Advanced Palladium-Catalyzed Synthesis of Polycyclic Spiroindoline Intermediates for Commercial Scale
The pharmaceutical industry is constantly seeking robust and scalable methods to access complex molecular scaffolds that possess significant biological activity. Patent CN110437238B introduces a groundbreaking approach to the synthesis of polycyclic spiroindoline compounds, a class of molecules that are structurally related to potent natural products like vincadifformine and aspidospermidine. These compounds are characterized by their intricate [6,5,5,6]-ABCD tetracyclic indoline skeletons, which are notoriously difficult to construct using traditional organic synthesis methods. The patent discloses a series of new polycyclic spiroindoline derivatives that demonstrate promising cytotoxicity against cancer cell lines, specifically the human oral epidermoid carcinoma cells KB. This technological breakthrough represents a significant leap forward in the field of medicinal chemistry, offering a viable pathway to develop a new class of anticancer drug candidates. By leveraging a palladium-catalyzed reaction between 3-(2-isocyanoethyl)-indoles and various diazo compounds, this method provides a versatile platform for generating diverse chemical libraries.
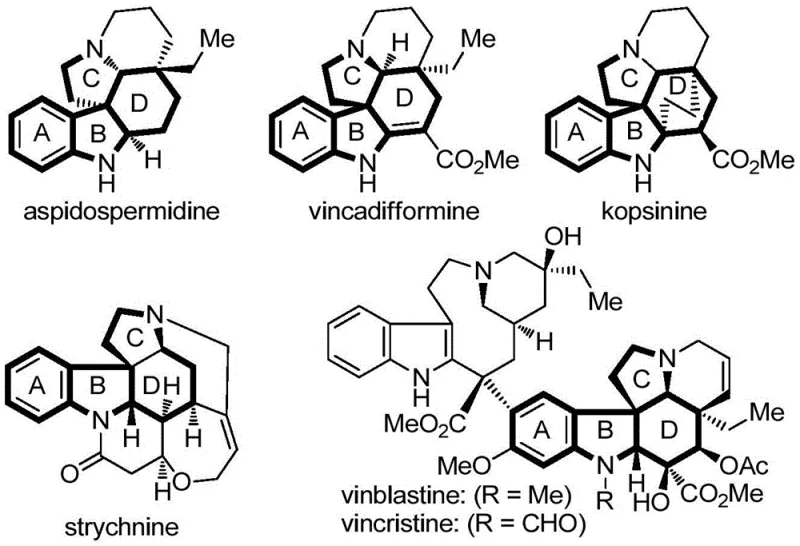
Historically, the acquisition of polycyclic spiroindoline structures has been heavily reliant on extraction from natural plant sources, a process fraught with inefficiencies and supply chain vulnerabilities. Natural products containing these skeletons, such as the alkaloid kopsinine, are often found in very low concentrations within the plant matrix, necessitating the processing of vast quantities of biomass to isolate minute amounts of the target molecule. Furthermore, the growth cycles of these plants are typically long, making it difficult to respond rapidly to market demands or scale up production for clinical trials. The variability in natural sources also introduces significant risks regarding batch-to-batch consistency and purity, which are critical parameters for regulatory approval in the pharmaceutical sector. In contrast, the novel approach detailed in the patent utilizes a de novo total synthesis strategy that bypasses these biological limitations entirely. By constructing the core skeleton chemically, manufacturers can achieve a level of control over the process that is simply impossible with extraction methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The conventional reliance on plant extraction for obtaining polycyclic spiroindoline alkaloids presents a multitude of challenges that hinder the efficient development of new therapeutics. One of the most pressing issues is the extremely low abundance of these target molecules in their natural hosts, which drives up the cost of goods significantly and creates a bottleneck for large-scale production. Additionally, the extraction process often involves the use of large volumes of organic solvents and complex purification steps to separate the desired alkaloid from a myriad of structurally similar impurities, leading to substantial environmental waste and increased operational costs. The structural complexity of these natural products, which often includes multiple chiral centers and quaternary carbons, makes semi-synthetic modification difficult, limiting the ability of medicinal chemists to optimize the pharmacological properties of the lead compounds. Consequently, the supply of these valuable scaffolds is often inconsistent, posing a serious risk to the continuity of drug development programs that depend on them.
The Novel Approach
The synthetic methodology disclosed in patent CN110437238B offers a transformative solution to these challenges by enabling the efficient construction of the polycyclic spiroindoline core through a catalytic cascade reaction. This approach utilizes readily available starting materials, specifically 3-(2-isocyanoethyl)-indoles and diazo compounds, which can be synthesized or procured in large quantities with high purity. The reaction conditions are remarkably mild, often proceeding at temperatures ranging from 25°C to 150°C, which reduces the energy consumption and safety risks associated with high-temperature processes. Furthermore, the method exhibits excellent functional group tolerance, allowing for the introduction of various substituents at different positions on the molecular scaffold without the need for extensive protecting group strategies. This modularity not only accelerates the structure-activity relationship (SAR) studies but also facilitates the rapid optimization of the synthesis process for commercial manufacturing.
Mechanistic Insights into Palladium-Catalyzed Cyclization
The core of this innovative synthesis lies in the palladium-catalyzed reaction mechanism that facilitates the formation of the complex spirocyclic framework. The reaction initiates with the coordination of the palladium catalyst to the diazo compound, leading to the generation of a reactive metal-carbene intermediate. This electrophilic species then undergoes a nucleophilic attack by the isonitrile group of the indole derivative, triggering a cascade of bond-forming events that ultimately result in the construction of the tetracyclic system. The versatility of the palladium catalyst allows for fine-tuning of the reaction pathway, ensuring high selectivity for the desired spiroindoline product over potential side reactions. The use of various palladium sources, such as tetrakis(triphenylphosphine)palladium or tris(dibenzylideneacetone)dipalladium, provides chemists with the flexibility to optimize the catalytic cycle for specific substrate combinations.
