Scalable Manufacturing of High-Purity EGFR Inhibitor Intermediates via Optimized Reduction and Acylation
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex oncology intermediates, particularly those targeting resistant mutations like EGFR T790M. Patent CN116217495A discloses a groundbreaking preparation method for the compound N-((5-((5-chloro-4-((naphthalene-2-yl) amino)) pyrimidin-2-yl) amino)-2-((N-methyl-N-dimethylaminoethyl) amino)-4-methoxyphenyl) acrylamide, designated as Formula I. This molecule serves as a critical intermediate in the synthesis of third-generation EGFR inhibitors, designed to overcome drug resistance induced by earlier therapies such as gefitinib. The disclosed technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to impurity profiles and operational scalability that have plagued previous synthetic routes.
Unlike conventional methods that rely on harsh reduction conditions or unstable acylating agents, this invention introduces a refined catalytic system and a novel acylation-elimination strategy. By shifting away from traditional metal powder reductions and direct acryloylation, the process achieves exceptional purity levels, with single impurities controlled below 0.10 percent. For procurement managers and supply chain directors, this translates to a reliable pharmaceutical intermediate supplier capability that ensures consistent quality without the bottlenecks of complex purification steps. The ability to scale this reaction to 1000L reactors while maintaining high yield and quality underscores its readiness for commercial adoption in the competitive landscape of API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those described in CN105601573B, typically employ reduction systems involving iron powder with ammonium chloride or hydrogenation using palladium on carbon and Raney nickel. While chemically feasible on a small scale, these methods present severe drawbacks for industrial application. The iron powder-ammonium chloride system generates substantial quantities of solid waste sludge, creating significant environmental disposal burdens and complicating post-reaction workup procedures. Furthermore, when scaled to larger vessels like 200L reactors, the iron powder tends to settle at the bottom, causing severe stirring difficulties that prevent the reaction from reaching completion. Alternatively, catalytic hydrogenation with Pd/C or Raney nickel often leads to the formation of specific byproducts like Int3-Imp7, which subsequently convert into the difficult-to-remove impurity Imp14 in the final product.
Additionally, the traditional final step involves direct reaction with acryloyl chloride. This highly reactive reagent inevitably promotes dimerization, leading to the formation of Imp24, a structural analog similar to osimertinib dimers. Removing Imp24 requires cumbersome and costly purification processes, drastically reducing overall process efficiency. These cumulative issues result in lower yields, higher production costs, and inconsistent batch quality, making the conventional route unsuitable for the rigorous demands of modern cost reduction in API manufacturing. The inability to effectively control these specific impurities poses a significant risk to regulatory compliance and supply continuity for downstream drug producers.
The Novel Approach
The innovative methodology presented in CN116217495A fundamentally reengineers the synthesis pathway to bypass these critical failure points. Instead of using iron powder or precious metal hydrogenation, the new process utilizes a ferric chloride-catalyzed reduction system with hydrazine hydrate in the presence of activated carbon. This homogeneous-like catalytic environment ensures efficient mixing and reaction kinetics, completely avoiding the settling issues associated with solid iron powder. More importantly, this specific catalytic combination suppresses the formation of the Int3-Imp7 byproduct, thereby eliminating the downstream generation of Imp14. The result is a much cleaner reaction profile that simplifies isolation and enhances the purity of the intermediate Formula III.
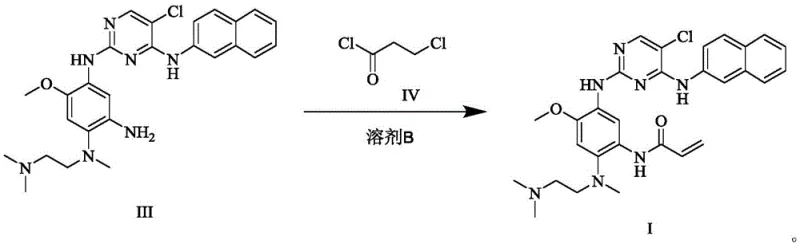
For the final transformation, the patent introduces a clever two-stage sequence involving 3-chloropropionyl chloride followed by base-mediated elimination, rather than direct acryloylation. This strategic modification prevents the uncontrolled dimerization that creates Imp24. As illustrated in the reaction scheme, the intermediate amide is formed under mild conditions and then converted to the acrylamide functionality in situ. This approach not only improves safety by handling less volatile reagents but also ensures that the final product, Formula I, is obtained with purity exceeding 99.8 percent and yields approaching 98 percent. This novel approach offers a commercially viable pathway for the high-purity pharmaceutical intermediate required for next-generation cancer therapies.
Mechanistic Insights into FeCl3-Catalyzed Reduction and Acylation-Elimination
The core of this technological advancement lies in the mechanistic precision of the reduction step. The use of ferric chloride (FeCl3) as a catalyst, combined with activated carbon and hydrazine hydrate, creates a highly selective reducing environment for the nitro group in Formula II. The activated carbon likely serves as a support that enhances the dispersion of the catalyst and facilitates electron transfer, allowing hydrazine to reduce the nitro moiety to an amine without affecting other sensitive functional groups on the pyrimidine ring. This selectivity is crucial because over-reduction or side reactions on the heterocyclic core would lead to the dreaded Int3-Imp7 impurity. By maintaining the reaction temperature between 60°C and 65°C and carefully controlling the molar ratio of hydrazine to substrate (6:1 to 10:1), the process maximizes the conversion to Formula III while keeping impurity levels undetectable by HPLC.
