Advanced Synthesis of SeTTP and STTP for High-Fidelity Nucleic Acid Detection and Commercial Scale-Up
Advanced Synthesis of SeTTP and STTP for High-Fidelity Nucleic Acid Detection and Commercial Scale-Up
In the rapidly evolving landscape of molecular diagnostics and genomic research, the fidelity of nucleic acid amplification is paramount for accurate disease identification and environmental monitoring. Patent CN113004358A introduces a groundbreaking approach to synthesizing selenium or thiothymidine-5'-triphosphate, specifically targeting the persistent challenge of base-pairing mismatches in DNA polymerization. This technology leverages a Selenium or sulfur Atom specific Modification (SAM) strategy to fundamentally alter the hydrogen bonding dynamics of thymine bases. By substituting the 2-oxygen atom with selenium or sulfur, the resulting nucleotides, SeTTP and STTP, exhibit superior specificity, effectively inhibiting the undesirable T/G wobble pairing that often plagues conventional PCR assays. For global supply chain leaders and R&D directors, this represents a critical advancement in reliable nucleic acid intermediate supplier capabilities, offering a pathway to drastically reduce false-negative results in pathogen detection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional nucleic acid detection methods, such as standard RT-qPCR, rely heavily on the precise base pairing of natural nucleotides. However, a significant pain point in the industry is the inherent tendency of thymine to form wobble pairs with guanine (T/G mismatch) due to the hydrogen bonding capability of the 2-oxygen atom. This abnormal pairing diversifies RNA structure and can lead to mutations or, more critically in a diagnostic context, reduced specificity during DNA synthesis. When detecting pathogens with low copy numbers, such as in early-stage viral infections, these mismatches generate background noise that can overwhelm the weak signal of the target nucleic acid. Consequently, this leads to false-negative results, which is a catastrophic failure mode in epidemic control and clinical diagnosis. The reliance on standard thymidine triphosphates limits the sensitivity of commercial kits, creating a bottleneck for high-stakes applications where every copy counts.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these limitations through the strategic incorporation of selenium or sulfur atoms into the thymidine base. This SAM strategy creates a steric and electronic environment that disfavors mismatched pairing while maintaining compatibility with DNA polymerases like Klenow and Taq. The synthesis route is designed for efficiency, utilizing a one-pot method to convert the protected nucleoside intermediates directly into the final triphosphate products, SeTTP and STTP. This approach not only enhances the accuracy of base pairing by approximately 10,000 times compared to standard TTP but also simplifies the manufacturing workflow. For procurement managers focused on cost reduction in molecular diagnostic manufacturing, this dual benefit of enhanced performance and streamlined synthesis offers a compelling value proposition for next-generation sequencing and diagnostic kit formulation.
Mechanistic Insights into Selenium-Induced Specificity Enhancement
The core of this technological breakthrough lies in the fundamental atomic properties of selenium compared to oxygen. As illustrated in the atomic radius data, selenium possesses a significantly larger atomic radius than oxygen, which fundamentally alters its interaction with neighboring bases. 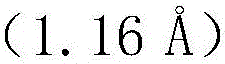 . Due to this larger size and the poor hydrogen bonding capability of selenium, the 2-seleno-thymine base is sterically and electronically prevented from forming stable hydrogen bonds with the guanine base. While the 2-oxygen in natural thymine participates in wobble pairing with guanine, the 2-selenium atom does not, thereby eliminating the T/G mismatch pathway. Crucially, this modification does not interfere with the correct Watson-Crick base pairing with adenine, ensuring that the polymerase can still efficiently incorporate the modified nucleotide during legitimate DNA synthesis. This selective inhibition of mismatching is the key mechanism driving the improved sensitivity and accuracy of the SAM strategy.
. Due to this larger size and the poor hydrogen bonding capability of selenium, the 2-seleno-thymine base is sterically and electronically prevented from forming stable hydrogen bonds with the guanine base. While the 2-oxygen in natural thymine participates in wobble pairing with guanine, the 2-selenium atom does not, thereby eliminating the T/G mismatch pathway. Crucially, this modification does not interfere with the correct Watson-Crick base pairing with adenine, ensuring that the polymerase can still efficiently incorporate the modified nucleotide during legitimate DNA synthesis. This selective inhibition of mismatching is the key mechanism driving the improved sensitivity and accuracy of the SAM strategy.
Furthermore, the synthesis mechanism ensures high purity by leveraging specific deprotection and phosphorylation conditions that minimize side reactions. The process involves a careful deprotection step using trichloroacetic acid to remove the dimethoxytrityl (DMTr) group, followed by a highly controlled one-pot phosphorylation. This sequence is critical for maintaining the integrity of the sensitive seleno-modified base, which could otherwise be susceptible to oxidation or degradation under harsher conditions. The use of reagents like 2-chloro-4H-1,3,2-benzodioxaphosphol-4-one allows for the efficient formation of the triphosphate chain without requiring multiple isolation steps that could lead to product loss. For R&D teams, understanding this mechanistic robustness is essential for scaling the process, as it indicates a high tolerance for variation and a clear path to achieving the stringent purity specifications required for enzymatic applications.
