Advanced Synthesis of Fexofenadine Intermediates: High-Yield Cyclization for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for high-volume antihistamines, particularly for second-generation compounds like Fexofenadine hydrochloride. Patent CN101585782B, published in 2012, introduces a transformative methodology for synthesizing N-methyl-N-methoxy-2-(4-cyclopropylcarbonylphenyl)-2-methylpropanamide, a critical Weinreb amide intermediate in this value chain. Unlike traditional approaches that suffer from low throughput and complex purification requirements, this invention leverages a straightforward base-mediated intramolecular cyclization to construct the cyclopropyl moiety directly from a chlorobutyryl precursor. The technical breakthrough lies in the optimization of reaction stoichiometry and solvent systems, allowing for yields exceeding 90% under mild thermal conditions. For R&D directors and process chemists, this represents a significant opportunity to streamline the supply chain for allergy medication ingredients by adopting a protocol that minimizes waste and maximizes material efficiency without requiring exotic catalysts or high-pressure equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in U.S. Patent 006242606B1, have historically plagued manufacturers with suboptimal economic and operational metrics. The conventional synthetic route typically achieves an overall yield of merely 28%, which is commercially unsustainable for high-demand active pharmaceutical ingredients. Furthermore, the isolation of the target molecule in these older processes necessitates rigorous and energy-intensive purification techniques, specifically molecular distillation followed by recrystallization. These additional unit operations not only extend the production cycle time significantly but also introduce potential points of failure where product degradation or loss can occur. The reliance on such complex downstream processing inflates the cost of goods sold (COGS) and creates bottlenecks in manufacturing capacity, making it difficult for suppliers to respond agilely to market fluctuations in the antihistamine sector.
The Novel Approach
The innovative strategy detailed in CN101585782B circumvents these historical inefficiencies by employing a direct cyclization mechanism using readily available alkali metal hydroxides. By utilizing sodium hydroxide or potassium hydroxide in an alcoholic medium, the process facilitates the formation of the cyclopropane ring through an intramolecular nucleophilic substitution without the need for transition metal catalysts. The reaction conditions are remarkably mild, operating effectively between 20°C and 50°C, which reduces energy consumption compared to high-temperature alternatives. Most critically, the post-reaction workup is drastically simplified to standard liquid-liquid extraction and solvent evaporation, completely eliminating the need for molecular distillation. This shift from complex physical separation to simple chemical conversion allows for a streamlined workflow that enhances throughput and significantly lowers the barrier to entry for commercial-scale production of this key intermediate.
Mechanistic Insights into Base-Mediated Intramolecular Cyclization
The core chemical transformation involves the deprotonation of the alpha-carbon adjacent to the carbonyl group or the direct nucleophilic attack by a carbanion species generated in situ, leading to the displacement of the chloride leaving group and the formation of the three-membered cyclopropyl ring. The patent specifies a molar ratio of alkali metal hydroxide to substrate ranging from 6:1 to 10:1, which is crucial for driving the equilibrium towards product formation and neutralizing the hydrochloric acid byproduct generated during the cyclization. This excess base ensures that the reaction environment remains sufficiently alkaline to sustain the nucleophilic species throughout the 10 to 30-hour reaction window. The choice of alcoholic solvents like methanol or ethanol serves a dual purpose: acting as a polar medium to dissolve the ionic base and the organic substrate, while also stabilizing the transition state of the cyclization. Understanding this mechanism is vital for process chemists aiming to replicate the high purity profiles reported in the patent examples.
Control of impurities in this synthesis is inherently managed by the specificity of the intramolecular reaction and the simplicity of the workup. In conventional methods, side reactions often lead to complex mixtures requiring distillation to separate; however, the high selectivity of this base-mediated route minimizes the formation of polymeric byproducts or open-chain hydrolysis products. The use of dichloromethane for extraction effectively partitions the lipophilic Weinreb amide product from inorganic salts and residual base remaining in the aqueous phase. This phase separation acts as a built-in purification step, ensuring that the final crude product possesses high chemical purity before any final drying or crystallization. For quality assurance teams, this implies a more consistent impurity profile, reducing the analytical burden and facilitating faster release testing for regulatory compliance in pharmaceutical manufacturing.
