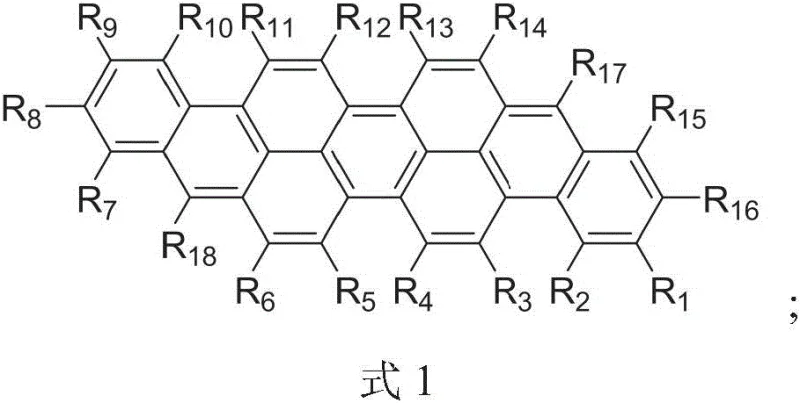
Advanced Isoviolanthrene Derivatives: Technical Breakthroughs and Commercial Scalability for OLED Applications
The landscape of organic optoelectronic materials is undergoing a significant transformation driven by the demand for higher efficiency and color purity in display technologies. Patent CN108047003B introduces a groundbreaking class of isoviolanthrene derivatives that address critical limitations found in conventional polycyclic aromatic hydrocarbons. These novel compounds are characterized by a unique structural formula allowing for extensive substitution at multiple positions, which fundamentally alters their physical and optical properties. Unlike traditional planar aromatics that suffer from poor solubility and aggregation-caused quenching, these derivatives feature a curved molecular architecture that enhances solubility and prevents detrimental pi-pi stacking interactions. This structural innovation is pivotal for applications requiring solution-processable materials, such as organic light-emitting diodes (OLEDs) and advanced fluorescent dyes. The patent details a robust synthetic pathway that enables precise molecular tailoring, ensuring that the resulting materials meet the stringent requirements of modern electronic chemical manufacturing. For industry leaders seeking a reliable OLED material supplier, understanding the technical nuances of this patent provides a strategic advantage in sourcing next-generation red emitters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
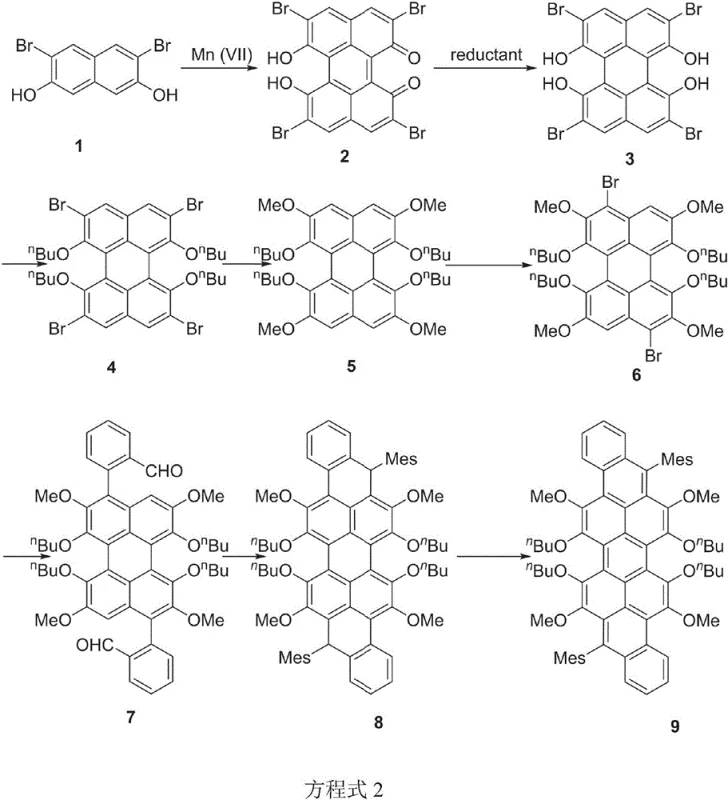
Historically, the synthesis of violanthracene and isoviolanthrene relied on methods such as zinc dust melting, which were first reported in early German literature. These conventional techniques are severely restricted by their inability to introduce functional groups onto the aromatic core, resulting in single, unsubstituted products with limited utility. The lack of substituents leads to strong intermolecular pi-pi interactions, causing severe fluorescence quenching and poor solubility in common organic solvents. Furthermore, the reaction conditions for these traditional methods are often harsh, requiring high temperatures that limit substrate adaptability and increase energy consumption. The resulting materials typically exhibit poor optical properties, such as negligible fluorescence, rendering them unsuitable for high-performance applications like electroluminescence or optical sensing. Additionally, the purification of these unsubstituted compounds often necessitates extensive recrystallization processes, which are inefficient and difficult to scale for commercial production. These inherent drawbacks create a significant bottleneck for manufacturers aiming to integrate red-emitting materials into full-color display schemes.
