Advanced Synthesis of 5-DNSA: Enhancing Labetalol Intermediate Quality and Commercial Scalability
The escalating global demand for cardiovascular therapeutics has placed intense scrutiny on the supply chain reliability of key pharmaceutical intermediates, particularly those required for the synthesis of Labetalol Hydrochloride. Patent CN101560171A introduces a transformative methodology for preparing 5-(N,N-dibenzylglycyl) salicylamide, a critical precursor that directly influences the efficacy and safety profile of the final antihypertensive agent. This technical disclosure addresses longstanding inefficiencies in traditional synthetic routes by optimizing reaction conditions to achieve yields consistently exceeding 85% while maintaining purity levels above 99.0%. For R&D Directors and Procurement Managers, understanding the nuances of this patented approach is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory standards. The innovation lies not merely in the chemical transformation but in the holistic process design that mitigates hazardous byproduct formation and simplifies downstream purification protocols. By leveraging this advanced chemistry, manufacturers can significantly enhance the cost reduction in API manufacturing while ensuring a robust supply of high-purity labetalol intermediate for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of this key intermediate relied heavily on the 5-acetobrom wintergreen oil method, a pathway fraught with significant operational and economic drawbacks that hindered efficient commercial scale-up of complex pharmaceutical intermediates. As illustrated in the reaction scheme below, this legacy process necessitates a multi-step sequence involving esterification followed by ammonolysis, resulting in a cumulative yield that rarely surpasses 31% due to material losses at each stage. Furthermore, this route generates persistent impurities such as 5-(N,N-dibenzyl amino acetyl) Whitfield's ointment derivatives and methyl ester variants, which are notoriously difficult to remove and often exceed the strict 0.3% threshold required for pharmaceutical grade materials. The reliance on harsh reaction conditions and extended process cycles also escalates energy consumption and waste generation, creating substantial environmental compliance burdens for production facilities. These technical bottlenecks translate directly into higher procurement costs and unpredictable lead times for downstream drug manufacturers seeking consistent quality. Consequently, the industry has urgently required a streamlined alternative that eliminates these structural inefficiencies and impurity risks.
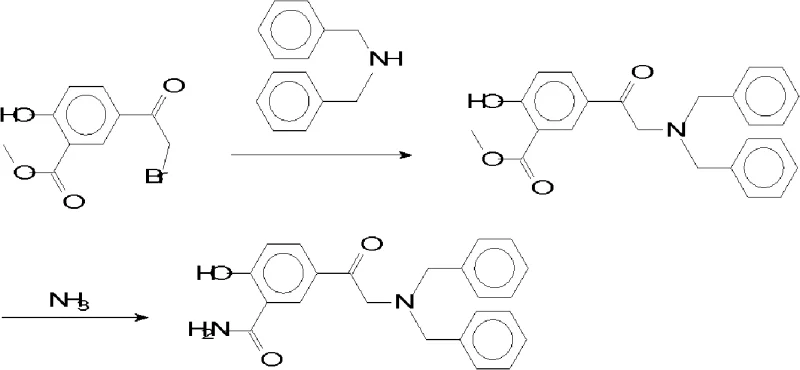
The Novel Approach
In stark contrast to the cumbersome ester-based pathways, the novel approach detailed in the patent utilizes a direct amination strategy starting from 5-acetobrom salicylamide, fundamentally restructuring the synthesis logic to prioritize atom economy and operational simplicity. This method bypasses the need for protective group manipulations and ester hydrolysis steps, thereby shortening the overall production timeline and reducing the equipment footprint required for manufacturing. By employing a specific molar ratio of dibenzylamine to substrate greater than or equal to 2, the reaction drives the equilibrium towards the desired product while minimizing the presence of unreacted starting materials that complicate purification. The implementation of a methanol-water solvent system represents a critical deviation from traditional anhydrous organic solvents, offering a safer and more cost-effective medium that facilitates easier product isolation through centrifugation. This strategic shift not only enhances the yield to impressive levels ranging between 85% and 90% but also ensures that the final product meets the rigorous appearance and melting point specifications demanded by international pharmacopoeias. Such improvements are vital for partners aiming for cost reduction in electronic chemical manufacturing or pharma sectors where margin compression is a constant challenge.
