Revolutionizing N-cyanoethylaniline Manufacturing: A Technical Breakdown of Clean Production and Commercial Scalability
The global demand for high-purity dye intermediates is driving a significant shift towards sustainable manufacturing protocols, as evidenced by the technological advancements detailed in Chinese patent CN102241606B. This specific intellectual property outlines a groundbreaking clean production method for N-cyanoethylaniline, a critical precursor in the synthesis of disperse dyes such as C.I. Disperse Orange 30 and C.I. Disperse Orange 288. Unlike traditional synthetic routes that struggle with excessive wastewater generation and difficult-to-remove impurities, this patented approach leverages a sophisticated combination of acid catalysis and steam distillation to achieve exceptional product quality. For R&D directors and procurement specialists alike, understanding the nuances of this process is vital, as it represents a tangible move away from high-pollution batch processing toward a more circular, efficient chemical economy. The core innovation lies not just in the reaction itself, but in the downstream processing which systematically recovers valuable raw materials like acrylonitrile and aniline, thereby transforming what was once waste into a reusable resource.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-cyanoethylaniline has been plagued by significant environmental and efficiency bottlenecks that hinder scalable operations. Conventional techniques typically employ acetic acid or hydrochloric acid alongside zinc chloride to catalyze the Michael addition between aniline and acrylonitrile, followed by a crude workup involving water addition and cooling crystallization. While these methods can achieve yields around 94%-95%, they suffer from severe drawbacks regarding impurity profiles and waste management. Specifically, the resulting product often contains 3%-4% of the unwanted byproduct N,N-dicyanoethylaniline, alongside substantial residual aniline that is difficult to eliminate through simple washing. Furthermore, the environmental footprint is staggering; for every ton of product manufactured, approximately 4 tons of mother liquor with a Chemical Oxygen Demand (COD) exceeding 80,000 mg/L are generated, creating a massive burden on wastewater treatment facilities and escalating operational costs due to disposal fees.
The Novel Approach
In stark contrast, the methodology described in patent CN102241606B introduces a paradigm shift by integrating steam distillation and precise pH control to overcome these legacy issues. Instead of relying solely on crystallization, the novel process subjects the reaction mixture to steam bubbling distillation at temperatures between 100°C and 150°C after the initial separation of the aqueous layer. This thermal treatment is highly effective at volatilizing and removing residual aniline, reducing its content to negligible levels of ≤0.05%, which is critical for preventing discoloration in downstream dye applications. Moreover, the process incorporates a strategic recycling loop where the distillate is extracted with benzene to recover unreacted aniline and product, while the separated mother liquor water is reused in subsequent batches. This holistic approach not only enhances the purity of the final N-cyanoethylaniline to 96%-97% but also dramatically curtails the volume of high-concentration wastewater discharged, aligning perfectly with modern green chemistry mandates.
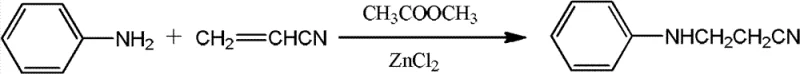
Mechanistic Insights into HCl/ZnCl2-Catalyzed Michael Addition
The core chemical transformation in this process is a classic Michael addition, where the nucleophilic nitrogen of aniline attacks the electron-deficient beta-carbon of acrylonitrile. The presence of hydrochloric acid and zinc chloride serves a dual purpose: protonating the nitrile group to increase its electrophilicity and stabilizing the transition state through Lewis acid coordination. In the patented procedure, the reaction is initiated at 60°C and gradually ramped to 95°C over a period of 10 hours, ensuring controlled kinetics that minimize the formation of the double-addition byproduct, N,N-dicyanoethylaniline. The careful regulation of temperature is paramount; excessive heat can accelerate the secondary addition of acrylonitrile to the mono-substituted product, whereas insufficient heat leads to incomplete conversion. By maintaining this specific thermal profile and utilizing a molar excess of aniline relative to acrylonitrile, the process inherently favors the formation of the desired mono-cyanoethylated species, setting the stage for high selectivity before purification even begins.
Beyond the reaction kinetics, the purification mechanism relies heavily on the physical properties of the components involved, specifically the volatility differences between aniline and the heavier N-cyanoethylaniline. The introduction of steam distillation acts as a continuous extraction process, where water vapor carries the volatile aniline out of the organic phase. This is chemically superior to water washing because it physically removes the impurity rather than just diluting it. Following the distillation, the mixture is neutralized with sodium bicarbonate or sodium carbonate to a pH of 5-8. This step is crucial for quenching any remaining acidic catalyst and preventing the hydrolysis of the nitrile group, which could occur under strongly acidic or basic conditions at elevated temperatures. The result is a chemically stable, high-purity intermediate that meets the stringent specifications required for the synthesis of sensitive azo dyes, ensuring consistent color strength and hue in the final textile applications.
