Advanced Cyano-Benzoxazine Intermediates: Enabling High-Tg Resins for Electronic and Aerospace Industries
Advanced Cyano-Benzoxazine Intermediates: Enabling High-Tg Resins for Electronic and Aerospace Industries
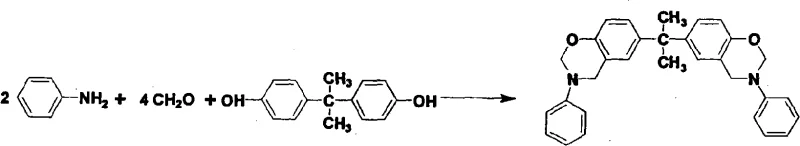
The landscape of high-performance thermosetting resins is undergoing a significant transformation driven by the demand for materials capable of withstanding extreme thermal environments without compromising structural integrity. Patent CN101265322A introduces a groundbreaking methodology for the preparation of cyano-containing benzoxazine intermediates and their subsequent resins, addressing critical limitations in traditional polymer chemistry. This innovation leverages a specific synthetic route utilizing cyano-containing monohydric phenols, primary amine compounds, and formaldehyde to generate intermediates that, upon curing and cross-linking, yield resins with exceptional properties. The resulting materials exhibit a glass transition temperature (Tg) exceeding 300°C, alongside superior heat resistance, flame retardancy, and carbon residue rates. For R&D directors and procurement specialists in the electronic and aerospace sectors, this technology represents a viable pathway to achieving reliable electronic chemical supplier status by offering materials that meet the rigorous demands of next-generation electronic packaging and structural components. The ability to operate effectively at temperatures above 250°C positions this chemistry as a cornerstone for advanced manufacturing where thermal stability is non-negotiable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of benzoxazine resins has relied heavily on specific precursor routes that, while effective to a degree, impose significant constraints on final material performance and economic feasibility. Early iterations, such as the 'dihydric phenol' route reported in the mid-1990s, succeeded in producing bicyclic intermediates that yielded polymers with a Tg of approximately 170°C. While this was a milestone that enabled applications in space vehicle interiors and sealing resins, it falls short for modern high-heat applications requiring stability well beyond 200°C. Furthermore, alternative strategies involving the introduction of polymerizable groups often utilized cyano-containing amines as raw materials. Although these cyano-amine routes could push Tg values to the 250-300°C range, they are plagued by substantial economic and supply chain drawbacks. Cyano-containing amines are notoriously expensive, possess limited structural variety, and are not conducive to large-scale industrial production due to sourcing complexities. These factors create a bottleneck for procurement managers seeking cost reduction in electronic chemical manufacturing, as the reliance on scarce and costly amine precursors inflates the bill of materials and introduces supply volatility.
The Novel Approach
The methodology outlined in CN101265322A circumvents these historical bottlenecks by shifting the source of the cyano functionality from the amine component to the phenolic component. By selecting cyano-containing phenols as the primary raw material, the process capitalizes on the fact that these compounds are easier to synthesize, relatively inexpensive, and available in a wide variety of commercial grades. This strategic substitution does not merely lower costs; it fundamentally enhances the scalability of the production process. The novel approach employs a 'diamine' route, which has been intellectually property-protected and refined to synthesize benzoxazine resins with higher heat resistance than the traditional 'dihydric phenol' routes. This shift allows for the production of high-performance resins that maintain the processing advantages of benzoxazines, such as near-zero volume shrinkage during curing, while achieving thermal properties comparable to polybismaleimides but at a significantly lower cost basis. For supply chain heads, this translates to a more robust and reliable polymer additive supplier network, reducing lead time for high-purity intermediates and ensuring continuity of supply for critical aerospace and electronic applications.
