Advanced Synthesis of N,N'-Diphenyl-N-(9,9-dimethyl-2-fluorenyl) Derivatives for OLEDs
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that offer superior thermal stability and controlled morphological properties to ensure long device lifespans. Patent CN101987823B discloses a groundbreaking synthesis method for N,N'-diphenyl-N-(9,9-dimethyl-2-fluorenyl)-N'-(9',9'-dimethyl-7'-(2''-naphthyl)-2'-fluorenyl)-benzidine, a specialized hole transport material designed to address these critical performance metrics. This technical insight report analyzes the proprietary three-segment synthetic strategy outlined in the patent, which utilizes 9,9-dimethyl-2-acetyl aminofluorene, 9,9-dimethyl-2-bromine fluorine, and 2-bromonaphthalene as key starting materials. By strategically employing Ullmann condensation and Suzuki coupling reactions, the process achieves a final product with purity exceeding 99%, demonstrating significant potential for scalable industrial application in the display sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for triarylamine-based hole transport materials often struggle with balancing thermal stability and crystallinity, which are paramount for the longevity of OLED devices. Conventional methods frequently result in materials with high molecular symmetry that promote excessive crystallization, leading to morphological instability under the heat generated during device operation. This high crystallinity allows an overabundance of holes to penetrate the luminescent layer, causing degradation of the emissive materials and significantly shortening the operational life of the display panel. Furthermore, older synthetic pathways may rely on harsh conditions or complex purification steps that introduce impurities, compromising the electronic properties of the final film and resulting in inconsistent batch-to-batch performance for manufacturers.
The Novel Approach
The innovative approach detailed in the patent overcomes these deficiencies by introducing a naphthyl group into the molecular structure, effectively breaking the molecular symmetry to reduce crystallinity while enhancing thermal stability. This structural modification ensures that the material maintains an amorphous state more effectively, preventing the formation of crystals that could disrupt