Advanced Synthesis of Bisphenol AF: Enhancing Yield and Sustainability for Global Supply Chains
Advanced Synthesis of Bisphenol AF: Enhancing Yield and Sustainability for Global Supply Chains
The global demand for high-performance fluoropolymers continues to surge, driving the need for robust and efficient supply chains for critical intermediates like Bisphenol AF (2,2-di(4-hydroxyphenyl)hexafluoropropane). A pivotal advancement in this domain is detailed in patent CN101870641B, which outlines a refined manufacturing method utilizing hexafluoroacetone, phenol, and hydrogen fluoride. This technology addresses long-standing challenges in the industry, specifically focusing on improving reaction yields while simultaneously mitigating the environmental burden associated with traditional synthesis routes. By optimizing the molar ratios of reactants and implementing a strategic recovery system for catalysts and unreacted materials, this process offers a compelling value proposition for manufacturers seeking to enhance operational efficiency. For R&D directors and procurement specialists, understanding the nuances of this patented approach is essential for securing a reliable fluorine materials supplier capable of meeting stringent quality and sustainability standards in the competitive landscape of advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Bisphenol AF has been plagued by inefficiencies that impact both cost structures and environmental compliance. Conventional methods typically operate under harsh conditions, often requiring temperatures around 100°C and pressures near 1.0 MPa, yet they frequently struggle to exceed yields of approximately 85%. A significant drawback of these legacy processes is the substantial presence of unreacted phenol and various byproducts in the crude reaction mixture. This necessitates extensive and costly purification steps, often involving large volumes of water for washing, which generates wastewater with high Chemical Oxygen Demand (COD). The disposal of such organic-rich wastewater imposes a heavy load on environmental treatment facilities and increases the overall operational expenditure. Furthermore, the inability to efficiently recover and recycle expensive raw materials like hexafluoroacetone and hydrogen fluoride results in significant material loss, eroding profit margins and complicating supply chain stability for high-purity OLED material and polymer additive manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN101870641B introduces a paradigm shift by meticulously controlling the stoichiometry of the reaction system. By adjusting the molar ratio of hexafluoroacetone to phenol to a range of 0.55 to 2.0, and more preferably between 0.6 and 1.0, the process minimizes the formation of unwanted byproducts while maximizing the conversion of phenol. Crucially, the usage of hydrogen fluoride is optimized to between 8 and 200 moles per mole of phenol, acting not just as a catalyst but as a solvent that facilitates a cleaner reaction profile. This novel approach enables the reaction to proceed with greater selectivity, significantly reducing the amount of unreacted phenol that ends up in the wastewater. Additionally, the integration of a distillation step at temperatures lower than the reaction temperature allows for the separation and recovery of unreacted hexafluoroacetone and hydrogen fluoride in a form suitable for direct reuse. This closed-loop capability drastically simplifies the refining process and aligns perfectly with the goals of cost reduction in advanced materials manufacturing.
Mechanistic Insights into HF-Catalyzed Condensation
The core of this synthesis lies in the electrophilic aromatic substitution mechanism facilitated by the superacidic nature of anhydrous hydrogen fluoride. In this reaction environment, hydrogen fluoride activates the carbonyl group of hexafluoroacetone, making it highly susceptible to nucleophilic attack by the electron-rich aromatic ring of phenol. The precise control of temperature, maintained ideally between 80°C and 120°C, is critical; temperatures below 50°C result in sluggish reaction kinetics, while exceeding 200°C promotes the formation of colored impurities and secondary byproducts. The reaction proceeds through the formation of an intermediate alcohol, 2-(4-hydroxyphenyl)hexafluoro-2-propanol, which subsequently reacts with a second molecule of phenol to form the final Bisphenol AF structure. The presence of excess hydrogen fluoride ensures that the equilibrium is driven towards the product side while suppressing the polymerization of hexafluoroacetone, a common side reaction that can foul reactors and reduce overall throughput.
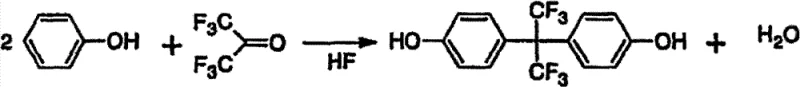
Impurity control is another vital aspect of this mechanistic pathway, particularly concerning the formation of ortho-isomers and mono-substituted intermediates. The patent data indicates that by strictly adhering to the specified molar ratios and reaction times (typically 2 to 5 hours), the formation of the undesirable 2-(2-hydroxyphenyl) isomer is kept to minimal levels, often below 3%. The subsequent purification strategy leverages the solubility differences between the target Bisphenol AF and these polar impurities. Since unreacted phenol and mono-substituted intermediates are water-soluble, a simple aqueous wash effectively removes them from the hydrophobic Bisphenol AF product. This mechanism ensures that the final product achieves purity specifications exceeding 98%, which is paramount for applications in sensitive sectors like semiconductor processing or high-performance engineering plastics where trace impurities can compromise material integrity.
