Advanced Synthesis of 2,7-Dibromo-N-Perfluorobutylethylcarbazole for High-Performance OLED Applications
Advanced Synthesis of 2,7-Dibromo-N-Perfluorobutylethylcarbazole for High-Performance OLED Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands continuous innovation in small molecule materials, particularly those offering enhanced stability and charge transport properties. Patent CN103232385A introduces a significant advancement in this domain by disclosing a robust preparation method for 2,7-dibromo-N-perfluoro-butylethylcarbazole. This specific fluorinated carbazole derivative represents a critical building block for next-generation photoelectric materials, addressing the limitations of traditional carbazole scaffolds. By shifting the substitution pattern from the conventional 3,6-positions to the 2,7-positions, the material achieves a larger conjugated system, which is pivotal for improving thermostability and photoconductivity. Furthermore, the strategic incorporation of a perfluoro butyl ethyl chain exploits the inherent hydrophobicity of fluorine, endowing the final product with exceptional water resistance, a crucial factor for the longevity of electronic devices.
For R&D directors and procurement specialists evaluating new supply chains, understanding the synthetic accessibility of such complex intermediates is paramount. The patented route outlined in CN103232385A offers a streamlined three-step sequence starting from readily available 4,4'-dibromobiphenyl. This approach not only simplifies the manufacturing workflow but also ensures high purity standards essential for electronic grade chemicals. As the market for high-performance display and lighting technologies expands, securing a reliable source of specialized intermediates like 2,7-dibromo-N-perfluoro-butylethylcarbazole becomes a strategic imperative. This report delves into the technical nuances of this synthesis, contrasting it with conventional methods to highlight its commercial viability and potential for cost reduction in OLED material manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbazole-based OLED materials has predominantly focused on modifications at the 3, 6, and 9 positions. This preference stems from the fact that the hydrogen atoms at the 3 and 6 positions of the carbazole ring are chemically activated, making them susceptible to electrophilic substitution reactions. Consequently, achieving substitution at the 2 and 7 positions has been synthetically challenging and often requires harsh conditions or multi-step protection-deprotection strategies that lower overall yields. Furthermore, standard carbazole derivatives often lack sufficient environmental stability, particularly regarding moisture sensitivity, which can degrade device performance over time. Traditional routes may also rely on expensive catalysts or difficult-to-source precursors, creating bottlenecks in the supply chain for high-purity OLED material production. These factors collectively increase the cost of goods sold and complicate the scale-up process for manufacturers aiming to meet the rigorous demands of the optoelectronics sector.
The Novel Approach
The methodology presented in patent CN103232385A circumvents these historical hurdles by utilizing a clever precursor strategy starting with 4,4'-dibromobiphenyl. Instead of attempting direct substitution on a pre-formed carbazole ring, the process constructs the carbazole core via a reductive cyclization of a nitro-biphenyl intermediate. This allows for precise control over the substitution pattern, ensuring the bromine atoms are locked in the desired 2,7-positions before the ring closure occurs. Additionally, the final alkylation step introduces the perfluoro butyl ethyl group, which not only modifies the electronic properties but also imparts significant hydrophobicity. This novel approach transforms a complex structural challenge into a manageable three-step sequence using commodity chemicals. By avoiding exotic reagents and leveraging standard organic transformations like nitration and nucleophilic substitution, the process offers a clear pathway for cost reduction in photoelectric material manufacturing while delivering a superior product profile.
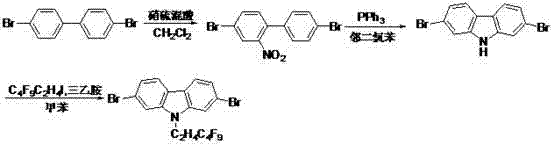
Mechanistic Insights into Triphenylphosphine-Mediated Reductive Cyclization
The core of this synthetic strategy lies in the second step, where the linear 2-nitro-4,4'-dibromobiphenyl is converted into the fused 2,7-dibromocarbazole system. This transformation is achieved through a reductive cyclization mediated by triphenylphosphine (PPh3) in orthodichlorobenzene. Mechanistically, the triphenylphosphine acts as an oxygen scavenger, reducing the nitro group to a reactive nitrene or amine intermediate which subsequently undergoes intramolecular C-N bond formation with the adjacent aromatic ring. The use of orthodichlorobenzene as a high-boiling solvent is critical here, as it facilitates the reflux conditions necessary to drive this energetically demanding cyclization to completion. This metal-free cyclization method is particularly advantageous for electronic materials, as it avoids the introduction of transition metal contaminants that could act as quenching sites in the final OLED device. The result is a clean conversion that preserves the integrity of the bromine handles needed for subsequent functionalization.
Following the formation of the carbazole core, the final step involves the N-alkylation of the nitrogen atom with perfluoro butyl iodoethane. This reaction proceeds via a standard nucleophilic substitution mechanism where the deprotonated carbazole nitrogen attacks the alkyl iodide. The presence of triethylamine serves as a base to neutralize the generated hydroiodic acid, driving the equilibrium towards the product. The choice of toluene as a solvent and the temperature range of 80-120°C ensures sufficient solubility and kinetic energy for the reaction without degrading the sensitive perfluoro chain. This step is crucial for imparting the unique hydrophobic characteristics to the molecule. The fluorine atoms create a low-energy surface that repels water molecules, thereby enhancing the material's stability against humidity-induced degradation. This mechanistic understanding underscores the robustness of the process, making it suitable for the commercial scale-up of complex organic small molecules required for advanced display technologies.
