Advanced One-Pot Synthesis of Aryl Alkyl Selenides for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries have long recognized the critical value of organoselenium compounds, particularly aryl alkyl selenides, which serve as potent bioactive scaffolds in drug discovery. As illustrated in 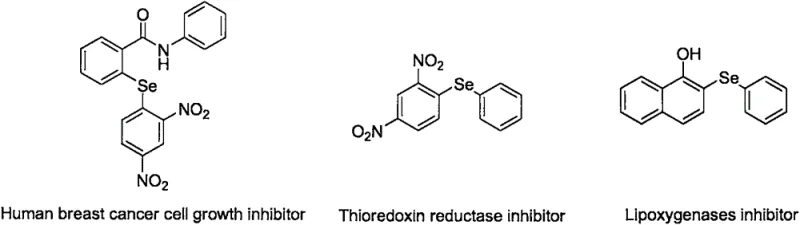 , these structures are foundational to anti-inflammatory agents like Ebselen and various anticancer candidates. However, the widespread adoption of these molecules has historically been bottlenecked by complex, hazardous, and costly synthetic routes. A groundbreaking solution to this challenge is detailed in Chinese Patent CN108658822B, filed in 2020, which introduces a highly efficient, one-pot tandem reaction strategy. This novel methodology transforms the synthesis landscape by utilizing inexpensive elemental selenium, readily available anilines, and epoxides, thereby offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their supply chain. The patent demonstrates that by leveraging a specific combination of tert-butyl nitrite and a base, manufacturers can achieve high yields and exceptional purity without the need for pre-functionalized selenium reagents, marking a significant paradigm shift in fine chemical manufacturing.
, these structures are foundational to anti-inflammatory agents like Ebselen and various anticancer candidates. However, the widespread adoption of these molecules has historically been bottlenecked by complex, hazardous, and costly synthetic routes. A groundbreaking solution to this challenge is detailed in Chinese Patent CN108658822B, filed in 2020, which introduces a highly efficient, one-pot tandem reaction strategy. This novel methodology transforms the synthesis landscape by utilizing inexpensive elemental selenium, readily available anilines, and epoxides, thereby offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their supply chain. The patent demonstrates that by leveraging a specific combination of tert-butyl nitrite and a base, manufacturers can achieve high yields and exceptional purity without the need for pre-functionalized selenium reagents, marking a significant paradigm shift in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the carbon-selenium bond in aryl alkyl selenides has relied on methodologies that are fraught with logistical and economic inefficiencies. Traditional approaches often necessitate the prior preparation of highly reactive and unstable organometallic selenium species. For instance, earlier literature describes the use of phenylselenyl bromide reacted with elemental zinc to generate a phenylselenolate zinc reagent, as depicted in 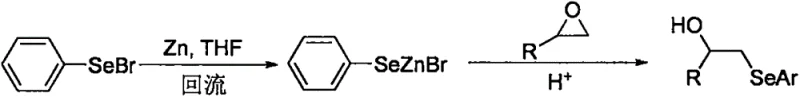 . This multi-step prerequisite not only increases the overall lead time for high-purity pharmaceutical intermediates but also introduces significant safety hazards associated with handling pyrophoric zinc and toxic bromine compounds. Furthermore, alternative methods employing rare earth metals like ytterbium to activate diselenides suffer from prohibitive costs and severe environmental concerns regarding heavy metal waste disposal. These conventional pathways typically require stringent anhydrous conditions, cryogenic temperatures, or excessive stoichiometric amounts of metal reductants, rendering them unsuitable for the commercial scale-up of complex polymer additives or drug precursors where cost reduction in fine chemical manufacturing is paramount.