charge transport or damage adjacent layers. The synthesis is divided into three distinct segments that are independently optimized before being coupled, allowing for precise control over impurities at each stage. This modular strategy not only improves the overall yield but also simplifies the purification process, making it a robust solution for the mass production of high-performance electronic chemicals.
Mechanistic Insights into CuI-Catalyzed Ullmann and Pd-Catalyzed Suzuki Couplings
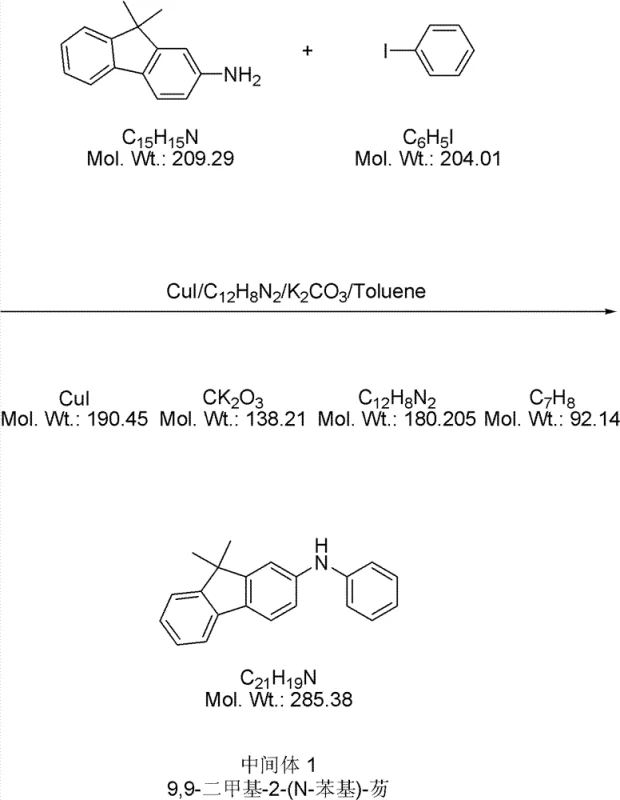
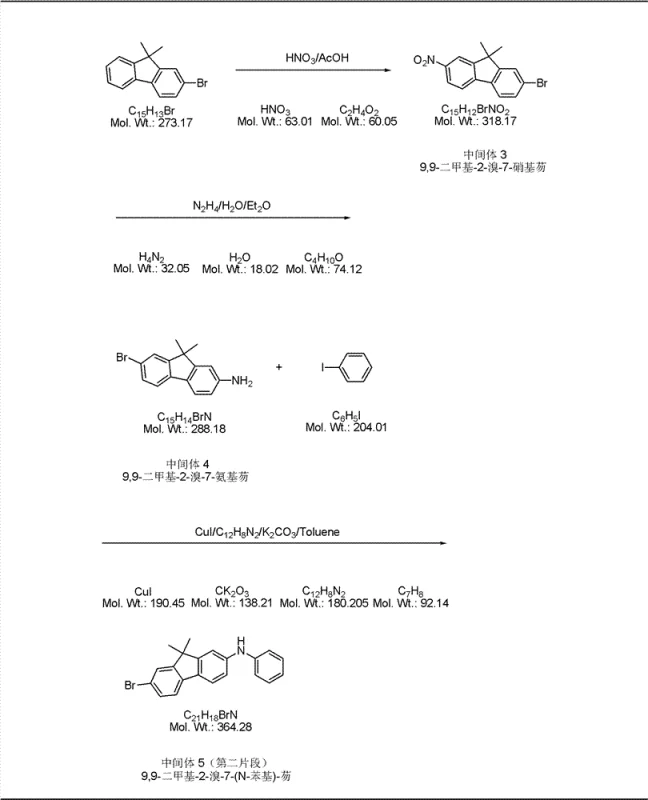
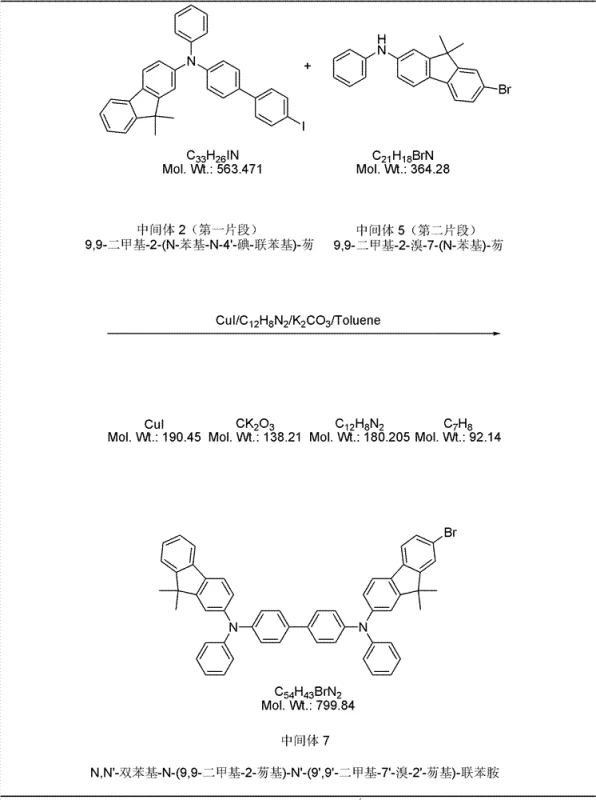
The core of this synthesis relies on the precise execution of copper-catalyzed Ullmann condensation reactions to build the nitrogen-carbon bonds essential for the triarylamine backbone. In the formation of Intermediate 1 and Intermediate 2, cuprous iodide (CuI) acts as the catalyst in the presence of 1,10-phenanthroline and potassium carbonate within a toluene solvent system. This catalytic cycle facilitates the nucleophilic attack of the amine on the aryl halide, forming the C-N bond under reflux conditions. The choice of ligands and base is critical here, as it stabilizes the copper species and promotes the oxidative addition and reductive elimination steps necessary for high conversion rates without requiring excessively high temperatures that could degrade sensitive functional groups.
Following the construction of the amine fragments, the final assembly utilizes a palladium-catalyzed Suzuki coupling reaction to link the fluorenyl-biphenyl core with the naphthyl group. This cross-coupling mechanism involves the reaction of an aryl halide (Intermediate 7) with an aryl boronic acid (Intermediate 6) in the presence of tetrakis(triphenylphosphine)palladium(0). The reaction proceeds through a catalytic cycle involving oxidative addition of the halide to the palladium center, transmetallation with the boronate species activated by the base, and finally reductive elimination to form the carbon-carbon bond. This step is crucial for introducing the sterically bulky naphthyl group that defines the material's low-crystallinity characteristics, and the mild conditions (90-100°C) ensure the integrity of the complex molecular architecture is maintained throughout the process.
