Advanced High-Char Benzoxazine Resins: Technical Breakthroughs for Electronic Insulation Manufacturing
Introduction to Next-Generation Electronic Insulation Materials
The relentless miniaturization and increasing power density of modern electronic devices have placed unprecedented demands on thermal management and fire safety materials. In this context, Patent CN114031616B represents a significant technological leap in the field of thermosetting resins, specifically addressing the critical need for high carbon residue and enhanced flame retardancy in electronic packaging. This patent discloses a novel benzoxazine resin architecture that strategically incorporates both ethyl acetate and triazole ring structures into the molecular backbone. Unlike conventional phenolic or epoxy systems which often suffer from brittleness or insufficient char yield during combustion, this new class of polybenzoxazines leverages molecular design to achieve superior performance metrics. By integrating nitrogen-rich heterocycles and ester functionalities, the material not only boosts the residual carbon rate—a key indicator of thermal stability and fire resistance—but also effectively lowers the curing temperature required for processing. For R&D directors and procurement specialists in the electronic materials sector, this innovation offers a compelling pathway to developing next-generation encapsulants and laminates that meet rigorous safety standards without compromising manufacturability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional thermosetting resins, such as standard phenolics and early-generation benzoxazines, have long served as the backbone of the electrical insulation industry. However, they face inherent limitations when subjected to the extreme thermal environments of modern high-power electronics. A primary deficiency lies in their relatively low nitrogen content, which directly correlates to poor char formation during thermal degradation. When exposed to fire, these conventional polymers tend to decompose rapidly, releasing volatile combustible gases and leaving behind a fragile, porous carbonaceous layer that fails to protect the underlying substrate. Furthermore, achieving high thermal stability in legacy systems often requires the addition of halogenated flame retardants, which are increasingly scrutinized due to environmental regulations and toxicity concerns. The synthesis of modified resins to overcome these issues frequently involves complex multi-step reactions, harsh acidic conditions, or the use of expensive precursors that drive up manufacturing costs and complicate supply chain logistics. These factors collectively hinder the ability of manufacturers to produce cost-effective, high-performance materials that can withstand the rigorous demands of advanced semiconductor packaging and PCB lamination.
The Novel Approach
The methodology outlined in Patent CN114031616B circumvents these historical bottlenecks through a clever dual-modification strategy. By introducing a triazole ring via click chemistry, the resin backbone is enriched with nitrogen atoms that act as intrinsic flame retardants, promoting the formation of a robust, intumescent char layer upon heating. Simultaneously, the incorporation of ethyl acetate groups provides a secondary mechanism for fire suppression; under high-temperature conditions, these ester moieties decompose to release carbon dioxide, a non-flammable gas that dilutes oxygen concentration near the burning surface and further inhibits combustion. This synergistic effect allows the material to achieve a remarkably high carbon residue rate while maintaining excellent mechanical integrity. Moreover, the synthetic route is designed for efficiency, utilizing a straightforward condensation reaction followed by a catalytic cycloaddition. This streamlined approach not only reduces the number of processing steps but also operates under milder conditions compared to traditional modification techniques, thereby preserving the structural integrity of the monomers and ensuring a high-purity final product suitable for sensitive electronic applications.
Mechanistic Insights into Cu-Catalyzed Azide-Alkyne Cycloaddition and Thermal Curing
The core of this technological advancement lies in the precise construction of the monomer architecture using Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC), widely recognized as a premier example of click chemistry. The synthesis begins with the formation of an ethynyl-functionalized benzoxazine intermediate. This is achieved through a Mannich-like condensation reaction where Bisphenol A serves as the phenolic source, reacting with paraformaldehyde and 3-ethynylaniline. The reaction proceeds efficiently under a nitrogen atmosphere at moderate temperatures, typically around 80°C, yielding the alkyne-bearing precursor with high fidelity. This intermediate retains the characteristic oxazine ring, which is crucial for the subsequent thermal polymerization, while presenting a reactive terminal alkyne group ready for functionalization. The robustness of this initial step ensures that the bulk of the molecular framework is established using commodity chemicals, laying a cost-effective foundation for the entire synthesis.