Mechanistic Insights into Cyano-Functionalized Mannich Condensation
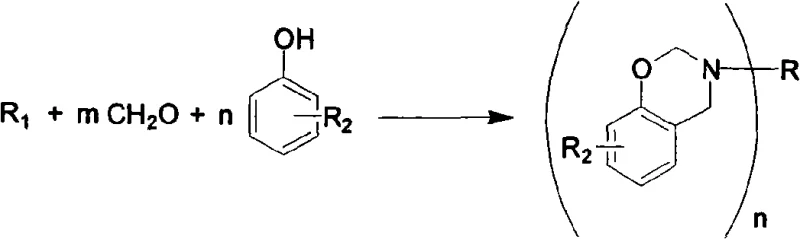
The core of this technological advancement lies in the precise control of the Mannich reaction and subsequent ring-closing polymerization. The synthesis begins with the formation of an N-dimethylol compound through the reaction of a primary amine and formaldehyde in a solvent system, typically dioxane or dimethylformamide. Crucially, the reaction temperature is maintained below 30°C, and the pH is adjusted to a range of 7-9 using triethylamine. This careful control ensures the stability of the N-dimethylol intermediate in the solution, preventing premature polymerization or side reactions that could compromise purity. Once this stable intermediate is established, the cyano-containing monohydric phenol is introduced. The mixture is then heated to reflux, where the N-dimethylol compound undergoes a condensation reaction with the phenolic hydroxyl group and the ortho position on the benzene ring. This step is critical for the formation of the six-membered oxazine ring, which is the defining structural feature of the benzoxazine intermediate. The presence of the cyano group on the phenolic ring significantly influences the electronic environment of the molecule, enhancing the cross-linking density upon subsequent curing.
Following the reflux period of 5 to 6 hours, the reaction mixture contains the desired cyano-containing benzoxazine intermediate. The workup process is designed to maximize yield and purity, involving precipitation with a fat-soluble solvent such as methanol or ethanol. The precipitated product is filtered and washed multiple times with a 1N NaHCO3 aqueous solution to remove acidic impurities and unreacted starting materials. Final purification is achieved through reduced pressure distillation to remove solvents, followed by vacuum drying. The resulting intermediate is a high-purity powder that can be cured in a staged process, typically ramping from 100°C to 260°C in increments. This curing profile facilitates the ring-opening polymerization, creating a dense, nitrogen-containing network structure similar to phenolic resins but with superior mechanical properties. The mechanism ensures that no small molecules are released during curing, which is vital for preventing voids in electronic encapsulation applications. The high char yield observed in these resins is a direct result of the cyano groups participating in the cross-linking network, providing exceptional thermal stability and flame retardancy without the need for additional halogenated additives.
How to Synthesize Cyano-Benzoxazine Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters defined in the patent to ensure reproducibility and high yield. The process is designed to be scalable, moving from laboratory glassware to industrial reactors with minimal modification to the core chemistry. The key to success lies in the sequential addition of reagents and the precise control of pH and temperature during the initial formation of the N-dimethylol species. Deviations in these early stages can lead to oligomerization or incomplete ring closure, affecting the thermal performance of the final resin. The detailed standardized synthesis steps provided below outline the exact stoichiometry and conditions required to achieve yields exceeding 80% with high purity specifications suitable for electronic grade applications.
- Prepare the reaction solvent system by mixing dioxane or dimethylformamide with paraformaldehyde and a primary amine compound, maintaining the temperature below 30°C.
- Adjust the pH to 7-9 using triethylamine to stabilize the N-dimethylol compound intermediate before adding the cyano-containing monohydric phenol.
- Heat the mixture to reflux for 5-6 hours to facilitate condensation and ring closure, followed by precipitation, washing, and vacuum drying to isolate the intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this cyano-phenol based synthesis route offers compelling strategic advantages that extend beyond mere technical performance. The primary benefit is the substantial cost savings derived from the raw material selection. By utilizing cyano-containing phenols instead of expensive cyano-containing amines, the overall cost of goods sold is drastically reduced. This cost efficiency does not come at the expense of quality; rather, it enhances the economic viability of producing high-Tg resins for mass-market applications in the automotive and electronics sectors. Furthermore, the availability of diverse cyano-phenol derivatives allows for greater flexibility in formulation, enabling manufacturers to tailor resin properties to specific customer requirements without incurring prohibitive costs. This flexibility is crucial for maintaining a competitive edge in the specialty chemical market where customization is often a key differentiator.
