Optimizing Hydroxyacetophenone Production: A Technical Breakthrough in Solvent-Free Fries Rearrangement
Optimizing Hydroxyacetophenone Production: A Technical Breakthrough in Solvent-Free Fries Rearrangement
In the highly competitive landscape of fine chemical manufacturing, the efficient synthesis of critical intermediates like o-hydroxyacetophenone and p-hydroxyacetophenone remains a pivotal challenge for R&D and procurement teams alike. These compounds serve as essential building blocks for a wide array of pharmaceutical agents, including the Class Ia antiarrhythmic drug propafenone hydrochloride and the widely used analgesic paracetamol. A significant technological advancement in this domain is detailed in patent CN102093189B, which outlines a novel, solvent-free preparation method that addresses many of the inefficiencies plaguing traditional synthesis routes. By leveraging a refined Fries rearrangement protocol, this technology offers a pathway to higher yields, improved isomer ratios, and a drastically reduced environmental footprint, positioning it as a superior choice for modern industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hydroxyacetophenones has relied heavily on classical two-step processes involving esterification followed by Fries rearrangement in the presence of organic solvents. Traditional methodologies often utilize chlorobenzene or similar halogenated solvents to manage the exothermic nature of the Lewis acid catalysis, typically employing aluminum trichloride. However, these conventional approaches suffer from significant drawbacks, including low yields for the ortho-isomer, which is often the more valuable target for specific pharmaceutical syntheses. Furthermore, the reliance on volatile organic compounds (VOCs) introduces severe environmental compliance issues and necessitates complex, energy-intensive solvent recovery systems. The separation of isomers in these traditional setups is also notoriously difficult, often requiring multiple crystallization steps that erode overall process efficiency and increase the cost of goods sold (COGS) due to material loss.
The Novel Approach
The innovative method described in the referenced patent fundamentally reengineers the rearrangement step by eliminating the solvent entirely, thereby creating a more concentrated and thermally efficient reaction environment. Instead of relying on dilute solutions in chlorobenzene, the process utilizes a melt of phenyl acetate and Lewis acid catalysts, such as anhydrous aluminum chloride or zinc dichloride, at elevated temperatures ranging from 160°C to 170°C. This solvent-free condition not only accelerates the reaction kinetics but also allows for precise tuning of the ortho-to-para isomer ratio, achieving a favorable distribution of up to 1.4:1. The downstream processing is equally ingenious, employing a combination of freezing filtration to isolate the para-isomer and steam distillation coupled with vacuum rectification for the ortho-isomer, ensuring high purity without the burden of solvent removal.
Mechanistic Insights into Lewis Acid-Catalyzed Fries Rearrangement
To fully appreciate the technical superiority of this solvent-free protocol, one must understand the underlying mechanistic nuances of the Fries rearrangement as facilitated by strong Lewis acids. The reaction initiates with the coordination of the Lewis acid, typically AlCl3, to the carbonyl oxygen of the phenyl acetate ester, forming a stable complex that activates the acyl group for migration. As the temperature increases, the C-O bond of the ester undergoes heterolytic cleavage, generating a reactive acylium ion paired with a phenoxy-aluminum species. This acylium ion acts as a potent electrophile, attacking the aromatic ring of the phenoxy moiety in an intramolecular or tightly coupled intermolecular fashion. The regioselectivity of this electrophilic aromatic substitution is governed by thermodynamic and kinetic factors, where lower temperatures generally favor the para-product, while higher temperatures, as utilized in this patent, promote the formation of the thermodynamically stable ortho-isomer through chelation control.
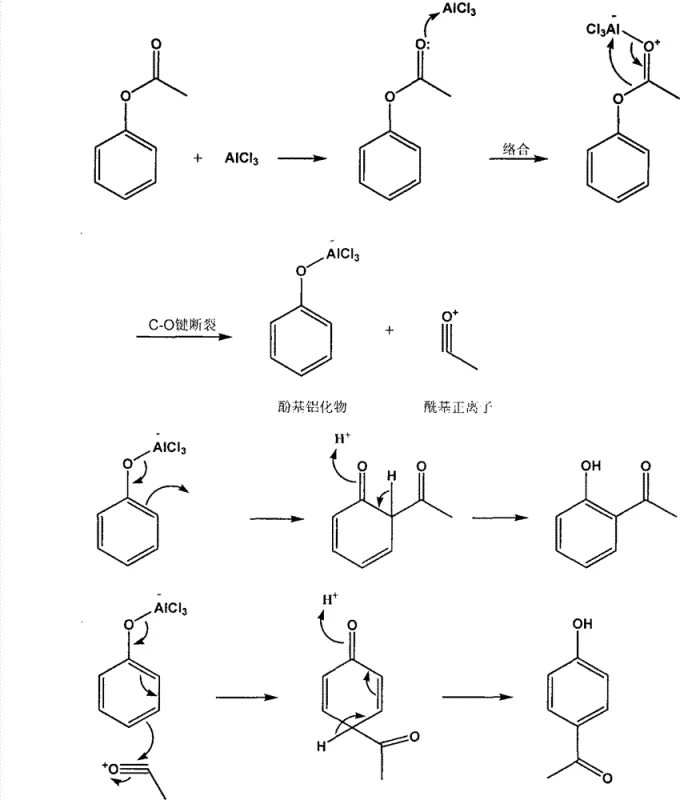
Impurity control in this system is critically dependent on the management of the acidic byproducts and the prevention of poly-acylation. The patent specifies a hydrolysis step using ice-cold hydrochloric acid, which serves to quench the aluminum complexes and liberate the free phenolic products while minimizing side reactions such as resinification or tar formation. The use of specific molar ratios of Lewis acid (1:2 to 1:3 relative to the ester) ensures complete conversion while maintaining a manageable viscosity in the reaction melt. Furthermore, the distinct physical properties of the resulting isomers are exploited for purification; the para-isomer's higher melting point allows for direct crystallization from the cooled reaction mass, effectively removing soluble impurities and residual catalysts before the ortho-isomer is recovered from the filtrate via distillation.
