Advanced Fluorine-Tagged Oligosaccharide Synthesis for High-Purity Pharmaceutical Intermediates
The landscape of oligosaccharide synthesis has long been challenged by the arduous task of isolating high-purity structures from natural sources or complex reaction mixtures. Patent CN112940058A introduces a transformative approach that merges the precision of enzymatic synthesis with the separation power of fluorine labeling. This technology addresses the critical bottlenecks in producing structurally defined oligosaccharides, which are essential for understanding biological functions and developing new therapeutics. By utilizing a novel fluorine carrier, specifically a lactose-based acceptor tagged with a fluorous chain, the invention enables rapid and efficient assembly of various oligosaccharide chains. The core innovation lies in the structural formula G-R, where G represents the carbohydrate moiety and R is a specialized fluorine-containing group designed for optimal solubility and extraction properties. This strategic integration allows for the seamless transition from laboratory-scale discovery to commercial-scale production, offering a reliable pathway for manufacturing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of oligosaccharides has relied heavily on techniques such as Fluorine Liquid-Liquid Extraction (FLLE) or traditional Solid Phase Extraction (SPE), both of which present significant drawbacks for industrial application. FLLE often requires the use of expensive, high-fluorinated solvents to achieve effective phase separation, driving up operational costs and complicating waste management protocols. Furthermore, heavy fluoro labels used in these processes frequently exhibit poor solubility in common organic solvents, limiting their utility in diverse synthetic routes. On the other hand, existing light fluorine labels, while more soluble, often suffer from instability under acidic or basic conditions and possess low loading capacities. Traditional solid-phase synthesis, while popular for peptides, struggles in carbohydrate chemistry due to heterogeneous reaction systems that hinder glycosylation efficiency and make real-time monitoring nearly impossible. These cumulative inefficiencies result in lower overall yields and extended production timelines, creating a substantial barrier for the reliable supply of complex sugar structures.
The Novel Approach
The methodology outlined in the patent data overcomes these historical hurdles by introducing a robust fluorine label that functions effectively in a homogeneous reaction system. As illustrated in the structural representation of the core lactose acceptor, the new fluorine carrier is engineered for stability and ease of manipulation. 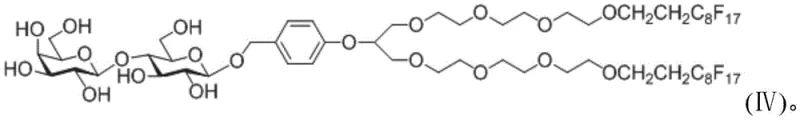 This specific configuration, often referred to as Formula IV in the technical documentation, combines a lactose base with a double fluorine chain receptor, ensuring compatibility with standard organic solvents while retaining strong affinity for fluorinated stationary phases. The homogeneous nature of the reaction environment allows chemists to monitor progress using conventional analytical techniques, thereby ensuring consistent quality control throughout the synthesis. Moreover, the design prioritizes the facile removal and regeneration of the fluorine tag, a critical feature for cost reduction in pharmaceutical intermediate manufacturing. By shifting away from heterogeneous solid supports and expensive solvent systems, this novel approach streamlines the workflow, significantly simplifying the purification process and enhancing the overall throughput of oligosaccharide production.
This specific configuration, often referred to as Formula IV in the technical documentation, combines a lactose base with a double fluorine chain receptor, ensuring compatibility with standard organic solvents while retaining strong affinity for fluorinated stationary phases. The homogeneous nature of the reaction environment allows chemists to monitor progress using conventional analytical techniques, thereby ensuring consistent quality control throughout the synthesis. Moreover, the design prioritizes the facile removal and regeneration of the fluorine tag, a critical feature for cost reduction in pharmaceutical intermediate manufacturing. By shifting away from heterogeneous solid supports and expensive solvent systems, this novel approach streamlines the workflow, significantly simplifying the purification process and enhancing the overall throughput of oligosaccharide production.