The Novel Approach
In stark contrast, the methodology disclosed in CN108047003B employs a sophisticated multi-step synthesis that allows for precise modification at eight distinct positions on the isoviolanthrene backbone. By utilizing tetrabromoperylene derivatives as starting materials, the process enables the introduction of diverse functional groups such as alkoxy, alkyl, and aryl substituents through palladium-catalyzed cross-coupling reactions. This approach not only improves the solubility of the final product by disrupting molecular planarity but also enhances the fluorescence quantum yield by reducing aggregation. The synthetic route is designed to be modular, allowing chemists to tune the electronic properties of the molecule to achieve specific emission wavelengths, particularly in the red region of the spectrum. Moreover, the use of mild reaction conditions and standard purification techniques like column chromatography and recrystallization makes the process more adaptable to industrial scaling. This novel strategy effectively overcomes the technical barriers associated with traditional synthesis, paving the way for the development of practical red light-emitting materials with superior thermal stability and color purity.
Mechanistic Insights into Suzuki-Miyaura Coupling and Dehydroaromatization
The core of this technological advancement lies in the strategic application of the Suzuki-Miyaura reaction to construct the complex polycyclic framework. The process begins with the halogenation of a perylene precursor, creating reactive sites for subsequent cross-coupling with polysubstituted phenylboronic acids. This step is critical as it installs the bulky side chains that prevent molecular stacking, a common issue in planar aromatic systems. The catalytic cycle involves the oxidative addition of the palladium catalyst to the carbon-halogen bond, followed by transmetallation with the boronic acid species and reductive elimination to form the new carbon-carbon bond. The precision of this reaction ensures that substituents are placed exactly where needed to optimize the electronic distribution across the conjugated system. Following the coupling, the intermediate undergoes a cyclization reaction catalyzed by Lewis acids, which closes the rings to form the distorted isoviolanthrene core. This structural distortion is key to maintaining high solubility while preserving the extended conjugation required for efficient light emission.
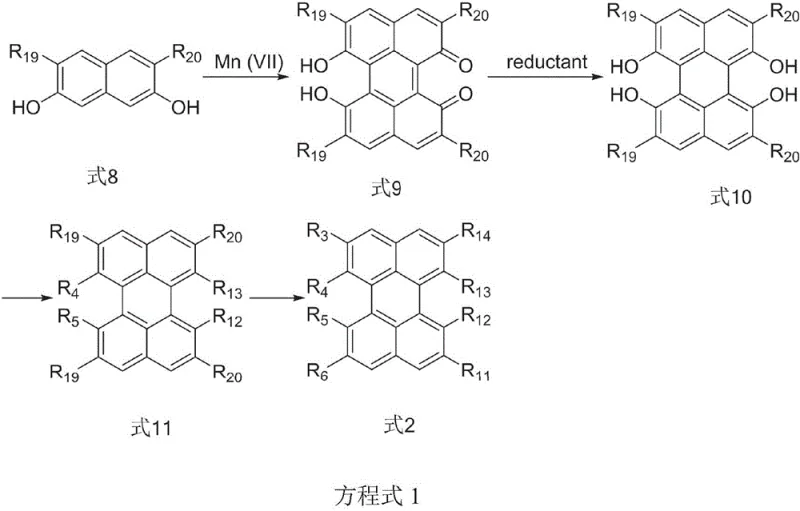
Impurity control is meticulously managed throughout the synthesis to ensure the high purity required for electronic applications. The initial oxidative coupling step uses heptavalent manganese compounds, which offer better selectivity and yield compared to traditional oxidants, minimizing the formation of side products. Subsequent purification steps, including acid precipitation and recrystallization from ethanol, effectively remove residual catalysts and unreacted starting materials. The final dehydroaromatization step, typically employing DDQ as an oxidant, is carefully monitored to ensure complete conversion to the aromatic system without over-oxidation or degradation of the sensitive functional groups. This rigorous control over the reaction pathway results in a product with a narrow emission band and high color saturation, essential characteristics for high-quality display materials. The ability to produce such high-purity compounds consistently is a major factor in reducing lead time for high-purity electronic chemical manufacturing, as it minimizes the need for extensive downstream processing.
