Scalable Stereocontrolled Synthesis of Adenosine Diphosphate Ribose Polypeptides for Drug Discovery
Scalable Stereocontrolled Synthesis of Adenosine Diphosphate Ribose Polypeptides for Drug Discovery
The field of chemical biology and drug discovery relies heavily on precise molecular tools to probe protein functions, particularly those involving post-translational modifications like ADP-ribosylation. A recent technological breakthrough, documented in patent CN112266410A, introduces a highly efficient and stereocontrolled method for synthesizing adenosine diphosphate ribose polypeptides. This innovation addresses the longstanding challenges associated with producing these complex biomolecules, which are critical for studying physiological processes such as DNA repair, chromosome structure regulation, and cell differentiation. By leveraging a streamlined two-step synthetic route starting from commercially available Nicotinamide Adenine Dinucleotide (NAD), this technology offers a robust platform for generating high-purity research tools. For pharmaceutical R&D teams, this represents a significant opportunity to accelerate the identification of new drug targets and the design of lead compounds with greater structural diversity and reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ADP-ribosyl compounds has been fraught with significant technical hurdles that impede both research throughput and commercial scalability. Traditional routes typically commence with base-modified nucleosides, necessitating a series of phosphorylation steps that require harsh reaction conditions and expensive, sensitive reagents. These multi-step processes often involve extensive protection and deprotection strategies to manage the high reactivity of hydroxyl groups, leading to low overall yields and cumbersome purification protocols. Furthermore, the resulting nucleotide intermediates possess high polarity and water solubility, making their isolation from organic reaction mixtures inefficient and prone to product loss. These inefficiencies not only drive up the cost of goods but also limit the structural diversity of the final polypeptide conjugates, restricting the scope of biological investigations.
The Novel Approach
In stark contrast to legacy methods, the novel approach disclosed in the patent utilizes a concise, two-step strategy that capitalizes on the inherent structure of commercial NAD. This method eliminates the need for complex protecting group manipulations, thereby drastically simplifying the synthetic workflow. The first step involves the conversion of NAD into an alpha-ADP-ribose azide intermediate under mild aqueous conditions, followed by a copper-catalyzed azide-alkyne cycloaddition (CuAAC) with alkynyl-functionalized polypeptides. This "click chemistry" approach ensures high reaction specificity and selectivity, facilitating the rapid assembly of diverse ADP-ribose polypeptide conjugates. The use of water as the primary solvent enhances biocompatibility and environmental safety, while the stereochemical control ensures the formation of the biologically relevant alpha-configuration glycosidic bond.
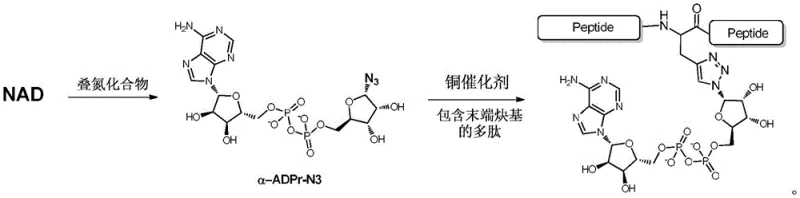
Mechanistic Insights into Stereocontrolled Azide Substitution and Bioconjugation
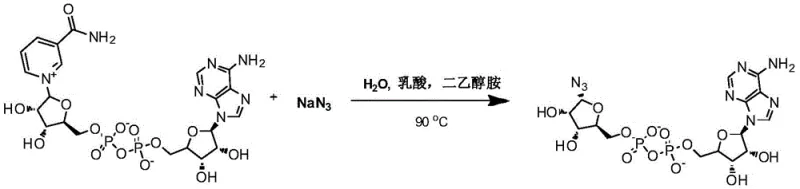
The core of this technological advancement lies in the precise mechanistic control exerted during the transformation of the nicotinamide riboside moiety. In the initial step, the reaction of NAD with sodium azide in the presence of lactic acid and diethanolamine at 90°C facilitates a nucleophilic substitution that effectively replaces the nicotinamide group with an azide functionality. Crucially, this transformation proceeds with high stereochemical fidelity, preserving or establishing the alpha-configuration at the anomeric center of the ribose ring. This is a pivotal achievement, as the biological activity of ADP-ribose derivatives is strictly dependent on their stereochemistry. The reaction conditions are optimized to minimize side reactions such as hydrolysis of the pyrophosphate linkage, ensuring that the integrity of the diphosphate bridge is maintained throughout the synthesis.
Following the generation of the alpha-ADPr-N3 intermediate, the second phase of the synthesis employs a copper(I)-catalyzed azide-alkyne cycloaddition to ligate the nucleotide derivative to the target polypeptide. This bioorthogonal reaction is renowned for its tolerance to aqueous environments and functional group compatibility, making it ideal for modifying sensitive biomolecules. The mechanism involves the formation of a copper-acetylide complex which reacts with the azide to form a stable 1,2,3-triazole linkage. This triazole ring serves as a robust mimic of the native amide or glycosidic bonds found in biological systems, providing metabolic stability to the conjugate. The mild reaction temperature of 37°C further preserves the structural integrity of the polypeptide chain, preventing denaturation or degradation that might occur under harsher chemical conditions.
