Optimizing Tenofovir Intermediate Production: A High-Selectivity Route for (R)-9-(2-Hydroxypropyl) Adenine
The global demand for antiretroviral therapies continues to drive the need for robust, scalable, and cost-effective manufacturing routes for key pharmaceutical intermediates. Patent CN103408548A introduces a significant technological advancement in the synthesis of (R)-9-(2-hydroxypropyl) adenine, a critical precursor for the blockbuster HIV medication Tenofovir Disoproxil. This intellectual property addresses a longstanding challenge in nucleoside analog chemistry: the regioselective alkylation of the purine ring. By implementing a strategic protection-deprotection sequence, the disclosed method effectively suppresses the formation of undesirable isomers, offering a pathway that aligns perfectly with the rigorous quality and efficiency standards required by modern reliable API intermediate suppliers. The technical breakthrough lies not just in the yield, but in the fundamental control of reaction selectivity, which translates directly into simplified downstream processing and enhanced supply chain stability for generic drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct alkylation of adenine with propylene carbonate under alkaline conditions has been the standard approach for accessing hydroxypropyl adenine derivatives. However, this conventional methodology suffers from inherent chemoselectivity issues due to the presence of multiple nucleophilic sites on the adenine scaffold. Specifically, the exocyclic primary amino group at the C-6 position competes with the ring nitrogen atoms for alkylation. This competition inevitably leads to the co-formation of the unwanted (R)-6-(2-hydroxypropyl) adenine isomer alongside the desired N9-alkylated product. The structural similarity between these isomers makes their separation exceptionally difficult and costly, often requiring complex chromatographic techniques that are impractical for large-scale industrial application. Furthermore, the formation of this by-product significantly depresses the overall yield of the target molecule, leading to inefficient raw material utilization and increased waste generation, which are critical pain points for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative strategy detailed in the patent circumvents these selectivity issues through a clever three-step sequence that prioritizes regiocontrol. The process initiates with the selective acetylation of the primary amino group at the C-6 position of adenine. By temporarily masking this reactive site with an acetyl group, the molecule is rendered inert at the C-6 position, effectively directing the subsequent alkylation reaction exclusively to the N9 nitrogen atom. Following the successful N-hydroxypropylation with propylene carbonate, the protecting group is removed via hydrolysis to reveal the final product. This approach not only drastically minimizes the generation of the N6-isomer but also simplifies the isolation procedure, as the intermediate can be purified via straightforward crystallization rather than complex separation methods. 
Mechanistic Insights into Regioselective Acetylation and Alkylation
The core of this synthetic success relies on the precise manipulation of nucleophilicity within the adenine heterocycle. In the first step, the use of 4-Dimethylaminopyridine (DMAP) as a nucleophilic catalyst is paramount. DMAP accelerates the acetylation of the exocyclic amine by forming a highly reactive acetylpyridinium intermediate, which transfers the acetyl group to the amine much faster than uncatalyzed acetic anhydride would. This ensures that the protection step is complete before the temperature is raised for the alkylation phase. The reaction conditions, specifically maintaining a pH of 9 to 10 during the second step, are carefully tuned to deprotonate the N9-H proton, generating the nucleophilic anion required to attack the epoxide ring of the propylene carbonate, while the acetylated amine remains non-nucleophilic. This electronic differentiation is the key to achieving the reported high selectivity.
Furthermore, the process incorporates a sophisticated crystallization protocol to ensure high purity without the need for column chromatography. After the alkylation step, the addition of toluene to the reaction mixture at elevated temperatures (70-80°C) serves as an anti-solvent, inducing the precipitation of the acetylated intermediate. This solid-liquid separation effectively removes soluble impurities and unreacted starting materials. The final deacetylation step utilizes aqueous sodium hydroxide, a cheap and readily available reagent, to cleave the amide bond. The mechanism here involves nucleophilic attack by the hydroxide ion on the carbonyl carbon of the acetyl group, regenerating the free amine and producing acetate as a water-soluble by-product. 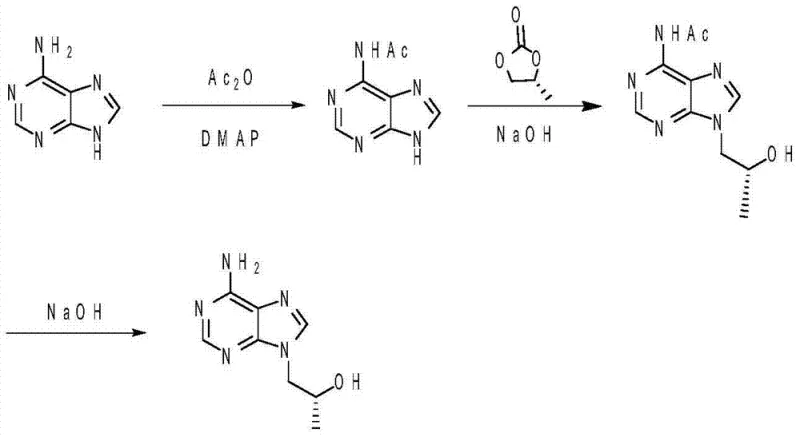
How to Synthesize (R)-9-(2-Hydroxypropyl) Adenine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and stoichiometric precision. The process begins with the dissolution of adenine in polar aprotic solvents such as DMF or NMP, followed by the controlled addition of acetic anhydride at low temperatures to manage exothermicity. The subsequent alkylation requires careful pH monitoring to maintain the optimal window for N9 attack. Finally, the hydrolysis step demands thermal energy to drive the deprotection to completion. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Dissolve adenine in an organic solvent (DMF/NMP/DMSO), add base and DMAP catalyst, then dropwise add acetic anhydride at 0-10°C followed by heating to 80-85°C to achieve primary amino acetylation.
