impurities, ensuring higher consistency in supply.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps results in substantial cost savings by reducing solvent consumption, silica gel usage, and processing time. Additionally, the ability to use a single universal intermediate (Formula I-1) for the production of multiple derivatives (where R varies) streamlines inventory management and reduces the capital tied up in diverse raw materials. The use of inexpensive inorganic bases like potassium carbonate instead of costly organometallic reagents further drives down the bill of materials, making the overall cost of goods sold (COGS) significantly more competitive.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate moderate temperatures and common solvents like DMSO, reduces the dependency on specialized equipment or cryogenic cooling systems. This flexibility allows for manufacturing in a wider range of facilities, diversifying the supply base and reducing the risk of disruption. The high purity achieved directly from crystallization minimizes the need for reprocessing or recycling off-spec material, ensuring that delivery timelines are met consistently without unexpected delays caused by quality control failures.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by reducing the volume of hazardous waste associated with chromatography fractions. The solvents used, primarily DMSO and ethyl acetate, are easier to recover and recycle compared to the complex mixtures generated in older routes. This facilitates compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and waste disposal. The scalability is further evidenced by the successful execution of the reaction on multi-gram to kilogram scales in the patent examples, demonstrating readiness for ton-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the feasibility and advantages of adopting this route for API manufacturing.
Q: Why is the new process for Macitentan intermediates superior to conventional methods?
A: Conventional methods often require ethylene glycol in late stages, which is difficult to remove due to its high boiling point, necessitating complex purification like column chromatography. The new process introduces the sulfonamide group in the final step on a pre-functionalized intermediate, avoiding excess ethylene glycol and enabling simpler crystallization purification.
Q: What role does the fluoride source play in this synthesis?
A: The pyrimidine ring in the intermediate is electronically deactivated by the alkoxy side chain, making standard nucleophilic substitution difficult. The addition of tetra-n-butylammonium fluoride (TBAF) or cesium fluoride activates the system, allowing the sulfonamide nucleophile to successfully displace the halogen leaving group under moderate conditions.
Q: Can this process be scaled for commercial production?
A: Yes, the process utilizes robust reagents like potassium carbonate and common polar aprotic solvents (DMSO). It avoids sensitive catalysts and allows for isolation via crystallization rather than chromatography, which is a critical factor for scalable, GMP-compliant commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Macitentan Intermediate Supplier
The technical advancements detailed in CN107162988B represent a significant leap forward in the synthesis of pyrimidine sulfonamide derivatives, offering a clearer path to high-purity Macitentan intermediates. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative patent technologies into robust, commercial-grade manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this fluoride-mediated coupling are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required by global regulatory bodies.
We invite pharmaceutical partners to leverage our expertise to optimize their supply chains for pulmonary hypertension treatments. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of high-quality intermediates that drive your drug development programs forward with confidence.