How to Synthesize Multi-Substituted Isoviolanthrene Efficiently
The synthesis of these advanced materials follows a logical progression designed to maximize yield and purity while maintaining operational simplicity. The process starts with the preparation of the perylenequinone core, followed by sequential functionalization and ring-closing steps. Each stage is optimized to use commercially available reagents and standard laboratory equipment, making the technology accessible for scale-up. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the results.
- Perform oxidative coupling of naphthol compounds using heptavalent manganese compounds to form the perylenequinone core structure.
- Execute Suzuki-Miyaura coupling with polysubstituted phenylboronic acids to introduce functional groups at specific positions.
- Complete the synthesis via cyclization and dehydroaromatization using DDQ to obtain the final fluorescent isoviolanthrene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical performance. The shift from harsh, non-selective traditional methods to this modular catalytic approach significantly simplifies the manufacturing process, leading to drastic cost savings in electronic chemical manufacturing. By eliminating the need for extreme temperatures and specialized reduction agents like zinc dust, the process reduces energy consumption and equipment wear, directly impacting the bottom line. Furthermore, the improved solubility of the intermediates allows for easier handling and purification, which streamlines production workflows and reduces waste generation. This efficiency translates into a more reliable supply chain, as the risk of batch failure due to insolubility or purification issues is markedly decreased. The use of common reagents such as potassium permanganate and sodium methoxide ensures that raw material sourcing is stable and less susceptible to market volatility.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and the use of recyclable solvents contribute to a leaner production cost structure. The ability to purify intermediates via recrystallization rather than preparative HPLC significantly lowers operational expenses associated with solvent consumption and waste disposal. Additionally, the high yield of the oxidative coupling step minimizes raw material waste, further enhancing the economic viability of the process. These factors combined result in a competitive pricing model for high-performance OLED materials without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on widely available chemical feedstocks, reducing dependency on niche suppliers and mitigating supply chain risks. The robustness of the reaction conditions means that production can be maintained consistently even under varying environmental conditions, ensuring steady output. This reliability is crucial for meeting the tight delivery schedules of the consumer electronics industry, where delays can have cascading effects on product launches. By partnering with a supplier who utilizes this resilient manufacturing method, companies can secure a continuous flow of critical materials.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production without significant re-engineering. The reduced use of hazardous reagents and the implementation of aqueous workup procedures align with increasingly strict environmental regulations. This compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile of the end product. The ability to scale up complex polymer additives and small molecule emitters efficiently ensures that supply can meet growing global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isoviolanthrene derivatives. The answers are derived directly from the patent specifications and practical implementation data to provide clarity for decision-makers.
Q: How does this patent overcome the solubility issues of traditional isoviolanthrene?
A: The patent introduces multi-substitution at positions 3, 4, 5, 6, 11, 12, 13, and 14 using alkoxy and aryl groups. This structural modification disrupts planar stacking, significantly enhancing solubility in organic solvents compared to unsubstituted parent compounds.
Q: What are the key advantages for OLED red light-emitting materials?
A: The derivatives exhibit high fluorescence quantum yield, narrow emission bands, and excellent thermal stability. Specifically, the emission wavelength around 634nm makes them highly suitable for efficient red light-emitting layers in OLED displays.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the method utilizes standard reactions like Suzuki-Miyaura coupling and oxidation with common reagents like DDQ and potassium permanganate. The purification relies on recrystallization rather than complex chromatography, facilitating large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoviolanthrene Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of synthesizing curved aromatic systems and understands the critical importance of stringent purity specifications for optoelectronic applications. We operate rigorous QC labs equipped with state-of-the-art analytical instruments to ensure every batch meets the highest standards of quality and consistency. Our commitment to excellence ensures that clients receive materials that perform reliably in their final devices, whether for OLED displays or advanced fluorescent coatings.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our capabilities can support your product development goals. Request a Customized Cost-Saving Analysis to understand how our optimized synthetic routes can reduce your overall material costs. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Partner with us to secure a stable supply of high-performance isoviolanthrene derivatives and drive innovation in your electronic material applications.