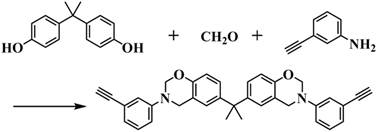
Following the preparation of the alkyne intermediate, the critical functionalization step occurs. The ethynyl benzoxazine is reacted with ethyl azidoacetate in the presence of a catalytic amount of Copper(I) Iodide (CuI). This reaction is highly regioselective, forming a 1,2,3-triazole linkage that bridges the benzoxazine core and the ethyl acetate side chain. The use of CuI is particularly advantageous for industrial applications; it is an inexpensive, readily available catalyst that drives the reaction to completion at room temperature, eliminating the need for energy-intensive heating protocols often associated with thermal Huisgen cycloadditions. The resulting triazole ring is chemically stable yet thermally responsive, contributing significantly to the nitrogen content of the final polymer. This modular approach allows for precise tuning of the resin's properties by simply varying the azide component, offering a versatile platform for material scientists to tailor performance characteristics for specific electronic end-uses.
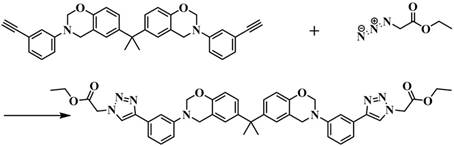
The final transformation involves the thermal curing of the functionalized monomer into a cross-linked polybenzoxazine network. Upon heating, the oxazine rings undergo a ring-opening polymerization mechanism. This process is autocatalytic and does not require additional curing agents or initiators, which simplifies the formulation and reduces the risk of impurity introduction. As the temperature rises, the oxazine rings open to form a Mannich bridge structure (-CH2-NR-CH2-) linking the phenolic units, creating a dense, three-dimensional network. The presence of the triazole and ester groups influences this curing behavior; the nitrogen atoms in the triazole ring can participate in hydrogen bonding, potentially lowering the activation energy for ring opening and thus reducing the overall curing temperature. This lower processing temperature is a critical advantage for electronic packaging, as it minimizes thermal stress on sensitive components during the encapsulation process. The resulting polymer exhibits a highly cross-linked structure with exceptional thermal stability, as evidenced by its high char yield and resistance to thermal degradation.
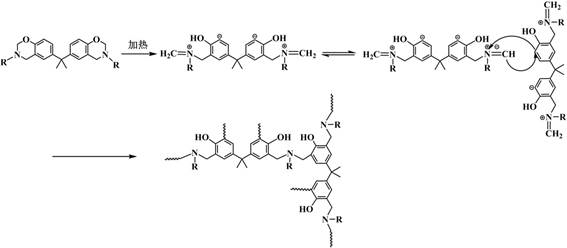
How to Synthesize High Carbon Residue Benzoxazine Efficiently
The synthesis protocol described in the patent offers a robust and scalable pathway for producing these advanced monomers. The process is divided into distinct stages that ensure high purity and yield at each step, starting from the condensation of raw materials to the final thermal curing. The initial condensation requires careful control of stoichiometry and temperature to maximize the formation of the ethynyl intermediate while minimizing side reactions. Subsequent purification steps, such as solvent separation and vacuum drying, are critical to removing unreacted amines or phenols that could act as plasticizers or degrade the thermal properties of the final resin. The click chemistry step is notably forgiving, proceeding efficiently at ambient temperatures, which reduces energy consumption and equipment requirements. For detailed operational parameters, including specific solvent volumes, molar ratios, and gradient curing profiles, please refer to the standardized synthesis guide below.
- Condense Bisphenol A, paraformaldehyde, and 3-ethynylaniline in an organic solvent at 80°C under nitrogen to form the ethynyl benzoxazine intermediate.
- React the ethynyl benzoxazine intermediate with ethyl azidoacetate using 5 mol% CuI catalyst at room temperature for 24 hours to introduce the triazole and ester groups.