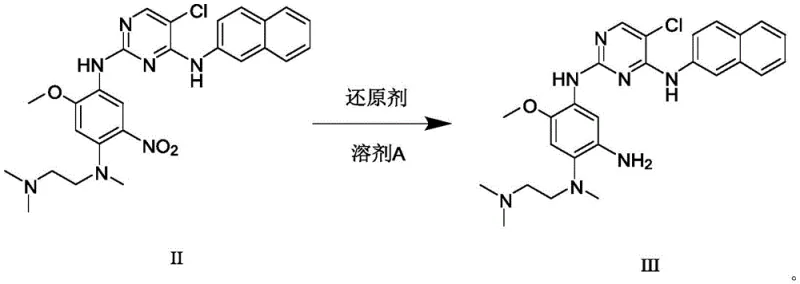
In the subsequent acylation-elimination phase, the mechanism shifts to nucleophilic substitution followed by dehydrohalogenation. The amine group of Formula III attacks the carbonyl carbon of 3-chloropropionyl chloride (Formula IV) to form a chloro-propionamide intermediate. This step is conducted at low temperatures (0°C to 5°C) to minimize side reactions. Following this, the addition of a strong base, such as sodium hydroxide, at elevated temperatures (65°C to 70°C) induces an E2 elimination reaction. The base abstracts a proton from the alpha-carbon relative to the carbonyl, causing the expulsion of the chloride ion and the formation of the carbon-carbon double bond characteristic of the acrylamide group. This stepwise generation of the double bond is far more controlled than direct acylation with acryloyl chloride, effectively suppressing the radical or nucleophilic pathways that lead to the dimeric Imp24 impurity.
How to Synthesize N-((5-((5-Chloro-4-((Naphthalene-2-yl)amino))pyrimidin-2-yl)amino)-2-((N-methyl-N-dimethylaminoethyl)amino)-4-methoxyphenyl)acrylamide Efficiently
The synthesis of this complex EGFR inhibitor intermediate requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high quality. The process is divided into two distinct stages: the catalytic reduction of the nitro precursor and the subsequent acylation-elimination cascade. Operators must pay close attention to temperature control during the exothermic addition of hydrazine and the precise stoichiometry of the base in the elimination step. Detailed standard operating procedures regarding solvent volumes, addition rates, and crystallization conditions are critical for achieving the reported 98 percent plus yields. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol outlined below.
- Perform catalytic reduction of Formula II using ferric chloride and activated carbon with hydrazine hydrate in THF at 60°C to obtain Formula III.
- React Formula III with 3-chloropropionyl chloride in THF at 0-5°C to form the amide intermediate.
- Add hydrochloric acid followed by sodium hydroxide solution and heat to 65°C to effect elimination, yielding the final Formula I compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By eliminating the need for expensive noble metal catalysts like palladium on carbon, the raw material costs are significantly reduced, and the risk of heavy metal contamination in the final API is mitigated. Furthermore, the removal of the iron powder-ammonium chloride system eradicates the generation of hazardous solid waste sludge, which simplifies waste management protocols and reduces environmental compliance costs. These factors collectively contribute to a more sustainable and economically efficient manufacturing process, aligning with the industry's push towards greener chemistry and cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts and the avoidance of complex purification steps for impurity removal lead to direct savings in material and processing costs. The simplified post-treatment, which involves straightforward filtration and crystallization rather than extensive chromatography or extraction, reduces labor and solvent consumption. Additionally, the high yield achieved in the 1000L scale-up demonstrates that the process is robust enough to minimize batch failures and reprocessing, further enhancing overall production economics.
- Enhanced Supply Chain Reliability: The use of readily available reagents such as ferric chloride, hydrazine hydrate, and 3-chloropropionyl chloride ensures a stable supply of raw materials, reducing the risk of shortages associated with specialized catalysts. The scalability of the process, proven from laboratory scale up to 1000L reactors, guarantees that production can be ramped up quickly to meet market demand without the engineering hurdles posed by solid handling issues in traditional methods. This reliability is essential for maintaining continuous supply chains for critical oncology medications.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale operation, with solvent-to-substrate ratios and mixing conditions optimized for industrial reactors. The absence of heavy metal sludge and the reduction of organic waste through higher efficiency make this method more environmentally compliant. This facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, which is increasingly important for corporate social responsibility and long-term operational licensing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does the new method control Imp14 and Imp24 impurities?
A: The method avoids hydrogenation catalysts like Pd/C which generate Int3-Imp7 (precursor to Imp14). It also replaces acryloyl chloride with 3-chloropropionyl chloride followed by elimination, preventing the formation of the dimer impurity Imp24.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scaling to a 1000L reactor (Example 27) with high yield (98.63%) and purity (99.91%), overcoming stirring issues associated with traditional iron powder reduction.
Q: What are the environmental benefits of this synthesis route?
A: By replacing the iron powder-ammonium chloride system, the process eliminates significant amounts of solid waste sludge, simplifying post-treatment and reducing environmental disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-((5-((5-Chloro-4-((Naphthalene-2-yl)amino))pyrimidin-2-yl)amino)-2-((N-methyl-N-dimethylaminoethyl)amino)-4-methoxyphenyl)acrylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest standards for impurity control, specifically targeting the elimination of difficult byproducts like Imp14 and Imp24 as demonstrated in this patent.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your production of this vital EGFR inhibitor intermediate is both efficient and commercially competitive.