How to Synthesize Bisphenol AF Efficiently
Implementing this synthesis route requires careful attention to reactor material selection and process sequencing to ensure safety and efficiency. The corrosive nature of hydrogen fluoride mandates the use of specialized alloys such as Hastelloy, Monel, or nickel-lined vessels, along with fluoropolymer gaskets and seals. The process begins with the charging of phenol and hydrogen fluoride into the reactor under cooling conditions to manage the exotherm, followed by the controlled addition of hexafluoroacetone. Maintaining the system under autogenous pressure, typically ranging from 0.2 to 2 MPa, is essential to keep the volatile reactants in the liquid phase. Detailed standardized synthetic steps, including specific heating ramps, agitation speeds, and quenching protocols, are critical for reproducibility and safety. For a comprehensive breakdown of the operational parameters and safety measures required for scale-up, please refer to the technical guide below.
- Charge a corrosion-resistant reactor with phenol and anhydrous hydrogen fluoride, maintaining a molar ratio of HF to phenol between 8: 1 and 200:1.
- Introduce hexafluoroacetone (HFA) to the mixture at a molar ratio of 0.55 to 2.0 relative to phenol, preferably between 0.6 and 1.0.
- Heat the reaction mixture to 80-120°C under autogenous pressure (0.2-2 MPa) for 2-5 hours, followed by distillation to recover unreacted HFA and HF.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized manufacturing process translates into tangible strategic benefits that extend beyond simple unit pricing. The ability to recover and recycle unreacted hexafluoroacetone and hydrogen fluoride represents a major breakthrough in raw material utilization. In traditional processes, these valuable chemicals are often lost or require energy-intensive purification before reuse. By contrast, this method allows for their recovery in a high-purity state suitable for direct re-introduction into the reaction cycle. This circular approach significantly reduces the consumption of fresh raw materials, leading to substantial cost savings over the lifecycle of production. Furthermore, the reduction in wastewater COD means lower fees for environmental treatment and a smaller regulatory footprint, enhancing the long-term viability of the supply source.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the efficient recycling of hydrogen fluoride and hexafluoroacetone drive down the variable costs of production. By minimizing the loss of high-value fluorinated reagents, manufacturers can offer more competitive pricing without compromising on quality. The simplified purification workflow also reduces energy consumption associated with distillation and drying, contributing to a leaner cost structure. These efficiencies allow for a more resilient pricing model that can better withstand fluctuations in the global commodity markets for fluorine chemicals.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route ensures consistent output quality, which is critical for maintaining uninterrupted production schedules for downstream customers. The ability to operate with a wider margin of safety regarding reactant ratios reduces the risk of batch failures due to minor feedstock variations. Moreover, the reduced dependency on complex waste treatment infrastructure minimizes the risk of production stoppages due to environmental compliance issues. This reliability makes the manufacturer a preferred partner for long-term contracts requiring steady volumes of high-purity intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated in autoclave systems that mimic industrial batch reactors. The significant reduction in organic load in the wastewater simplifies the requirements for effluent treatment plants, making it easier to obtain and maintain environmental permits. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening regulations but also appeals to end-users who prioritize sustainable sourcing in their supply chains. The ease of scaling from pilot to commercial tonnage ensures that supply can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Bisphenol AF using this specific HF-catalyzed methodology. These insights are derived directly from the experimental data and claims found in the underlying patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their own formulations or supply networks. The answers reflect the balance between high-yield chemistry and practical engineering constraints found in modern fine chemical manufacturing.
Q: What are the optimal molar ratios for Bisphenol AF synthesis to minimize waste?
A: According to patent CN101870641B, using a hexafluoroacetone to phenol ratio of 0.6-1.0 and a hydrogen fluoride to phenol ratio of 10-50 significantly reduces unreacted phenol, lowering the COD of wastewater.
Q: How does this process improve upon traditional Bisphenol AF manufacturing methods?
A: Traditional methods often yield around 85% with high organic waste. This optimized process achieves yields exceeding 92% and allows for the efficient recovery and reuse of expensive hexafluoroacetone and hydrogen fluoride.
Q: What purification steps are required to achieve high-purity Bisphenol AF?
A: The process involves distilling off volatile components (HFA/HF) at temperatures lower than the reaction temperature, followed by water washing to remove soluble impurities, resulting in purity levels above 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol AF Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation fluoropolymers and electronic materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering Bisphenol AF that meets stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our facility is designed to handle corrosive fluorine chemistry safely and efficiently, adhering to the highest international standards for safety and environmental protection.
We invite you to collaborate with us to optimize your supply chain for fluorine-based materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project goals. Let us be your partner in achieving both technical excellence and commercial success in the rapidly evolving field of advanced fluorine materials.