. This multi-step prerequisite not only increases the overall lead time for high-purity pharmaceutical intermediates but also introduces significant safety hazards associated with handling pyrophoric zinc and toxic bromine compounds. Furthermore, alternative methods employing rare earth metals like ytterbium to activate diselenides suffer from prohibitive costs and severe environmental concerns regarding heavy metal waste disposal. These conventional pathways typically require stringent anhydrous conditions, cryogenic temperatures, or excessive stoichiometric amounts of metal reductants, rendering them unsuitable for the commercial scale-up of complex polymer additives or drug precursors where cost reduction in fine chemical manufacturing is paramount.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the technology disclosed in CN108658822B presents a streamlined, atom-economical solution that bypasses the need for pre-activated selenium reagents entirely. The core innovation lies in a tandem reaction system where elemental selenium powder serves directly as the selenoylation source. As shown in the general reaction scheme in 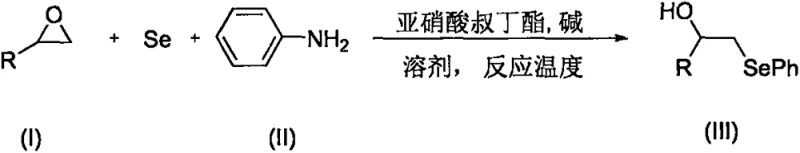 , the process combines an epoxide, aniline, and elemental selenium in a single vessel. Under the synergistic promotion of tert-butyl nitrite and a mild base such as sodium carbonate, the reaction proceeds smoothly to yield the target aryl alkyl selenide. This approach drastically simplifies the operational workflow by eliminating the isolation of intermediate organometallic species. The use of common organic solvents like toluene and the ability to run the reaction at a mild 30°C further underscore its practicality. This represents a transformative advancement for any entity aiming for cost reduction in electronic chemical manufacturing or pharmaceutical production, as it replaces expensive catalysts and complex setups with a simple, mix-and-heat protocol that delivers superior results.
, the process combines an epoxide, aniline, and elemental selenium in a single vessel. Under the synergistic promotion of tert-butyl nitrite and a mild base such as sodium carbonate, the reaction proceeds smoothly to yield the target aryl alkyl selenide. This approach drastically simplifies the operational workflow by eliminating the isolation of intermediate organometallic species. The use of common organic solvents like toluene and the ability to run the reaction at a mild 30°C further underscore its practicality. This represents a transformative advancement for any entity aiming for cost reduction in electronic chemical manufacturing or pharmaceutical production, as it replaces expensive catalysts and complex setups with a simple, mix-and-heat protocol that delivers superior results.
Mechanistic Insights into Tert-Butyl Nitrite Promoted Selenation
The efficacy of this novel synthesis route is rooted in a sophisticated yet elegant mechanistic pathway involving in situ nitrosation and radical generation. The reaction initiates with the interaction between aniline and elemental selenium, facilitated by tert-butyl nitrite (tBuONO). The tBuONO acts as a nitrosating agent, converting the aniline into a diazonium-like intermediate or a reactive nitroso species that effectively activates the elemental selenium surface. This activation generates a highly nucleophilic selenium species, likely a selenolate or a radical selenium intermediate, directly within the reaction medium. This reactive species then attacks the electrophilic carbon of the epoxide ring, triggering a ring-opening event. The presence of the base, specifically sodium carbonate, plays a dual role: it neutralizes acidic byproducts generated during the nitrosation process and assists in the deprotonation steps necessary to drive the equilibrium toward the final beta-hydroxy selenide product. This intricate balance ensures that the reaction proceeds with high chemoselectivity, minimizing the formation of homocoupled diselenide byproducts that often plague selenium chemistry.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material or API intermediates. Because the reactive selenium species is generated in situ and immediately consumed by the epoxide, the concentration of free, unreactive selenium species remains low, thereby suppressing side reactions such as the oxidation of the selenide to selenoxide or the formation of diaryl diselenides. The mild reaction temperature of 30°C further contributes to impurity suppression by preventing thermal degradation of sensitive functional groups on the epoxide or aniline substrates. The patent data indicates that this method tolerates a wide range of substituents, including halogens and ethers, without compromising yield. Consequently, the resulting crude product requires minimal purification, often needing only standard column chromatography to achieve analytical purity, which is a critical factor for maintaining stringent purity specifications in regulated industries.
How to Synthesize Aryl Alkyl Selenide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize the benefits of the tandem reaction system. The process is designed to be user-friendly, utilizing standard glassware and inert atmosphere techniques common in organic synthesis facilities. The key to success lies in the precise stoichiometric balance of the promoters and the maintenance of the nitrogen atmosphere to prevent oxidation of the sensitive selenium intermediates. By following the optimized conditions outlined in the patent, manufacturers can consistently achieve yields exceeding 90% for a variety of substrates, ensuring a reliable supply of these valuable intermediates. The detailed standardized synthesis steps are provided in the guide below to facilitate immediate technology transfer and process validation.