Mechanistic Insights into Dibenzylamine Nucleophilic Substitution
The core chemical innovation of this process resides in the precise control of nucleophilic substitution dynamics between the bromoacetyl group and the dibenzylamine nitrogen center within a biphasic solvent environment. The selection of methanol as a polar solvent aids in the solvation of reactants, while the introduction of water plays a pivotal role in dissolving the dibenzylamine hydrobromide salt formed during the reaction, effectively pulling the equilibrium forward and preventing the accumulation of acidic byproducts that could catalyze degradation. Crucially, the protocol mandates a feed temperature of ≤40°C during the addition of dibenzylamine, a constraint designed to suppress competitive side reactions at the phenolic hydroxyl and amide functionalities which are susceptible to alkylation under highly basic or thermal stress conditions. Following the addition, refluxing at 65°C provides the necessary activation energy to complete the substitution without inducing thermal decomposition of the sensitive salicylamide backbone. This delicate balance of thermodynamic and kinetic parameters ensures that the reaction proceeds with high selectivity, minimizing the formation of dibromo impurities and other structurally related analogues that compromise the safety profile of the intermediate. Understanding these mechanistic subtleties is paramount for technical teams aiming to replicate this high-purity OLED material or pharma intermediate synthesis with consistency.
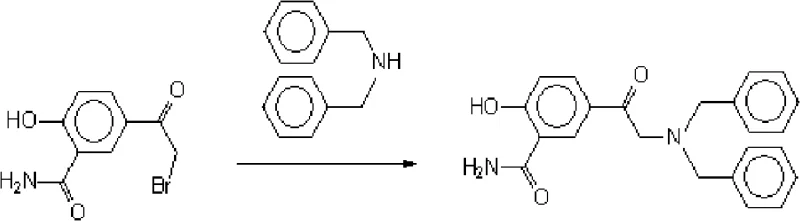
Impurity control is further augmented by the specific refinement process utilizing ethyl acetate, which selectively crystallizes the target molecule while leaving soluble impurities in the mother liquor. The patent data indicates that this recrystallization step is effective in reducing specific impurities like 5-(N,N-dibenzyl amino acetyl) wintergreen oil to levels as low as 0.01%, far below the acceptable limit of 0.3%. This level of purity is achieved without the need for expensive chromatographic separations or transition metal catalysts, which often introduce heavy metal contamination risks requiring additional scavenging steps. The absence of transition metals also simplifies the regulatory filing process for the final drug substance, as elemental impurity testing becomes less burdensome. For supply chain stakeholders, this translates to a more robust quality assurance framework where batch-to-batch variability is minimized through deterministic chemical controls rather than corrective post-processing. The result is a manufacturing process that is inherently stable and capable of delivering reducing lead time for high-purity pharmaceutical intermediates to meet just-in-time production schedules.
How to Synthesize 5-(N,N-dibenzylglycyl) salicylamide Efficiently
The practical implementation of this synthesis route requires strict adherence to the standardized operating procedures outlined in the patent to ensure reproducibility and safety across different production scales. Operators must prioritize the slow addition of reagents and precise temperature monitoring to maintain the reaction within the specified thermal window, as deviations can lead to exothermic events or increased impurity loads. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive walkthrough of the material charging sequences, agitation rates, and workup procedures necessary for successful execution. By following these guidelines, production teams can mitigate operational risks and maximize the recovery of valuable intermediates from the reaction mass. This structured approach is essential for maintaining the integrity of the supply chain and ensuring that every kilogram produced meets the stringent specifications required for cardiovascular drug manufacturing.
- Prepare 5-acetobrom salicylamide via controlled acylation of salicylamide followed by precise bromination using triethylamine to suppress dibromo impurities.
- React 5-acetobrom salicylamide with dibenzylamine in a methanol-water solvent system maintaining feed temperature below 40°C to prevent side reactions.
