Advanced Diazabenzofluoranthene Compounds for High-Performance OLED Displays
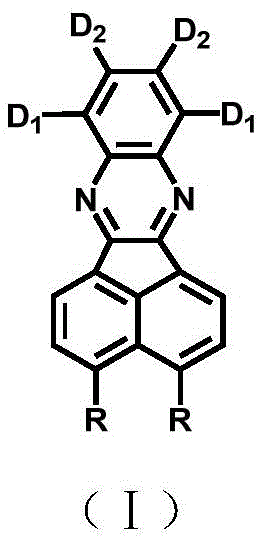
The rapid evolution of organic light-emitting diode (OLED) technology has created an urgent demand for high-performance emissive materials capable of delivering full-color displays with superior efficiency. Patent CN113683601B introduces a groundbreaking class of diazabenzofluoranthene compounds designed to overcome the spectral limitations of existing orange and red emitters. By utilizing a pyrazine moiety as a robust electron-accepting core and strategically integrating multiple electron-donating groups, this invention achieves a tunable emission spectrum ranging from 531nm to 713nm. This technological leap addresses the critical industry bottleneck of narrow emission bands, offering a versatile platform for next-generation solid-state lighting and smart material applications. The structural versatility allows for precise modulation of charge transfer states, resulting in fluorescence quantum yields as high as 96%, which signifies a major advancement in material efficiency.
For procurement specialists and R&D directors seeking a reliable OLED material supplier, this patent represents a significant opportunity for cost reduction in electronic chemical manufacturing. The ability to fine-tune emission colors without compromising quantum efficiency means fewer material variants are needed to cover the entire visible spectrum, simplifying inventory management and device fabrication processes. Furthermore, the synthetic route described relies on established organic transformations that are amenable to large-scale production, ensuring that the transition from laboratory discovery to commercial availability can be executed with minimal friction. This combination of high performance and manufacturability positions these diazabenzofluoranthene derivatives as a cornerstone for future display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic发光 materials has been heavily skewed towards blue and green emitters, leaving a significant gap in the performance of orange and red materials required for high-quality full-color displays. Existing solutions, such as those based on simple pyrazine acceptors, often suffer from a narrow spectral range, typically confined between 580nm and 620nm, which restricts the color gamut achievable in OLED devices. Moreover, conventional long-wave emission small molecules frequently rely on strong electron-donating groups to achieve the desired redshift, but this often leads to planar molecular structures that promote tight π-π stacking. This tight packing results in severe concentration quenching, where the excited state energy is dissipated non-radiatively, drastically lowering the device efficiency and lifespan. Additionally, the rigid planar nature of many traditional emitters limits the ability to finely tune the emission color across a broad spectrum, forcing manufacturers to blend multiple materials to achieve the desired hue, which complicates the device architecture.
The Novel Approach
The novel approach detailed in this patent fundamentally reengineers the molecular architecture by introducing a diazabenzofluoranthene core that inherently supports a D-A or D-π-A charge transfer structure. Unlike prior art, this method allows for the introduction of multiple different electron donors (D1 and D2) at specific positions, creating multiple charge transfer channels that enhance intramolecular charge transfer efficiency. A key innovation is the strategic selection of donor groups that induce specific torsion angles relative to the central benzene nucleus. These torsion angles prevent the molecules from packing too densely, effectively suppressing the π-π stacking interactions that cause fluorescence quenching in conventional materials. This structural design not only enables a wide spectral tuning range from green to near-infrared but also maintains high fluorescence quantum yields, solving the long-standing trade-off between emission wavelength and efficiency in organic semiconductors.
Mechanistic Insights into Pd-Catalyzed Coupling and Condensation
The synthesis of these advanced materials relies on a sophisticated sequence of reactions that build the complex polycyclic aromatic framework with high precision. The process begins with the formation of the core diazabenzofluoranthene skeleton through a condensation reaction between a fluorinated phenylenediamine derivative and an acenaphthenequinone compound. This step is critical as it establishes the rigid, planar acceptor unit that defines the electronic properties of the final emitter. Following the core formation, the methodology employs palladium-catalyzed cross-coupling reactions to attach the electron-donating groups. This modular approach allows for the systematic variation of D1 and D2 groups, enabling fine-tuning of the HOMO-LUMO energy gaps without altering the core synthesis. The use of mild reaction conditions and standard catalysts ensures that the process is robust and reproducible, which is essential for maintaining batch-to-batch consistency in commercial production.