How to Synthesize Adenosine Diphosphate Ribose Polypeptide Efficiently
The implementation of this synthesis protocol requires careful attention to reaction parameters to maximize yield and purity, particularly given the sensitivity of the pyrophosphate bond. The process begins with the preparation of the alpha-ADPr-N3 intermediate, where precise stoichiometry of sodium azide and buffering agents like lactic acid is essential to drive the equilibrium towards the desired product while suppressing hydrolysis. Subsequent purification via preparative HPLC ensures the removal of unreacted starting materials and byproducts before the conjugation step. The final coupling reaction demands a controlled catalytic environment, utilizing copper sulfate and sodium ascorbate to generate the active Cu(I) species in situ. Detailed standardized operating procedures for scaling this reaction from milligram to kilogram quantities are provided in the technical guide below.
- Dissolve Nicotinamide Adenine Dinucleotide (NAD) and sodium azide in water with lactic acid and diethanolamine, then heat at 90°C to form alpha-ADPr-N3.
- Prepare an aqueous solution containing the alkynyl-functionalized polypeptide and the synthesized alpha-ADPr-N3 intermediate.
- Add copper sulfate pentahydrate and sodium ascorbate catalysts to the mixture and stir at 37°C for 12 hours to complete the bioconjugation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and operational benefits over traditional sourcing models. The elimination of multiple protection and deprotection steps significantly reduces the consumption of specialized reagents and solvents, leading to a substantial reduction in raw material costs. Furthermore, the simplified workflow shortens the overall production cycle time, allowing for faster turnaround on custom orders and improved responsiveness to R&D demands. The use of water as a solvent not only lowers waste disposal costs associated with hazardous organic solvents but also aligns with increasingly stringent environmental regulations, mitigating compliance risks for the supply chain.
- Cost Reduction in Manufacturing: The streamlined two-step process eliminates the need for expensive chiral catalysts and protecting group reagents that characterize conventional nucleoside synthesis. By starting directly from bulk commodity chemicals like NAD, the material cost basis is significantly lowered. Additionally, the high selectivity of the click chemistry step minimizes the formation of difficult-to-separate impurities, reducing the burden on downstream purification units and lowering the overall cost of goods sold for high-purity intermediates.
- Enhanced Supply Chain Reliability: Reliance on commercially available starting materials such as NAD and sodium azide ensures a stable and resilient supply chain, free from the bottlenecks often associated with custom-synthesized building blocks. The robustness of the aqueous reaction conditions allows for manufacturing in facilities with standard chemical processing capabilities, reducing dependency on specialized contract manufacturers. This decentralization potential enhances supply security and reduces lead times for critical research materials needed in drug discovery pipelines.
- Scalability and Environmental Compliance: The transition from organic solvents to an aqueous-based system dramatically simplifies scale-up operations, removing the safety hazards associated with large volumes of flammable solvents. This "green chemistry" approach reduces the volume of hazardous waste generated per kilogram of product, facilitating easier regulatory approval for manufacturing sites. The process is inherently scalable from laboratory benchtop to industrial reactor sizes without requiring fundamental changes to the reaction engineering, ensuring consistent quality across different production batches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ADP-ribose polypeptides using this patented methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these nuances is essential for R&D directors evaluating the suitability of these intermediates for their specific assay development needs and for procurement teams assessing the long-term viability of this supply source.
Q: How does this method improve stereocontrol compared to traditional nucleoside phosphorylation?
A: This novel route utilizes a direct transformation of commercial NAD, bypassing the complex protection and deprotection sequences typically required for base-modified nucleosides. The reaction conditions inherently favor the formation of the alpha-configuration glycosidic bond between the triazole and ribose, ensuring high stereoselectivity without needing chiral auxiliaries.
Q: What are the purification advantages of using water as the primary solvent?
A: Traditional methods often rely on organic solvents that complicate the isolation of highly polar nucleotide intermediates. By conducting the reaction in an aqueous system, the process aligns with the natural solubility profile of ADP-ribose derivatives, simplifying downstream processing and reducing the need for hazardous organic waste disposal.
Q: Is this synthesis method compatible with diverse polypeptide structures?
A: Yes, the method employs a modular copper-catalyzed azide-alkyne cycloaddition (CuAAC) strategy. This allows for the convenient realization of structural diversification, enabling researchers to attach the ADP-ribose moiety to various polypeptides containing terminal alkynyl groups under mild, biocompatible conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adenosine Diphosphate Ribose Polypeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chemical biology tools play in accelerating the pace of pharmaceutical innovation. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your research needs can be met with industrial-grade reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stereochemical purity and structural integrity of complex nucleotide-peptide conjugates, adhering to the most stringent purity specifications required for preclinical and clinical applications. We are committed to delivering consistency and excellence in every batch we produce.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug discovery project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data for our ADP-ribose intermediates and to discuss route feasibility assessments for your custom polypeptide conjugation needs. Let us help you streamline your supply chain and focus on what matters most: discovering the next generation of life-saving therapies.