- Purify the resulting monomer via solvent separation and vacuum drying, followed by gradient thermal curing to obtain the final polybenzoxazine network.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the technology disclosed in Patent CN114031616B presents a highly attractive value proposition for procurement managers and supply chain leaders in the specialty chemicals sector. The primary driver of cost efficiency is the reliance on commodity feedstocks. Bisphenol A, paraformaldehyde, and aniline derivatives are produced on a massive global scale, ensuring a stable and competitive supply base that is less susceptible to the volatility often seen with exotic fine chemical intermediates. Furthermore, the synthetic route avoids the use of precious metal catalysts such as palladium or platinum, which are not only expensive but also subject to significant price fluctuations and geopolitical supply risks. Instead, the process utilizes copper iodide, a base metal salt that is abundant and inexpensive. This substitution drastically reduces the raw material cost per kilogram of the final product, enabling manufacturers to offer high-performance electronic materials at a price point that competes effectively with traditional epoxy and phenolic systems.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the ability to run the key functionalization step at room temperature significantly lower the operational expenditure (OPEX) associated with production. The absence of high-temperature requirements for the click chemistry step reduces energy consumption, while the high yields reported in the patent minimize waste generation and the need for complex recycling streams. Additionally, the solvent systems employed, such as DMF or toluene, are standard industrial solvents with well-established recovery and recycling infrastructures, further enhancing the economic viability of the process. By streamlining the synthesis into fewer steps with higher atom economy, manufacturers can achieve substantial cost savings that can be passed down to customers or reinvested in R&D.
- Enhanced Supply Chain Reliability: The reliance on widely available chemical building blocks ensures a resilient supply chain that is less prone to disruptions. Unlike specialized monomers that may be sourced from a single supplier, the precursors for this benzoxazine resin are available from multiple global vendors, providing procurement teams with leverage and flexibility. The simplicity of the reaction conditions also means that the technology can be easily transferred to different manufacturing sites without requiring specialized high-pressure or cryogenic equipment. This flexibility allows for decentralized production strategies, reducing lead times and transportation costs for regional markets. The robustness of the synthesis ensures consistent quality batch-to-batch, which is critical for maintaining long-term contracts with major electronics manufacturers who demand strict adherence to specifications.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to pilot plant and full commercial production. The use of standard organic solvents and ambient pressure reactions simplifies the engineering requirements for scale-up, reducing capital expenditure (CAPEX) for new production lines. From an environmental standpoint, the high efficiency of the click reaction minimizes the generation of hazardous byproducts, aligning with green chemistry principles. The final resin's enhanced flame retardancy reduces the need for additive-type flame retardants, many of which are facing regulatory restrictions. This intrinsic safety feature simplifies the regulatory compliance landscape for downstream users, facilitating faster time-to-market for electronic devices that must meet stringent fire safety standards like UL94.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-performance benzoxazine technology. These insights are derived directly from the patent specifications and are intended to clarify the practical implications for industrial adoption. Understanding the interplay between molecular structure and macroscopic properties is essential for evaluating the suitability of this material for specific electronic applications. The answers provided reflect the current state of the art as described in the intellectual property documentation.
Q: How does the triazole structure improve the flame retardancy of benzoxazine resins?
A: The triazole ring introduces a high density of nitrogen atoms into the polymer backbone. Upon thermal decomposition, these nitrogen species promote char formation and release non-flammable gases, significantly enhancing the flame retardant properties compared to traditional low-nitrogen phenolic resins.
Q: What are the key advantages of using CuI catalysis in this synthesis?
A: Copper(I) iodide facilitates a highly efficient Azide-Alkyne Cycloaddition (Click Chemistry) at room temperature. This eliminates the need for harsh conditions or expensive noble metal catalysts, simplifying the purification process and reducing overall production costs.
Q: Can this resin be scaled for industrial electronic packaging applications?
A: Yes, the synthesis utilizes readily available commodity chemicals like Bisphenol A and standard organic solvents. The process operates under mild conditions with high yields, making it highly suitable for commercial scale-up in the electronic materials sector.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazine Resin Supplier
As the global demand for high-performance electronic materials continues to surge, partnering with an experienced chemical manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in polymer synthesis and process optimization to deliver cutting-edge solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We understand that in the electronics sector, purity is paramount; therefore, our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of resin we produce. Whether you require custom modifications to the benzoxazine backbone or large-scale supply of standard grades, our infrastructure is designed to support your growth.
We invite you to collaborate with us to explore how this advanced triazole-functionalized benzoxazine technology can enhance your product portfolio. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific manufacturing setup, demonstrating exactly how switching to this novel resin can optimize your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By working together, we can accelerate the development of safer, more durable, and cost-effective electronic packaging solutions that meet the evolving needs of the global market.