Mechanistic Insights into Enzymatic Modular Assembly
The heart of this synthesis strategy lies in its sophisticated use of enzymatic modules that mimic biological pathways to construct complex glycosidic bonds with high stereochemical specificity. The process employs a series of glycosyltransferases, sugar nucleoside generating enzymes, and glucokinases, all sourced from prokaryotic organisms to ensure high expression levels and catalytic efficiency. For instance, the assembly of a trisaccharide involves the coordinated action of enzymatic Module A, which includes a fusion enzyme of N-acetamido hexokinase and a nucleoside sugar producing enzyme, alongside a specific beta-1-3-N-acetamido glucosyltransferase. This modular setup allows for the precise addition of sugar units, such as N-acetylglucosamine, to the fluorine-labeled lactose acceptor without the need for extensive protecting group chemistry typically required in chemical synthesis. The enzymes facilitate the conversion of simple monosaccharides into activated nucleoside donors in situ, driving the reaction forward with exceptional regioselectivity. 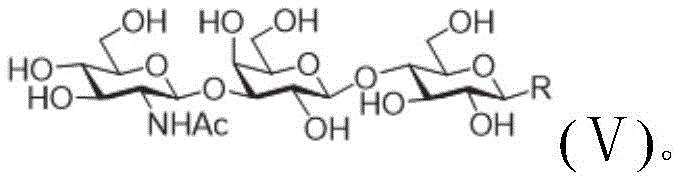 This mechanistic precision ensures that the resulting oligosaccharide chains, such as the trisaccharide shown in Formula V, possess the exact linkage patterns required for biological activity, eliminating the formation of unwanted isomers that complicate downstream purification.
This mechanistic precision ensures that the resulting oligosaccharide chains, such as the trisaccharide shown in Formula V, possess the exact linkage patterns required for biological activity, eliminating the formation of unwanted isomers that complicate downstream purification.
Impurity control is inherently managed through the dual mechanisms of enzymatic specificity and fluorine-based separation. Because the glycosyltransferases exhibit strict substrate recognition, side reactions are minimized at the molecular level, resulting in a cleaner crude reaction mixture compared to chemical glycosylation methods. Following the enzymatic assembly, the fluorine tag serves as a powerful handle for purification via Fluorine Solid Phase Extraction (FSPE). In this step, the fluorinated product binds strongly to the fluorinated silica column, while non-fluorinated impurities, unreacted enzymes, and salts are washed away using a methanol and water mixture. This orthogonal purification strategy effectively removes contaminants that would otherwise co-elute in standard chromatography. The combination of high-fidelity enzymatic synthesis and robust fluorine-based isolation creates a closed-loop system for quality assurance, ensuring that the final oligosaccharide products meet the stringent purity specifications demanded by the pharmaceutical industry.
How to Synthesize Fluorine-Labeled Oligosaccharides Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for producing complex oligosaccharides with high efficiency and reproducibility. The process begins with the chemical preparation of the fluorine-labeled lactose acceptor, followed by a series of enzymatic elongation steps that build the sugar chain unit by unit. Each step is optimized for mild reaction conditions, typically utilizing Tris-HCl buffers at physiological pH levels and temperatures around 37°C, which preserves the integrity of the sensitive carbohydrate structures. The detailed standardized synthesis steps below outline the specific reagents, concentrations, and incubation times required to achieve the high yields reported in the experimental data, serving as a foundational guide for process chemists looking to implement this technology.
- Chemically synthesize the beta-configuration lactoside fluorine label (Formula IV) by reacting lactose with acetic anhydride and sodium acetate, followed by activation with trichloroacetonitrile and coupling with a double fluorine chain receptor.
- Prepare the enzymatic reaction mixture by combining the fluorine label, sugar matrix (e.g., N-acetylglucosamine), nucleoside triphosphates (ATP/UTP), and MgCl2 in a Tris-HCl buffer solution.
- Add specific enzymatic modules (e.g., NahK/GlmU and HpLgtA) to the mixture, incubate at 37°C, and subsequently purify the product using fluorine solid phase extraction to separate non-fluorinated impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this fluorine-tag assisted enzymatic synthesis offers profound advantages in terms of cost structure and operational reliability. The shift from chemical synthesis to enzymatic assembly fundamentally alters the cost drivers of oligosaccharide production. By utilizing inexpensive monosaccharides as starting materials and generating expensive activated donors in situ through enzymatic conversion, the process drastically reduces the reliance on costly synthetic reagents. This intrinsic cost reduction in pharmaceutical intermediate manufacturing is further amplified by the elimination of complex protecting group strategies, which traditionally require multiple additional reaction steps and significant volumes of organic solvents. The streamlined workflow not only lowers direct material costs but also reduces the burden on waste treatment facilities, aligning with increasingly strict environmental compliance standards.