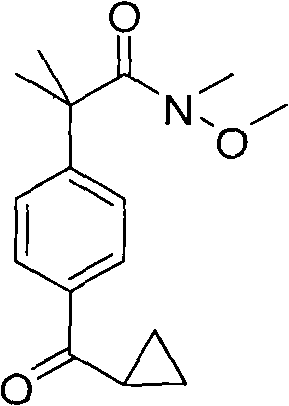
How to Synthesize N-Methyl-N-Methoxy-2-(4-Cyclopropylcarbonylphenyl)-2-Methylpropanamide Efficiently
To implement this high-yield synthesis in a laboratory or pilot plant setting, operators must strictly adhere to the stoichiometric ratios and temperature controls defined in the patent to ensure reproducibility. The process begins with the preparation of a concentrated alkaline alcoholic solution, followed by the controlled addition of the chlorobutyryl precursor to manage exothermicity and reaction kinetics. Maintaining the reaction temperature within the 20-50°C range is essential to balance reaction rate with selectivity, preventing thermal degradation of the sensitive Weinreb amide functionality. Detailed standardized operating procedures regarding mixing speeds, addition rates, and quenching protocols are critical for scaling this chemistry from gram to kilogram quantities. For a comprehensive guide on the exact step-by-step execution, please refer to the technical protocol outlined below.
- Prepare an alkaline alcoholic solution by dissolving alkali metal hydroxide (NaOH or KOH) in methanol or ethanol to achieve a concentration of 2-3.4 mol/L.
- Dissolve the precursor N-methyl-N-methoxy-2-[4-(4-chlorobutyryl)phenyl]-2-methylpropanamide in alcohol solvent to form a 1 mol/L solution.
- Dropwise add the precursor solution to the alkaline mixture maintaining a molar ratio of 6-10: 1, react at 20-50°C for 10-30 hours, then isolate via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers profound advantages in terms of cost structure and supply reliability. The dramatic improvement in yield from the historical baseline of 28% to over 90% fundamentally alters the raw material consumption metrics, meaning significantly less starting material is required to produce the same amount of finished intermediate. This efficiency gain translates directly into reduced raw material costs and lower waste disposal expenses, contributing to a leaner manufacturing model. Furthermore, the elimination of molecular distillation equipment reduces capital expenditure requirements and lowers the energy intensity of the production process, aligning with modern sustainability goals and reducing the overall carbon footprint of the supply chain.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the downstream processing train. By removing the need for high-vacuum molecular distillation and multiple recrystallization steps, manufacturers can significantly reduce utility costs associated with heating, cooling, and vacuum generation. The use of commodity chemicals such as sodium hydroxide and methanol instead of specialized catalysts further drives down the variable cost per kilogram. Additionally, the higher yield means that fixed costs related to labor and facility usage are amortized over a much larger output volume, resulting in substantial overall cost savings per unit of product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is greatly improved due to the robustness and simplicity of the reaction conditions. The reliance on stable, non-hazardous reagents like alkali hydroxides reduces the risk of supply disruptions often associated with specialized or imported catalysts. The shorter and more predictable reaction timeline, combined with a straightforward workup procedure, allows for faster batch turnover times, enabling suppliers to respond more rapidly to urgent demand spikes from downstream API manufacturers. This agility is crucial in the pharmaceutical sector, where inventory buffers are often kept low, and the ability to ramp up production quickly is a key competitive differentiator for reliable partners.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is highly favorable for scale-up. The absence of heavy metal catalysts eliminates the need for complex metal scavenging steps and reduces the toxic load in wastewater streams, simplifying effluent treatment and ensuring compliance with stringent environmental regulations. The solvent system, primarily consisting of alcohols and dichloromethane, is well-understood and easily recoverable through standard distillation, promoting a circular economy approach within the plant. The inherent safety of operating at near-ambient temperatures and atmospheric pressure minimizes operational risks, making the process easier to validate and license for commercial production in diverse global manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN101585782B, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. For further clarification on specific parameters or customization options, direct consultation with our technical team is recommended.
Q: What is the primary advantage of this synthesis method over prior art?
A: The primary advantage is a dramatic increase in yield from approximately 28% in previous methods to over 90%, coupled with the elimination of complex purification steps like molecular distillation.
Q: What are the critical reaction conditions for the cyclization step?
A: The reaction requires a significant excess of base (6-10 equivalents) in an alcoholic solvent at moderate temperatures (20-50°C) over a period of 10 to 30 hours to ensure complete ring closure.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes common reagents like sodium hydroxide and standard solvents like methanol, and the workup involves simple extraction and evaporation, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-N-Methoxy-2-(4-Cyclopropylcarbonylphenyl)-2-Methylpropanamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient production of life-saving medications like Fexofenadine. Our technical team has extensively analyzed the pathway described in CN101585782B and possesses the expertise to execute this high-yield cyclization with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of N-methyl-N-methoxy-2-(4-cyclopropylcarbonylphenyl)-2-methylpropanamide meets the highest industry standards for pharmaceutical applications.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