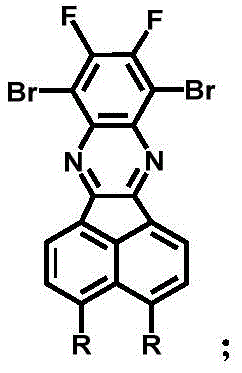
Impurity control is managed through the inherent selectivity of the condensation and coupling steps, combined with rigorous purification protocols such as silica gel column chromatography. The use of specific molar ratios, such as a 1:1 ratio between the diamine and quinone precursors, minimizes the formation of side products and oligomers. Furthermore, the inert atmosphere conditions employed during the heating steps prevent oxidative degradation of the sensitive intermediates, ensuring high purity of the final product. The mechanistic understanding of how the torsion angles influence molecular packing provides a rational basis for impurity profiling, as deviations in the donor group attachment could lead to planar impurities that might act as quenchers. This deep mechanistic insight allows for the implementation of targeted analytical methods to detect and remove such trace impurities, guaranteeing the high performance required for premium OLED applications.
How to Synthesize Diazabenzofluoranthene Efficiently
The preparation of these high-performance emitters involves a streamlined five-step sequence that balances yield and purity. The process initiates with the nitration and subsequent reduction of a dibromodifluorobenzene precursor to generate the key diamine intermediate. This is followed by a condensation reaction with acenaphthenequinone to form the central heterocyclic core. The final stages involve sequential palladium-catalyzed coupling reactions to install the electron-donating carbazole or triphenylamine units. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Nitration of 1,4-dibromo-2,3-difluorobenzene followed by reduction to form the diamine precursor.
- Condensation of the diamine with acenaphthenequinone derivatives to construct the diazabenzofluoranthene core.
- Sequential palladium-catalyzed coupling with electron donor groups (D1 and D2) to tune emission properties.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this diazabenzofluoranthene technology offers substantial advantages in terms of raw material security and process scalability. The synthesis pathway utilizes commodity chemicals such as brominated benzenes, nitric acid, and common heterocyclic amines, which are widely available from global chemical suppliers. This reduces the risk of supply chain disruptions associated with exotic or proprietary starting materials. Furthermore, the reaction conditions, while requiring inert atmospheres for the coupling steps, operate at temperatures and pressures that are compatible with standard stainless steel reactors found in most fine chemical manufacturing facilities. This compatibility eliminates the need for specialized equipment investments, facilitating a smoother and faster transition from pilot scale to full commercial production.
- Cost Reduction in Manufacturing: The synthetic route is designed to maximize atom economy and minimize the use of expensive reagents. By avoiding the need for complex multi-step functionalizations of the core ring system prior to coupling, the overall number of unit operations is reduced. The high yields reported in the patent examples, particularly in the condensation and coupling steps, directly translate to lower cost of goods sold (COGS). Additionally, the ability to achieve high quantum yields means that less material is required per device to achieve the same brightness, further driving down the effective cost per unit for the end-user. The elimination of heavy metal catalysts in the final purification steps also reduces waste disposal costs associated with hazardous metal residues.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations such as nitration, reduction, and Suzuki-Miyaura coupling ensures high process reliability. These reactions are less sensitive to minor fluctuations in temperature or reagent quality compared to more exotic catalytic cycles, leading to consistent batch outcomes. The modular nature of the synthesis allows for the decoupling of intermediate production; for instance, the core diazabenzofluoranthene scaffold can be manufactured in bulk and stored, while the final coupling with specific donor groups can be performed on demand to meet specific customer color requirements. This flexibility significantly reduces lead times for custom orders and enhances the responsiveness of the supply chain to market demands.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, with reaction parameters that can be easily translated from gram-scale laboratory synthesis to kilogram and ton-scale production. The use of common organic solvents like toluene, ethanol, and acetic acid simplifies solvent recovery and recycling systems, aligning with modern environmental sustainability goals. The absence of highly toxic reagents or extreme reaction conditions minimizes the environmental footprint of the manufacturing process. Moreover, the high purity of the final product reduces the burden on downstream device manufacturers, who often face challenges with material purification, thereby creating a more efficient and environmentally friendly value chain from synthesis to final device assembly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of diazabenzofluoranthene compounds in OLED manufacturing. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for adoption.
Q: What is the emission range of these diazabenzofluoranthene compounds?
A: The compounds exhibit a wide spectral range from 531nm to 713nm, covering green to near-infrared light, with fluorescence quantum yields reaching up to 96%.
Q: How does the molecular structure prevent fluorescence quenching?
A: The introduction of specific electron donor groups creates torsion angles with the central benzene nucleus, reducing molecular packing density and suppressing π-π stacking interactions that cause quenching.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily available starting materials such as 1,4-dibromo-2,3-difluorobenzene, acenaphthenequinone, and common boronic acid derivatives, ensuring supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazabenzofluoranthene Supplier
As the demand for high-efficiency red and orange OLED materials continues to surge, partnering with an experienced CDMO is crucial for securing a stable supply of high-purity intermediates. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our stringent purity specifications and rigorous QC labs guarantee that every batch of diazabenzofluoranthene compound meets the exacting standards required for next-generation display panels. We understand the critical nature of material consistency in OLED fabrication and have optimized our processes to deliver products with minimal batch-to-batch variation.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our capabilities can support your product roadmap. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can lower your overall material costs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and bring superior display technologies to market faster.