- Cost Reduction in Manufacturing: The elimination of expensive cyano-amine precursors in favor of readily available cyano-phenols results in a significantly lower raw material cost structure. This shift simplifies the supply chain by reducing dependency on niche chemical suppliers who often dictate pricing for specialized amines. Additionally, the mild reaction conditions and the use of common solvents like dioxane or DMF further contribute to operational cost efficiency. The process avoids the need for exotic catalysts or extreme pressure conditions, which reduces capital expenditure on specialized reactor equipment and lowers energy consumption during production. These factors combine to create a robust economic model that supports long-term pricing stability for downstream customers.
- Enhanced Supply Chain Reliability: Sourcing cyano-containing phenols is inherently more stable than sourcing specialized cyano-amines, as phenols are produced in larger volumes for various industrial applications. This abundance ensures a continuous supply of raw materials, mitigating the risk of production stoppages due to material shortages. For supply chain heads, this reliability translates to reduced lead time for high-purity intermediates and the ability to commit to tighter delivery schedules with confidence. The robustness of the synthesis route also means that production can be easily scaled up to meet surges in demand without compromising quality, ensuring that the supply chain remains resilient in the face of market fluctuations.
- Scalability and Environmental Compliance: The synthesis process is designed with scalability in mind, utilizing standard unit operations such as reflux, precipitation, and filtration that are well-understood in the chemical industry. This ease of scale-up facilitates the commercial scale-up of complex polymer additives from pilot plants to full-scale production facilities. Moreover, the process generates minimal hazardous waste compared to alternative high-performance resin syntheses. The absence of small molecule byproducts during the curing phase reduces volatile organic compound (VOC) emissions, aligning with increasingly stringent environmental regulations. This environmental compliance not only reduces the burden of waste disposal but also enhances the sustainability profile of the final product, a key consideration for modern procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this cyano-benzoxazine technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this material into their product lines and for procurement teams assessing the total cost of ownership.
Q: What are the thermal advantages of cyano-containing benzoxazine resins compared to traditional methyl-containing variants?
A: According to patent data, cyano-containing variants achieve a glass transition temperature (Tg) exceeding 300°C and a 5% thermal weight loss temperature around 383°C, significantly outperforming methyl-containing resins which typically show a Tg around 190°C.
Q: Why is the cyano-phenol route preferred over cyano-amine routes for industrial production?
A: Cyano-containing amines are often expensive and limited in variety, whereas cyano-containing phenols are easier to synthesize, more cost-effective, and available in a wider range of commercial specifications, facilitating large-scale manufacturing.
Q: What are the primary applications for these high-performance benzoxazine intermediates?
A: These intermediates are ideal for manufacturing structural materials, electrical insulation, electronic packaging, and flame-retardant materials that must withstand operating temperatures above 250°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyano-Benzoxazine Intermediate Supplier
The technical potential of cyano-containing benzoxazine intermediates is immense, offering a pathway to materials that can withstand the rigorous demands of modern high-temperature applications. NINGBO INNO PHARMCHEM stands ready to support your development and production needs as a trusted partner in this specialized field. As a CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory concept to industrial reality is seamless. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs that guarantee every batch meets the exacting standards required for electronic packaging and aerospace composites. We understand that consistency is key in high-performance polymer manufacturing, and our processes are designed to deliver that consistency reliably.
We invite you to engage with our technical procurement team to explore how this technology can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to our cyano-benzoxazine intermediates can impact your bottom line. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your unique application requirements. Whether you are developing next-generation circuit boards or high-heat structural components, our team is equipped to provide the technical support and material supply necessary to drive your innovation forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