How to Synthesize N,N'-diphenyl-N-(9,9-dimethyl-2-fluorenyl) Derivatives Efficiently
The synthesis protocol described in the patent offers a streamlined pathway for producing this complex OLED material through a sequence of eight well-defined steps. The process begins with the preparation of three key fragments: the first fragment involves double Ullmann coupling, the second fragment requires nitration and reduction followed by coupling, and the third fragment is a boronic acid derived from a Grignard reaction. These fragments are then sequentially coupled to build the final molecule. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Synthesize Intermediate 1 and 2 via Ullmann condensation of 9,9-dimethyl-2-aminofluorene with iodobenzene and 4,4'-diiodobiphenyl using CuI catalyst.
- Prepare Intermediate 5 by nitration and reduction of 9,9-dimethyl-2-bromofluorene followed by Ullmann coupling with iodobenzene.
- Generate Intermediate 6 (2-naphthalene boronic acid) via Grignard reaction of 2-bromonaphthalene followed by boration.
- Couple Intermediates 2 and 5 to form Intermediate 7, then perform final Suzuki coupling with Intermediate 6 to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits regarding cost efficiency and supply reliability. The modular nature of the three-segment synthesis allows for parallel processing of intermediates, which significantly reduces the overall lead time compared to linear synthesis methods. Additionally, the use of relatively common starting materials like 9,9-dimethyl-2-aminofluorene and 2-bromonaphthalene ensures a stable supply chain that is less susceptible to the volatility associated with exotic or highly specialized precursors. The high purity achieved (>99%) through standard recrystallization techniques minimizes the need for expensive and time-consuming chromatographic purifications on a large scale, directly translating to lower manufacturing costs.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of cost-effective catalysts like CuI and Pd(PPh3)4 contribute to a leaner production cost structure. By achieving high yields in each segment and utilizing simple solvent systems like toluene and ethanol, the process avoids the financial burden of specialized reagents or extreme reaction conditions. This efficiency allows for substantial cost savings in electronic chemical manufacturing, making the final OLED material more competitive in the marketplace without sacrificing performance quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available bulk chemicals for the starting materials ensures that production schedules are not disrupted by raw material shortages. The robustness of the Ullmann and Suzuki reactions means that the process is tolerant to minor variations in input quality, reducing the rejection rate of batches. This reliability is critical for maintaining continuous supply to display panel manufacturers who require consistent material availability to meet their own production targets and market demands.
- Scalability and Environmental Compliance: The synthesis is designed with scalability in mind, utilizing standard reflux and filtration operations that are easily transferred from laboratory to pilot and commercial scales. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through established recovery and treatment protocols, ensuring compliance with environmental regulations. The high atom economy of the coupling reactions further supports sustainable manufacturing practices by minimizing waste generation relative to the amount of product produced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this advanced hole transport material. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for integration into their supply chains.
Q: How does the introduction of the naphthyl group affect the material properties?
A: The introduction of the naphthyl group reduces molecular symmetry, which effectively lowers crystallinity and improves thermal stability, thereby extending the operational lifespan of OLED devices.
Q: What is the purity level achievable with this synthesis method?
A: The patented method achieves a product purity of greater than 99%, making it highly suitable for industrial production of high-performance electronic materials.
Q: Why is low crystallinity important for hole transport materials?
A: Low crystallinity prevents excessive holes from entering the luminescent layer, which protects the luminescent properties and significantly prolongs the service life of the OLED material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-diphenyl-N-(9,9-dimethyl-2-fluorenyl) Derivatives Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring complex synthetic routes like this from the laboratory to full-scale commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of OLED material meets the exacting standards required for high-end display applications, providing you with a partner dedicated to quality and performance.
We invite you to collaborate with us to optimize your material sourcing strategy and leverage the benefits of this advanced synthesis technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your next-generation OLED projects and drive your business forward.