How to Synthesize Hydroxyacetophenone Efficiently
Implementing this synthesis route requires strict adherence to thermal profiles and stoichiometric precision to maximize the yield of both isomers. The process begins with the high-yield esterification of phenol and acetic anhydride, followed by the critical solvent-free rearrangement step where temperature control is paramount. Operators must carefully manage the addition of the Lewis acid to prevent runaway exotherms, followed by a controlled heating phase to drive the rearrangement to completion. The subsequent workup involves a sophisticated separation strategy that leverages the differential solubility and volatility of the products. For a comprehensive understanding of the operational parameters, safety protocols, and specific equipment requirements necessary for successful execution, please refer to the standardized synthesis guide below.
- Esterify phenol with acetic anhydride using sulfuric acid catalysis to obtain phenyl acetate with high yield.
- Perform solvent-free Fries rearrangement using anhydrous aluminum chloride at 160-170°C to generate the isomeric mixture.
- Separate isomers via freezing filtration for the para-isomer and steam distillation followed by vacuum rectification for the ortho-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this solvent-free Fries rearrangement technology offers compelling advantages that directly impact the bottom line and supply chain resilience. By eliminating the need for large volumes of organic solvents like chlorobenzene, manufacturers can significantly reduce raw material procurement costs and mitigate the risks associated with volatile solvent pricing and availability. The simplified workflow reduces the number of unit operations required, translating to lower utility consumption and reduced labor hours per batch. Moreover, the ability to tune the isomer ratio provides flexibility in meeting specific customer demands without the need for separate, dedicated production lines, thereby enhancing asset utilization and overall operational efficiency in a multi-product facility.
- Cost Reduction in Manufacturing: The elimination of solvent purchase, storage, and recovery infrastructure results in substantial capital and operational expenditure savings. Without the need for complex distillation columns to recycle chlorobenzene, the process intensity is lowered, and the energy demand per kilogram of product is drastically reduced. Additionally, the high conversion rates and efficient separation techniques minimize material loss, ensuring that the theoretical yield is closely approached in commercial practice, which further drives down the effective cost per unit for high-purity hydroxyacetophenone.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically phenol and acetic anhydride, are commodity chemicals with robust global supply chains, reducing the risk of feedstock shortages. The simplified process flow, characterized by fewer steps and less sensitive handling requirements compared to solvent-based methods, enhances production reliability and reduces the likelihood of batch failures. This robustness ensures consistent delivery schedules, allowing procurement managers to maintain leaner inventory levels while securing a steady flow of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling this solvent-free process is inherently safer and more straightforward, as the absence of flammable solvents reduces fire hazards and simplifies reactor design. The environmental benefits are profound, as the process generates less hazardous waste and avoids the emission of VOCs, aligning with increasingly stringent global environmental regulations. The acidic wastewater generated during hydrolysis can be treated to recover valuable aluminum salts, turning a potential waste stream into a secondary revenue source or reducing disposal costs, thus supporting a circular economy approach within the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of hydroxyacetophenones using this advanced methodology. These insights are derived directly from the patent specifications and practical industrial experience, aiming to clarify the feasibility and benefits of adopting this route for large-scale manufacturing. Understanding these details is crucial for technical teams evaluating process transfer and for commercial teams negotiating supply agreements based on quality and sustainability metrics.
Q: What is the primary advantage of the solvent-free Fries rearrangement method?
A: The solvent-free approach eliminates the need for hazardous organic solvents like chlorobenzene, significantly reducing environmental pollution and downstream solvent recovery costs while maintaining high reaction yields.
Q: How is the ortho/para isomer ratio controlled in this process?
A: The ratio is primarily controlled by reaction temperature and the specific Lewis acid catalyst used. The patented method achieves an ortho-to-para ratio of up to 1.4:1 by optimizing the thermal conditions during the rearrangement step.
Q: What are the key purification steps for obtaining high-purity products?
A: The process utilizes a combination of freezing filtration to isolate crude p-hydroxyacetophenone, followed by water recrystallization. The o-hydroxyacetophenone is purified through steam distillation and subsequent vacuum rectification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxyacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate thermal and separation requirements of the Fries rearrangement are met with precision. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for these critical pharmaceutical intermediates. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable sourcing strategy available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