- Adjust the reaction mixture pH to 9-10, add propylene carbonate, and heat to 120-140°C for N-hydroxypropylation, followed by toluene addition for crystallization.
- Hydrolyze the solid intermediate in aqueous sodium hydroxide at 90-95°C, neutralize with hydrochloric acid to pH 6-8, and isolate the final product via filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this regioselective synthesis route offers profound advantages for procurement managers and supply chain directors focused on commercial scale-up of complex pharmaceutical intermediates. The primary value driver is the substantial increase in process yield, which reportedly exceeds 90%. In high-volume manufacturing, a yield improvement of this magnitude translates directly into a significant reduction in the cost of goods sold (COGS), as less raw material is required to produce the same amount of finished intermediate. Additionally, the elimination of the difficult-to-separate N6-isomer removes the need for expensive and time-consuming purification steps, such as preparative HPLC or multiple recrystallizations, further streamlining the production timeline and reducing operational expenditures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the efficient use of raw materials and the simplification of the purification workflow. By preventing the formation of the major by-product at the source, the process avoids the yield losses associated with trying to separate isomers later in the pipeline. The reagents used—acetic anhydride, propylene carbonate, and sodium hydroxide—are commodity chemicals with stable pricing and widespread availability, insulating the supply chain from volatility associated with exotic catalysts. Furthermore, the ability to isolate intermediates via simple filtration and crystallization reduces solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Supply continuity is critical for the production of antiretroviral drugs, and this synthetic route enhances reliability by relying on robust, well-understood chemical transformations. The avoidance of sensitive transition metal catalysts eliminates the risk of supply disruptions related to specialized reagent sourcing and removes the regulatory burden of demonstrating heavy metal clearance in the final API. The process conditions are compatible with standard stainless steel reactor equipment found in most multipurpose chemical plants, facilitating easy technology transfer between different manufacturing sites. This flexibility ensures that production can be scaled or shifted geographically without the need for specialized infrastructure, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is favorable for modern green chemistry initiatives. The reaction generates minimal hazardous waste, primarily consisting of aqueous salt solutions and recoverable organic solvents like toluene and DMF. The high atom economy of the alkylation step, combined with the high yield, means that the E-factor (mass of waste per mass of product) is significantly lower than that of traditional non-selective routes. This alignment with environmental compliance standards simplifies the permitting process for new production lines and supports the sustainability goals of multinational pharmaceutical companies seeking responsible partners for their active ingredient supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: How does this method improve selectivity over traditional alkylation?
A: Traditional methods often result in mixed N-alkylation at both the N9 and exocyclic N6 positions. This patented route utilizes a temporary acetyl protecting group on the primary amino group (C-6 position), effectively blocking side reactions and forcing alkylation exclusively at the N9 position, thereby eliminating the difficult-to-separate (R)-6-(2-hydroxypropyl) adenine by-product.
Q: What represents the critical quality attribute for this intermediate?
A: The critical quality attribute is the isomeric purity, specifically the absence of the N6-alkylated isomer. The described process achieves HPLC purity exceeding 98% and yields over 90%, which is essential for downstream phosphorylation steps in Tenofovir Disoproxil manufacturing where isomeric impurities can complicate purification and reduce final API potency.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It avoids the use of expensive transition metal catalysts or complex chromatographic separations. The workup involves simple pH adjustments and crystallization using common solvents like toluene, making it highly adaptable for multi-ton production scales typical in the generic antiviral supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-9-(2-Hydroxypropyl) Adenine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final antiviral medication depends heavily on the purity and consistency of its precursors. As a leading CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of critical intermediates. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical assessment of isomeric impurities that define the quality of (R)-9-(2-hydroxypropyl) adenine. We are committed to delivering products that meet the exacting standards of the global pharmaceutical industry.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you achieve significant efficiencies in your Tenofovir supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical expertise can support your long-term production goals.