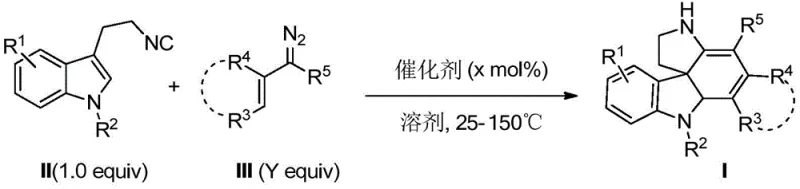
Impurity control is a critical aspect of this synthetic route, particularly given the potential for side reactions involving the highly reactive diazo species. The patent demonstrates that by carefully selecting the reaction solvent and temperature, the formation of by-products can be minimized, leading to crude products that are amenable to straightforward purification. For instance, the use of polar aprotic solvents like DMF or THF helps to stabilize the reaction intermediates and promote the desired cyclization pathway. Additionally, the stoichiometry of the reactants can be adjusted to ensure complete consumption of the limiting reagent, thereby reducing the burden on downstream purification processes. The robustness of this method is evidenced by the successful synthesis of a wide range of derivatives with varying electronic and steric properties, all of which were obtained in good to excellent yields. This level of control over the reaction outcome is essential for meeting the stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize Polycyclic Spiroindoline Efficiently
The practical implementation of this synthesis involves a straightforward procedure that can be adapted for both laboratory-scale discovery and pilot-scale production. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent the decomposition of sensitive reagents. The specific choice of catalyst and solvent is dictated by the nature of the substrates, with the patent providing extensive guidance on optimal conditions for different derivative classes. Following the reaction, the workup procedure is designed to be simple and efficient, typically involving direct column chromatography to isolate the pure product. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Mix 3-(2-isocyanoethyl)-indole derivatives with diazo compounds under nitrogen protection.
- Add a palladium catalyst such as Pd(PPh3)4 or Pd2(dba)3 and a suitable solvent like DMF or THF.
- Stir the reaction mixture at temperatures between 25°C and 150°C until completion, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that directly address the pain points of procurement and supply chain management in the pharmaceutical industry. The ability to synthesize these complex intermediates from cheap and easily obtainable raw materials significantly reduces the dependency on volatile natural product markets, thereby stabilizing the supply chain. The mild reaction conditions and operational simplicity translate to lower energy costs and reduced equipment wear, contributing to overall cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the high yield and purity achieved with this method minimize waste generation and simplify the purification process, aligning with modern environmental compliance standards and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The elimination of expensive extraction processes and the use of cost-effective catalysts lead to a significant decrease in the overall production cost of these valuable intermediates. By avoiding the need for large-scale biomass processing, manufacturers can allocate resources more efficiently and reduce the capital expenditure associated with extraction infrastructure. The high atom economy of the reaction also ensures that raw materials are utilized effectively, further driving down the cost per kilogram of the final product. This economic efficiency makes the commercialization of drugs based on this scaffold much more viable.
- Enhanced Supply Chain Reliability: Synthetic production ensures a consistent and reliable supply of polycyclic spiroindoline compounds, independent of seasonal variations or agricultural disruptions that affect plant-based sources. The scalability of the chemical process allows for rapid ramp-up of production volumes to meet sudden increases in demand, such as those encountered during clinical trial phases or market launch. This reliability is crucial for maintaining the continuity of drug development pipelines and ensuring that patients have access to life-saving medications without interruption. The ability to produce these intermediates on demand reduces the need for large inventory buffers, optimizing working capital.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, allowing for the transition from gram-scale laboratory synthesis to ton-scale commercial production with minimal process re-engineering. The use of standard organic solvents and the absence of highly toxic reagents simplify the handling of waste streams, making it easier to comply with increasingly stringent environmental regulations. The reduced environmental footprint of this synthetic route enhances the sustainability profile of the manufacturing process, which is becoming a key differentiator in the global pharmaceutical market. This alignment with green chemistry principles adds value to the supply chain beyond mere cost considerations.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis technology. These answers are derived directly from the patent data to provide accurate and reliable information for stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows. The insights provided here aim to clarify the operational requirements and potential benefits of adopting this novel methodology.
Q: What are the primary advantages of this synthetic route over plant extraction?
A: This synthetic method overcomes the low content and long growth cycles associated with plant extraction, providing a more reliable and scalable source of polycyclic spiroindoline skeletons.
Q: Which catalysts are compatible with this transformation?
A: The patent specifies a wide range of palladium catalysts including Pd(PPh3)4, Pd2(dba)3, and Pd(OAc)2, allowing for flexibility in process optimization.
Q: What is the biological potential of these compounds?
A: The synthesized compounds exhibit cytotoxicity against human oral epidermoid cancer cells KB, indicating their potential as lead compounds for new anticancer drugs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic Spiroindoline Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, possessing the technical expertise required to translate complex patent methodologies like CN110437238B into commercial reality. Our team of experienced chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the laboratory to the market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polycyclic spiroindoline intermediate meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for your pharmaceutical development needs.
We invite you to collaborate with us to optimize your supply chain and reduce costs through the adoption of this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements. Please contact us to request specific COA data and route feasibility assessments that will demonstrate the value of partnering with NINGBO INNO PHARMCHEM. Let us help you secure a stable and cost-effective supply of these critical anticancer drug intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