How to Synthesize N-cyanoethylaniline Efficiently
Implementing this synthesis route requires precise adherence to the sequential unit operations defined in the patent to maximize yield and minimize waste. The process begins with the charging of bottom water, zinc chloride, hydroquinone (as a polymerization inhibitor), and industrial hydrochloric acid into the reactor, followed by the slow addition of aniline and acrylonitrile. Once the reaction reaches completion, indicated by the stabilization of temperature and exotherm, the mixture is allowed to settle for phase separation. The detailed standardized synthesis steps, including specific mass ratios for catalysts and precise distillation parameters, are outlined in the technical guide below to ensure reproducibility at scale.
- Conduct the addition reaction of aniline and acrylonitrile catalyzed by HCl and ZnCl2 at 60-95°C, then separate the aqueous layer.
- Neutralize the mixture with sodium bicarbonate to pH 4-7 and recover unreacted acrylonitrile under vacuum.
- Perform steam distillation at 100-150°C to reduce aniline content below 0.05%, followed by final neutralization to pH 5-8.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this clean production technology offers profound strategic advantages that extend far beyond simple unit cost calculations. The primary value driver is the drastic reduction in raw material consumption achieved through the closed-loop recycling system. By recovering unreacted acrylonitrile under vacuum and extracting aniline from the distillate for reuse, the process significantly lowers the net input of expensive feedstocks per kilogram of finished product. This internal recycling capability insulates the supply chain from volatile market fluctuations in aniline and acrylonitrile prices, providing a more predictable cost structure for long-term contracts. Furthermore, the elimination of high-COD wastewater discharge reduces the reliance on external waste treatment services, which are becoming increasingly costly and regulated in major chemical manufacturing hubs, thereby enhancing the overall economic viability of the operation.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived principally from the optimization of material efficiency and the simplification of the purification train. By eliminating the need for extensive crystallization and washing steps that consume large volumes of fresh water and energy, the operational expenditure is substantially lowered. Additionally, the recovery of valuable organic solvents like benzene from the extraction phase further contributes to cost savings, creating a leaner manufacturing model. The avoidance of expensive transition metal catalysts, which often require specialized scavenging resins to meet ppm-level specifications, means that the downstream processing is both faster and less capital intensive, allowing for better margin retention in competitive markets.
- Enhanced Supply Chain Reliability: From a logistics and continuity perspective, this method offers superior robustness due to its reliance on commodity chemicals and standard unit operations. The use of industrial-grade hydrochloric acid and zinc chloride ensures that raw material sourcing is not constrained by niche supplier limitations, mitigating the risk of supply disruptions. The ability to recycle mother liquor water internally reduces the facility's dependency on external fresh water supplies, a critical factor in regions prone to drought or strict water usage quotas. This self-sufficiency translates to higher plant uptime and more reliable delivery schedules for customers, ensuring that production targets are met consistently without being hampered by utility constraints or waste disposal bottlenecks.
- Scalability and Environmental Compliance: As regulatory frameworks tighten globally regarding industrial effluent, the scalability of this process is a key asset for future-proofing production capacity. The design inherently minimizes the generation of hazardous waste, making it easier to obtain and maintain environmental permits for expansion. The simplified workflow, which combines reaction and purification in a streamlined sequence, facilitates easier scale-up from pilot plants to multi-ton commercial reactors without the need for complex re-engineering. This compliance-ready architecture not only protects the manufacturer from potential fines but also appeals to end-users in the dye and pigment industry who are increasingly auditing their supply chains for sustainability credentials and carbon footprint reduction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean production technology. These insights are derived directly from the experimental data and process descriptions found within the patent documentation, providing a factual basis for evaluating the feasibility of adopting this route for your specific manufacturing needs. Understanding these details is essential for making informed decisions about process integration and quality assurance protocols.
Q: How does the new process reduce aniline residue compared to traditional crystallization?
A: Traditional methods rely on cooling crystallization which often traps residual aniline. This patent utilizes steam bubbling distillation at 100-150°C, effectively stripping volatile aniline until content is ≤0.05%, ensuring superior purity for downstream dye synthesis.
Q: What is the environmental impact regarding wastewater COD in this method?
A: Conventional processes generate high-COD mother liquor (approx. 80,000 mg/L). This method implements a closed-loop recycling system where mother liquor water and recovered residues are reused in subsequent batches, drastically minimizing fresh water consumption and hazardous discharge.
Q: Can the catalyst system be optimized for large-scale production?
A: Yes, the use of industrial hydrochloric acid (31%) and zinc chloride is cost-effective and scalable. The process avoids expensive transition metals, simplifying post-reaction cleanup and allowing for robust commercial scale-up without complex metal scavenging steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-cyanoethylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to cleaner, more efficient chemical processes is not just a regulatory requirement but a strategic imperative for maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102241606B are fully realized in practical, industrial settings. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of N-cyanoethylaniline meets the exacting standards required for high-performance dye synthesis, guaranteeing consistency and reliability for our partners.
We invite you to engage with our technical procurement team to discuss how we can tailor this advanced synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your operation. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and sustainability can drive value for your supply chain.