How to Synthesize 2,7-Dibromo-N-Perfluorobutylethylcarbazole Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity and scalability, relying on three distinct unit operations that can be easily integrated into existing fine chemical facilities. The process begins with the controlled nitration of 4,4'-dibromobiphenyl, followed by the thermal cyclization and final alkylation. Each step has been optimized in the patent examples to balance reaction time and yield, utilizing standard workup procedures like extraction and recrystallization to ensure high purity. For technical teams looking to implement this route, the detailed standardized synthesis steps provided below outline the specific molar ratios, temperature profiles, and solvent systems required to replicate the successful outcomes described in the intellectual property.
- Nitration of 4,4'-dibromobiphenyl using a nitric-sulfuric acid mixture in methylene dichloride at 20-40°C to obtain 2-nitro-4,4'-dibromobiphenyl.
- Reductive cyclization of the nitro-intermediate with triphenylphosphine in orthodichlorobenzene under reflux conditions to form the 2,7-dibromocarbazole core.
- N-Alkylation of 2,7-dibromocarbazole with perfluoro butyl iodoethane using triethylamine in toluene at 80-120°C to yield the final fluorinated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of the synthesis route described in CN103232385A offers tangible benefits that extend beyond mere technical feasibility. The reliance on 4,4'-dibromobiphenyl as a starting material is a significant strategic advantage, as this feedstock is a commodity chemical with a stable global supply chain, mitigating the risk of raw material shortages. Furthermore, the elimination of precious metal catalysts in the cyclization step removes a major cost driver and simplifies the purification process, as there is no need for expensive metal scavenging resins or complex filtration protocols. This streamlining of the manufacturing process translates directly into improved margin structures and more competitive pricing for the final OLED material intermediate. Additionally, the robustness of the reaction conditions suggests a high tolerance for scale-up, reducing the likelihood of batch failures during technology transfer from lab to plant.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing triphenylphosphine for cyclization instead of palladium or copper catalysts, which are subject to volatile market pricing. By avoiding these expensive transition metals, the manufacturing protocol eliminates the need for costly downstream purification steps dedicated to removing metal residues to ppm levels. This reduction in processing complexity lowers both the capital expenditure on equipment and the operational expenditure on consumables. Moreover, the use of common solvents like dichloromethane, toluene, and orthodichlorobenzene ensures that solvent recovery and recycling can be managed efficiently, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: The synthetic route is built upon a foundation of widely available chemical building blocks, ensuring a resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. The starting material, 4,4'-dibromobiphenyl, is produced by multiple manufacturers globally, providing procurement managers with leverage in negotiations and backup options. The reaction conditions are relatively mild, operating at atmospheric pressure and moderate temperatures, which reduces the safety risks and regulatory burdens associated with high-pressure hydrogenation or cryogenic reactions. This operational safety profile facilitates smoother logistics and storage, allowing for consistent delivery schedules and reducing the lead time for high-purity OLED material intermediates.
- Scalability and Environmental Compliance: The three-step sequence is inherently scalable, with each reaction utilizing standard stirred-tank reactor configurations found in most multipurpose chemical plants. The absence of heavy metals simplifies waste stream management, as the effluent does not require specialized treatment for toxic metal removal, aligning with increasingly stringent environmental regulations. The workup procedures involve standard liquid-liquid extractions and crystallizations, which are well-understood unit operations that can be automated for consistent quality control. This ease of scale-up ensures that production volumes can be rapidly increased from kilogram to tonne scales to meet surging market demand for advanced display materials without compromising on the stringent purity specifications required by the electronics industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,7-dibromo-N-perfluorobutylethylcarbazole. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this material into their existing product portfolios and supply networks.
Q: What are the key advantages of 2,7-substituted carbazoles over 3,6-substituted analogues in OLED materials?
A: According to the background technology in patent CN103232385A, 2,7-substituted carbazoles possess a larger conjugated system compared to the more common 3,6-substituted variants. This structural difference results in superior thermostability and enhanced photoconductive properties, making them highly desirable for blue electroluminescent materials.
Q: How does the introduction of perfluoroalkyl chains affect the material properties?
A: The incorporation of perfluoro butyl ethyl groups leverages the strong hydrophobicity of fluorine atoms. This modification significantly improves the water resistance of the final organic small molecule material, addressing stability issues often encountered in optoelectronic device operation.
Q: Is the synthetic route scalable for industrial production?
A: Yes, the patent emphasizes that the preparation method is simple and utilizes accessible raw materials like 4,4'-dibromobiphenyl. The reaction conditions, such as temperatures between 20-120°C and standard solvents like toluene and dichloromethane, are compatible with large-scale chemical manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dibromo-N-Perfluorobutylethylcarbazole Supplier
As the demand for high-performance organic semiconductors continues to grow, partnering with a manufacturer that possesses deep technical expertise is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorinated organic synthesis, ensuring that every batch of 2,7-dibromo-N-perfluorobutylethylcarbazole meets stringent purity specifications. With our rigorous QC labs and commitment to process safety, we provide a secure and reliable supply chain for your critical OLED material needs, minimizing the risk of production delays.
We invite you to collaborate with us to optimize your sourcing strategy for advanced electronic chemicals. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timeline and secure a competitive edge in the rapidly evolving optoelectronics market.