How to Synthesize Selenium-Thymidine-5'-Triphosphate Efficiently
The synthesis of SeTTP described in the patent follows a logical, step-wise progression that balances chemical yield with operational simplicity. The process begins with the activation of the 2-thio functional group via alkylation, followed by the critical selenium substitution step using NaSeH. Subsequent deprotection reveals the free nucleoside, which is then subjected to the one-pot triphosphorylation. This route is designed to be scalable, utilizing common organic solvents like dichloromethane and DMF, and reagents that are readily available in fine chemical supply chains. The detailed standardized synthesis steps, including specific molar ratios, temperature controls (such as maintaining 0°C during NaSeH preparation), and purification protocols via HPLC, provide a robust framework for manufacturing. For technical teams looking to implement this, the following guide outlines the critical operational parameters derived directly from the patent examples.
- Alkylate 5'-DMTr-2-thiothymidine with methyl iodide to activate the 2-thio functional group, yielding Compound 2.
- React Compound 2 with freshly prepared NaSeH solution to perform selenium substitution, obtaining Compound 3.
- Deprotect Compound 3 using trichloroacetic acid to remove the DMTr group, yielding the nucleoside Compound 4Se.
- Perform a one-pot phosphorylation on Compound 4Se using tributyl pyrophosphate amine and 2-chloro-4H-1,3,2-benzodioxaphosphol-4-one to generate SeTTP.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this SAM-based synthesis route offers substantial advantages for supply chain stability and cost management. The elimination of complex transition metal catalysts, which are often required in alternative modification strategies, removes the need for expensive and time-consuming heavy metal clearance steps. This simplification directly translates to reduced processing time and lower consumption of specialized scavenging resins. Furthermore, the one-pot nature of the final phosphorylation step significantly reduces the number of unit operations, minimizing solvent usage and waste generation. For procurement managers, this means a more predictable cost structure and a reduced environmental footprint, aligning with modern green chemistry initiatives. The ability to produce high-purity intermediates with fewer purification cycles enhances the overall throughput of the manufacturing facility, allowing for better responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The synthetic route described avoids the use of precious metal catalysts and relies on cost-effective reagents such as selenium powder, sodium borohydride, and methyl iodide. By streamlining the synthesis into fewer steps, particularly the one-pot conversion of the nucleoside to the triphosphate, the process significantly lowers the operational expenditure associated with labor, energy, and solvent recovery. The high yields reported in the intermediate steps (e.g., 95% yield in deprotection) further contribute to material efficiency, ensuring that raw material costs are optimized. This qualitative improvement in process efficiency allows for competitive pricing strategies without compromising on the high purity required for diagnostic applications.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including protected nucleosides and phosphorus reagents, are commercially available from established fine chemical suppliers, reducing the risk of supply bottlenecks. The robustness of the reaction conditions, which tolerate standard laboratory temperatures and pressures, facilitates easy technology transfer between different manufacturing sites. This flexibility ensures continuity of supply, a critical factor for global diagnostic companies that cannot afford interruptions in their reagent supply chains. Additionally, the stability of the intermediates allows for strategic stockpiling, further buffering against market volatility and ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to kilogram and ton-scale commercial production. The use of standard organic solvents like dichloromethane and ethanol simplifies waste management and solvent recycling, aiding in compliance with strict environmental regulations. The reduction in the number of purification steps also decreases the volume of hazardous waste generated per unit of product. For supply chain heads, this scalability means that the technology can support the massive volume requirements of global pandemic response efforts or routine high-throughput screening, ensuring that the supply of these critical diagnostic intermediates remains uninterrupted and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium-modified nucleotide technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these nuances is vital for stakeholders evaluating the integration of SeTTP or STTP into their existing diagnostic platforms or research workflows. The answers provided reflect the specific advantages of the SAM strategy over conventional nucleotide usage.
Q: How does Selenium modification improve PCR specificity compared to standard Thymidine?
A: The substitution of the 2-oxygen atom with selenium increases the atomic radius and reduces hydrogen bonding capability. This prevents the formation of T/G wobble pairs, significantly inhibiting mismatches during DNA polymerization and improving detection accuracy.
Q: What is the primary advantage of the one-pot synthesis method described in the patent?
A: The one-pot synthesis method for converting the nucleoside to the triphosphate minimizes intermediate isolation steps, reduces solvent consumption, and streamlines the purification process, leading to improved operational efficiency and potential cost reductions.
Q: Can this technology detect low-copy viral RNA effectively?
A: Yes, by suppressing T/G mismatches, the SAM strategy enhances the signal-to-noise ratio in nucleic acid amplification. This allows for the accurate detection of pathogens even at extremely low copy numbers, addressing the issue of false negatives in traditional RT-qPCR kits.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SeTTP Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the SAM strategy in elevating the standards of molecular diagnostics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like selenium-substituted nucleotide synthesis are executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of SeTTP or STTP delivered meets the exacting standards required for enzymatic DNA synthesis and high-sensitivity PCR applications. We are committed to bridging the gap between innovative patent chemistry and reliable industrial supply.
We invite global partners to collaborate with us to optimize their supply chains and enhance their product performance. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure a stable source of high-performance nucleic acid intermediates that drive accuracy in detection and reliability in results.