- Combine aniline, elemental selenium, epoxide, tert-butyl nitrite, and sodium carbonate in toluene under a nitrogen atmosphere.
- Stir the reaction mixture at a controlled temperature of 30°C for approximately 24 hours to ensure complete conversion.
- Perform standard workup including extraction with ethyl acetate, washing with brine, drying, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages that extend far beyond simple chemical transformation. The shift from multi-step organometallic protocols to a direct one-pot synthesis fundamentally alters the cost structure and risk profile of producing aryl alkyl selenides. By removing the dependency on pre-synthesized, unstable reagents like phenylselenyl bromide, companies can significantly reduce their raw material inventory costs and eliminate the safety liabilities associated with storing hazardous intermediates. Furthermore, the use of commodity chemicals such as elemental selenium, aniline, and sodium carbonate ensures a stable and resilient supply chain, insulating production schedules from the volatility often seen with specialized organometallic reagents. This robustness is essential for maintaining continuous manufacturing operations and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive and wasteful pre-activation steps. Traditional methods often require stoichiometric amounts of zinc or rare earth metals like ytterbium, which not only add direct material costs but also generate substantial quantities of heavy metal waste that require costly disposal procedures. In contrast, this novel method utilizes catalytic or near-stoichiometric amounts of inexpensive promoters and avoids heavy metals entirely. The simplified workup procedure, which involves basic extraction and filtration rather than complex quenching of reactive metals, further reduces labor and utility costs. These cumulative efficiencies result in substantial cost savings per kilogram of product, enhancing the overall margin profile for high-value selenium-containing APIs.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the accessibility of the starting materials. Elemental selenium and aniline are bulk commodities produced at a global scale, ensuring that sourcing bottlenecks are virtually non-existent compared to niche organoselenium reagents. The mild reaction conditions (30°C) also reduce the energy burden on manufacturing facilities, allowing for production in standard reactors without the need for specialized cryogenic or high-pressure equipment. This flexibility enables manufacturers to diversify their production sites and scale up capacity rapidly in response to market demand, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent availability for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The absence of heavy metal contaminants like zinc or copper simplifies the wastewater treatment process and reduces the regulatory burden associated with metal residue limits in final drug substances. The reaction generates minimal hazardous waste, and the solvents used, such as toluene, are easily recoverable and recyclable. This eco-friendly profile not only lowers compliance costs but also enhances the corporate sustainability image of the manufacturer. The scalability is further evidenced by the patent's assertion that reaction efficiency improves upon amplification, making it an ideal candidate for transition from gram-scale R&D to multi-ton commercial production without the typical teething problems associated with exothermic organometallic reactions.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries based on the detailed experimental data provided in the patent documentation. These insights address critical concerns regarding substrate scope, reaction optimization, and product quality, ensuring that stakeholders have a clear understanding of the process capabilities. The following responses are derived directly from the comparative studies and optimization experiments conducted during the development of this innovative synthesis route.
Q: What are the key advantages of this selenium synthesis method over traditional zinc or ytterbium methods?
A: This method eliminates the need for pre-synthesized organometallic reagents like phenylselenyl bromide or expensive rare earth metals like ytterbium. It utilizes cheap elemental selenium and operates under mild conditions (30°C), significantly reducing environmental pollution and operational complexity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is suitable for large-scale industrial production. The reaction conditions are mild, the reagents are commercially available and inexpensive, and the post-processing involves standard extraction and chromatography techniques that are easily scalable.
Q: What is the role of tert-butyl nitrite in this reaction?
A: Tert-butyl nitrite acts as a promoter in conjunction with the base. It facilitates the in situ generation of reactive selenium species from elemental selenium and aniline, enabling the tandem reaction with the epoxide without requiring harsh activation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Alkyl Selenide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the lab are faithfully reproduced on an industrial scale. We understand the critical nature of selenium chemistry and have established stringent purity specifications and rigorous QC labs to guarantee that every batch of aryl alkyl selenide meets the highest international standards. Our facility is equipped to handle the specific nuances of this tandem reaction, from the safe handling of elemental selenium to the precise control of nitrosation conditions, delivering a product that is ready for immediate use in drug substance manufacturing.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cutting-edge technology for their next-generation projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of these critical selenium intermediates, driving your innovation forward while optimizing your bottom line.
