Advanced Aqueous Synthesis of Metamifop Intermediates for Commercial Scale Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN111732554A introduces a groundbreaking methodology for the production of the metamifop intermediate, specifically (R)-2-(4-(6-chloro-2-benzoxazolyloxy)phenoxy)propionic acid. This technical disclosure represents a significant paradigm shift from traditional organic solvent-based systems to a green, aqueous-phase synthesis protocol. By utilizing water as the primary reaction medium and employing a synergistic combination of phase transfer catalysis and mechanical grinding, this method addresses critical pain points regarding environmental compliance and product quality. For R&D directors and procurement strategists, this patent offers a viable pathway to achieving superior purity profiles while drastically simplifying downstream processing. The ability to produce a white solid with content ≥99.0% and yield ≥96.0% using such a benign solvent system underscores the potential for substantial operational expenditure (OPEX) reductions in large-scale manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical benzoxazole derivative has been plagued by significant environmental and quality control challenges. Traditional routes often rely heavily on polar aprotic solvents such as DMF or acetonitrile, which pose severe biological toxicity risks and complicate wastewater treatment protocols due to their high solubility and stability. Furthermore, alternative methods utilizing toluene and strong bases like sodium hydroxide frequently result in product instability, leading to degradation and the formation of colored impurities. Literature indicates that prior art methods often yield gray powder solids with unsatisfactory chromaticity and purity levels hovering around 95% to 98.2%. These impurities not only necessitate costly recrystallization steps but also create bottlenecks in the supply chain due to the generation of high-salt waste streams that are difficult to treat industrially. The reliance on volatile organic compounds (VOCs) also increases safety hazards and regulatory burdens for manufacturing sites aiming for green certification.
The Novel Approach
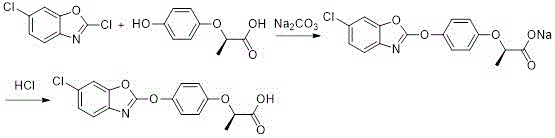
In stark contrast, the novel approach detailed in the patent leverages water as a safe, non-toxic, and inexpensive solvent, fundamentally altering the economic and environmental landscape of production. The process employs sodium carbonate as a mild base and 18-crown-6 as a phase transfer catalyst to facilitate the nucleophilic substitution between 2,6-dichlorobenzoxazole and (R)-2-(4-hydroxyphenoxy)propionic acid. Crucially, the introduction of an inert grinding medium, specifically zirconia beads, enhances the interfacial contact between reactants in the heterogeneous aqueous system. This mechanical activation allows the reaction to proceed efficiently at moderate temperatures of 50-55°C, avoiding the thermal degradation associated with harsher conditions. The result is a pristine white solid product that requires minimal purification, thereby streamlining the entire manufacturing workflow and eliminating the need for hazardous solvent recovery systems.
Mechanistic Insights into Aqueous Phase Transfer Catalysis
The core of this technological advancement lies in the sophisticated interplay between the phase transfer catalyst and the mechanical grinding media within the aqueous environment. 18-crown-6 acts as a molecular host for the sodium cations generated from sodium carbonate, effectively solubilizing the carbonate anion in the organic-like microenvironment surrounding the hydrophobic reactants. This "naked anion" effect significantly increases the nucleophilicity of the phenoxide oxygen, enabling it to attack the electron-deficient carbon of the benzoxazole ring with high precision. Simultaneously, the zirconia beads serve a dual purpose: they act as a physical grinding medium to reduce particle size and increase surface area, and they promote turbulent mixing that prevents the agglomeration of reactants. This synergy ensures that the reaction kinetics are optimized without the need for high-energy input or toxic co-solvents, resulting in a highly selective transformation that minimizes side reactions such as hydrolysis of the benzoxazole ring.
Impurity control is inherently built into this mechanism through the choice of mild reaction conditions and the specific solubility characteristics of the product. Unlike strong alkaline conditions that can degrade the benzoxazole moiety, the use of sodium carbonate at controlled temperatures preserves the structural integrity of the heterocyclic ring. The subsequent workup involves a simple pH adjustment to 3 using hydrochloric acid, which protonates the carboxylate salt to precipitate the free acid. Because the starting materials and byproducts remain soluble in the aqueous phase or are removed during the filtration of the grinding media, the precipitated product exhibits exceptional purity. This mechanistic elegance eliminates the need for complex chromatographic separations or multiple recrystallizations, ensuring a consistent impurity profile that meets the stringent specifications required for downstream agrochemical formulation.