- Cost Reduction in Manufacturing: The economic model of this technology is built on the efficiency of biocatalysis and the recyclability of the fluorine tag. Unlike traditional methods that consume stoichiometric amounts of expensive activators, the enzymatic modules regenerate cofactors and utilize cheap sugar precursors, leading to substantial cost savings. Additionally, the fluorine label is designed for easy cleavage and recovery, meaning the specialized fluorous reagents do not represent a sunk cost but rather a reusable asset. This circular approach to reagent usage significantly optimizes the bill of materials, making the production of high-value oligosaccharides more economically viable for large-scale applications.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of prokaryotic enzymes, which can be produced in high yields through fermentation, ensuring a stable and scalable source of biocatalysts. The robustness of the enzymatic modules reduces the risk of batch failures associated with sensitive chemical catalysts. Furthermore, the simplified purification process via FSPE shortens the overall production cycle time, allowing for faster turnaround from raw materials to finished goods. This agility enables suppliers to respond more rapidly to fluctuating market demands, reducing lead times for high-purity oligosaccharides and ensuring a steady flow of critical intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction system facilitates straightforward scale-up from laboratory benchtops to industrial reactors without the mass transfer limitations often encountered in solid-phase synthesis. The ability to monitor reactions in real-time ensures consistent quality across different batch sizes, mitigating the risks associated with commercial scale-up of complex polymer additives or drug conjugates. From an environmental perspective, the reduction in organic solvent usage and the avoidance of heavy metal catalysts simplify waste disposal protocols. This green chemistry profile not only lowers regulatory hurdles but also enhances the sustainability credentials of the supply chain, a key consideration for modern pharmaceutical sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of fluorine-labeled enzymatic synthesis. These insights are derived directly from the patent specifications and experimental data, providing clarity on the practical aspects of adopting this advanced manufacturing technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: What are the advantages of using fluorine labels over traditional solid-phase synthesis for oligosaccharides?
A: Traditional solid-phase synthesis often suffers from heterogeneous reaction conditions, leading to low glycosylation efficiency and difficult monitoring. The fluorine label method described in patent CN112940058A operates in a homogeneous system, allowing for conventional reaction monitoring and higher efficiency. Furthermore, the fluorine tag facilitates rapid separation via Fluorine Solid Phase Extraction (FSPE), avoiding the need for expensive high-fluorinated solvents required in liquid-liquid extraction.
Q: How does the enzymatic module approach reduce production costs for complex sugars?
A: The process utilizes prokaryotic source enzymes such as glycosyltransferases and sugar nucleoside generating enzymes, which offer high protein expression and catalytic efficiency. Crucially, the sugar nucleotide generating enzymes can efficiently convert cheap, easily obtained monosaccharides into expensive nucleoside-activated glycosyl donors in situ. This eliminates the need to purchase costly activated donors, significantly reducing the raw material costs associated with complex oligosaccharide manufacturing.
Q: Is the fluorine tag reusable after the synthesis process?
A: Yes, a key feature of this technology is the design of the fluorine label for easy removal and recovery. After the oligosaccharide chain is assembled and purified via fluorine solid phase extraction, the fluorine carrier can be cleaved from the final product. This recyclability contributes to the overall economic viability of the process, making it suitable for large-scale applications where reagent costs are a critical factor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine Label Oligosaccharide Supplier
The technological breakthroughs presented in patent CN112940058A represent a significant leap forward in the field of carbohydrate chemistry, offering a viable solution for the mass production of complex oligosaccharides. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these advanced synthesis methods to the global market. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of fluorine-labeled intermediates meets the exacting standards required for pharmaceutical applications. We understand the critical nature of supply chain stability and are dedicated to providing a consistent, high-quality source of these essential building blocks.
We invite potential partners to engage with our technical procurement team to explore how this technology can optimize your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your production volume. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these efficient enzymatic synthesis routes into your supply chain. Together, we can drive the next generation of glycan-based therapeutics forward with speed, precision, and economic efficiency.