- Isolate the crude product via centrifugation and refine using ethyl acetate recrystallization to achieve purity levels exceeding 99.0%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages for procurement managers and supply chain heads tasked with optimizing the cost structure and reliability of their raw material sourcing strategies. The elimination of complex multi-step sequences and the reduction in solvent usage directly contribute to significant cost savings in manufacturing, allowing for more competitive pricing models without sacrificing quality margins. Furthermore, the use of readily available commodity chemicals such as methanol, water, and ethyl acetate reduces dependency on specialized or volatile reagent markets, thereby enhancing supply chain resilience against geopolitical or logistical disruptions. The high yield and purity profile minimize the need for reprocessing or scrapping off-spec batches, which traditionally erode profitability and strain production capacity. These factors collectively create a more predictable and efficient supply environment, enabling partners to plan their inventory levels with greater confidence and reduce the working capital tied up in safety stock. Ultimately, this process represents a strategic asset for companies seeking to secure a reliable pharmaceutical intermediates supplier for long-term partnerships.
- Cost Reduction in Manufacturing: The streamlined single-step amination process eliminates the need for expensive esterification reagents and subsequent hydrolysis catalysts, drastically simplifying the bill of materials and reducing overall chemical consumption. By avoiding the use of transition metal catalysts, the process removes the costly and time-consuming heavy metal removal steps typically required in pharmaceutical synthesis, leading to substantial cost savings in downstream processing. The high conversion efficiency ensures that raw material utilization is maximized, minimizing waste disposal costs and improving the overall economic viability of the production line. Additionally, the ability to recover and recycle solvents like methanol and ethyl acetate further contributes to lowering the variable costs associated with large-scale manufacturing operations.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity solvents and common organic starting materials ensures that production is not bottlenecked by the availability of niche or regulated chemicals, fostering a more resilient supply network. The robustness of the reaction conditions allows for flexible scheduling and rapid scale-up capabilities, enabling suppliers to respond quickly to fluctuations in market demand without compromising product quality. Reduced process complexity also lowers the risk of operational failures or shutdowns, ensuring a continuous flow of materials to downstream customers. This stability is critical for maintaining the production schedules of life-saving medications where interruptions can have severe consequences for patient care and regulatory compliance.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard equipment such as centrifuges and distillation columns that are readily available in most chemical manufacturing facilities. The aqueous workup and reduced solvent load significantly lower the volume of hazardous waste generated, aligning with increasingly stringent environmental regulations and sustainability goals. The absence of toxic heavy metals simplifies wastewater treatment requirements and reduces the environmental footprint of the manufacturing site. These green chemistry attributes not only facilitate regulatory approvals but also enhance the corporate social responsibility profile of the supply chain partners involved in the production of these vital intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology for 5-DNSA production. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality benchmarks associated with this method. Understanding these details is crucial for technical evaluators and procurement specialists assessing the feasibility of integrating this intermediate into their existing supply chains. The answers provided reflect the consensus on best practices for achieving optimal yield and purity while maintaining cost efficiency and regulatory compliance.
Q: How does the new solvent system improve impurity profiles compared to conventional methods?
A: The use of a methanol-water mixture effectively dissolves dibenzylamine salts and controls the basicity of the reaction environment, significantly reducing the formation of phenolic hydroxyl side-reaction products that plague traditional anhydrous methods.
Q: What are the critical temperature controls required for high-yield production?
A: Maintaining the feed temperature at or below 40°C during the addition of dibenzylamine is crucial to prevent exothermic runaway and subsequent degradation, while refluxing at 65°C ensures complete conversion without compromising structural integrity.
Q: Is this process suitable for large-scale commercial manufacturing of Labetalol intermediates?
A: Yes, the process utilizes common industrial solvents like methanol and ethyl acetate and relies on standard unit operations such as centrifugation and distillation, making it highly adaptable for multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(N,N-dibenzylglycyl) salicylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving cardiovascular medications like Labetalol. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-(N,N-dibenzylglycyl) salicylamide delivered meets the highest international standards. We are committed to supporting your R&D and commercial goals through transparent communication, technical expertise, and a dedication to continuous improvement in our manufacturing processes. Partnering with us means gaining access to a supply chain that is both robust and responsive to the dynamic needs of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements and timelines. Request a Customized Cost-Saving Analysis today to understand how our optimized synthesis routes can enhance your bottom line while ensuring supply security. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us collaborate to drive innovation and efficiency in your pharmaceutical manufacturing operations, ensuring a healthier future for patients worldwide through reliable access to essential medicines.