How to Synthesize Metamifop Intermediate Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and physical parameters to maximize the benefits of the aqueous system. The patent outlines a robust protocol where the molar ratio of the hydroxy acid to sodium carbonate and the benzoxazole derivative is carefully balanced to drive the reaction to completion while minimizing excess reagent waste. The addition of the grinding media at the outset ensures that the reaction mixture remains homogeneous enough for effective heat and mass transfer throughout the process. Operators must maintain the temperature within the narrow window of 50-55°C to optimize the rate of substitution without triggering thermal decomposition. Following the reaction, the recovery of the zirconia beads via filtration is a critical step that not only cleans the product stream but also prepares the media for immediate reuse, enhancing the overall sustainability of the operation.
- Prepare the reaction mixture by combining water, 18-crown-6 catalyst, zirconia beads, (R)-2-(4-hydroxyphenoxy)propionic acid, and sodium carbonate in a reactor.
- Heat the mixture to 50-55°C and add 2,6-dichlorobenzoxazole, maintaining temperature until reaction completion.
- Filter to recover grinding media, acidify the filtrate with HCl to pH 3 to precipitate the white solid product, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous synthesis method offers profound strategic advantages beyond mere technical feasibility. The elimination of expensive and regulated organic solvents like DMF and acetonitrile directly translates to significant cost reduction in agrochemical manufacturing by removing the need for solvent recovery distillation columns and specialized VOC abatement systems. Furthermore, the use of water as a solvent drastically reduces the fire hazard classification of the production facility, potentially lowering insurance premiums and simplifying safety compliance audits. The ability to recycle the zirconia grinding media further contributes to long-term operational savings, reducing the consumption of consumable materials and minimizing solid waste disposal costs. These factors combine to create a leaner, more resilient supply chain capable of withstanding fluctuations in raw material pricing and regulatory pressures.
- Cost Reduction in Manufacturing: The shift to a water-based system eliminates the capital and operational expenditures associated with handling hazardous organic solvents. By removing the need for complex solvent recovery units and reducing the volume of hazardous waste requiring incineration, the overall cost of goods sold (COGS) is substantially optimized. Additionally, the high yield and purity achieved in a single step reduce the need for reprocessing, further driving down production costs and improving margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: Water is a universally available and inexpensive resource, insulating the manufacturing process from the volatility of petrochemical-derived solvent markets. The simplicity of the workup procedure, involving basic filtration and acidification, reduces the dependency on specialized equipment and skilled labor for complex separations. This robustness ensures consistent batch-to-batch quality and reliable delivery schedules, which are critical for maintaining just-in-time inventory levels for downstream herbicide formulators.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of standard stirred-tank reactors and the absence of exothermic risks associated with strong alkalis in organic media. The wastewater generated is primarily saline and easily treatable compared to streams contaminated with DMF or toluene, facilitating compliance with increasingly strict environmental regulations. This green chemistry profile enhances the corporate social responsibility (CSR) standing of the manufacturer, making it a preferred partner for global agrochemical companies seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for commercial production. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-making processes.
Q: What are the primary advantages of the aqueous synthesis method for metamifop intermediates?
A: The primary advantages include the elimination of toxic organic solvents like DMF and acetonitrile, resulting in easier wastewater treatment and reduced environmental impact. Additionally, the process yields a white solid with purity ≥99.0% and yield ≥96.0%, overcoming the poor chromaticity issues associated with traditional alkaline hydrolysis methods.
Q: How does the use of zirconia beads impact the reaction efficiency?
A: The inert grinding medium, specifically zirconia beads, enhances mass transfer and mixing efficiency within the aqueous phase. This mechanical assistance, combined with the phase transfer catalyst 18-crown-6, ensures uniform reaction conditions, leading to higher conversion rates and consistent product quality without the need for hazardous organic co-solvents.
Q: Is the grinding media reusable in this synthesis process?
A: Yes, the zirconia beads are recovered by simple filtration after the reaction is complete and before acidification. They can be directly reused in subsequent batches, which significantly reduces material costs and waste generation, contributing to a more sustainable and cost-effective manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metamifop Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous synthesis technology described in CN111732554A for the global agrochemical sector. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of this water-based process, including the management of grinding media and the precise control of phase transfer catalysis conditions. We are committed to delivering products with stringent purity specifications and rigorous QC labs testing every batch to guarantee consistency and performance for your final herbicide formulations.
We invite you to collaborate with us to leverage this advanced synthesis route for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green methodology can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective source of high-quality metamifop intermediates.
