Advanced Chiral Cobalt-Nitrogen Complexes for High-Efficiency Henry Reactions in Pharma Manufacturing
The landscape of asymmetric catalysis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in Chinese Patent CN103570768A, which discloses a novel chiral cobalt-nitrogen complex derived from L-phenylglycinol. This invention represents a significant leap forward in the design of multinuclear metal-organic frameworks that serve as potent catalysts for carbon-carbon bond-forming reactions. Specifically, the patent details the preparation and structural characterization of a hexa[(S)-phenylglycinol] cobalt acetate complex, designated as complex (I). For R&D directors and process chemists seeking robust catalytic systems, this material offers a compelling alternative to traditional noble metal catalysts, combining the abundance of cobalt with the precise stereochemical control afforded by chiral amino alcohol ligands. The ability to synthesize this complex in a single step with high reproducibility makes it an attractive candidate for integration into existing synthetic pipelines for API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral catalysts for Henry reactions (nitroaldol reactions) has often relied on expensive transition metals such as copper, zinc, or even rare earth elements, coupled with sophisticated, multi-step ligand synthesis protocols. These conventional approaches frequently suffer from several critical drawbacks that hinder their widespread adoption in large-scale manufacturing. Firstly, the ligands required to induce high enantioselectivity are often proprietary and costly, significantly inflating the raw material expenses for the final active pharmaceutical ingredient. Secondly, many traditional catalytic systems require stringent anhydrous conditions or cryogenic temperatures to maintain stability and selectivity, imposing heavy energy burdens and operational complexity on the production facility. Furthermore, the removal of residual heavy metals from the final product to meet regulatory standards (often below 10 ppm) necessitates additional purification steps, such as scavenging or recrystallization, which inevitably reduce overall yield and extend cycle times. These cumulative inefficiencies create bottlenecks in the supply chain, making the cost reduction in fine chemical manufacturing a persistent challenge for procurement teams.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology described in CN103570768A introduces a streamlined, one-pot synthesis strategy that leverages readily available chiral pool materials. The novel approach utilizes L-phenylglycinol, a commercially accessible chiral amino alcohol, which coordinates directly with cobalt acetate tetrahydrate in absolute ethanol. This method eliminates the need for pre-functionalized ligands or inert atmosphere gloveboxes, as the reaction proceeds efficiently under standard heating and reflux conditions. The resulting complex is a stable, brown-red crystalline solid that can be isolated through simple filtration and solvent volatilization, bypassing the need for column chromatography or complex workup procedures. By simplifying the catalyst preparation to a single thermal step, this innovation drastically reduces the operational overhead and technical barriers associated with catalyst production. Moreover, the use of cobalt, a base metal, inherently lowers the material cost profile compared to precious metal alternatives, aligning perfectly with the strategic goals of reliable pharmaceutical intermediate supplier networks aiming for sustainable cost optimization.
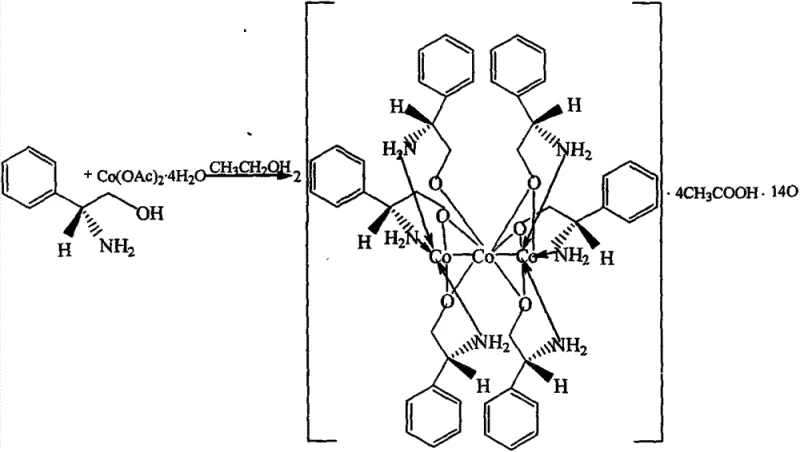
The mechanistic efficacy of this cobalt-nitrogen complex stems from its unique trinuclear architecture, which creates a highly defined chiral pocket around the metal centers. As evidenced by the single-crystal X-ray diffraction data provided in the patent, the complex features three cobalt atoms bridged by oxygen atoms from the phenylglycinol ligands, forming a rigid core that prevents ligand dissociation during catalysis. This structural rigidity is paramount for maintaining the integrity of the chiral environment throughout the reaction coordinate. In the context of the Henry reaction, where a nitroalkane adds to an aldehyde, the cobalt centers act as Lewis acids to activate the carbonyl group of the benzaldehyde, while the adjacent amine or hydroxyl groups of the ligand likely participate in hydrogen bonding or deprotonation of the nitromethane. This cooperative activation lowers the energy barrier for the nucleophilic attack, facilitating the formation of the beta-nitro alcohol product. The patent reports a conversion rate of 61.4% for the reaction between benzaldehyde and nitromethane at room temperature, demonstrating that the catalyst is active under mild conditions without the need for external heating or high pressure. Such mild operating parameters are crucial for preserving the integrity of sensitive functional groups often present in complex drug molecules.
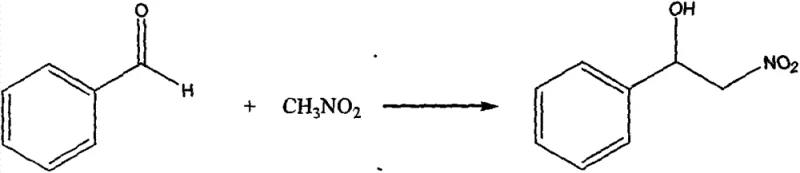
Beyond the primary catalytic cycle, the impurity profile of reactions mediated by this complex is expected to be favorable due to the homogeneous nature of the catalysis and the specific geometric constraints imposed by the ligand sphere. The chiral induction provided by the phenyl rings of the phenylglycinol ligands shields specific faces of the substrate, thereby suppressing the formation of unwanted diastereomers or racemic byproducts. For quality control laboratories, this translates to a cleaner crude reaction mixture, reducing the burden on downstream purification units. The stability of the Co-N and Co-O bonds within the complex ensures that metal leaching is minimized, which is a critical consideration for meeting the stringent purity specifications required by global regulatory bodies. By controlling the stereochemistry at the molecular level through this well-defined coordination geometry, manufacturers can achieve higher optical purity in their intermediates, directly impacting the efficacy and safety profile of the final therapeutic agent.
How to Synthesize Chiral Cobalt Complex Efficiently
The preparation of this high-value catalyst is designed for operational simplicity, allowing chemical engineers to reproduce the results with minimal training or specialized equipment. The protocol involves dissolving the chiral ligand in ethanol, adding the cobalt salt, and applying heat, followed by a straightforward isolation process. This ease of execution is a key factor in its potential for rapid technology transfer from R&D labs to pilot plants. For detailed procedural specifics regarding stoichiometry, temperature ramps, and crystallization kinetics, please refer to the standardized synthesis guide below.
- Dissolve 0.03mol of L-phenylglycinol in 50ml of absolute ethanol within a 100ml round-bottom flask under stirring.
- Add 0.01mol of Cobalt(II) acetate tetrahydrate (Co(OAc)2·4H2O) to the solution and heat the mixture to reflux for 48 hours.
- Filter the hot reaction solution, remove the solvent via rotary evaporation, redissolve the residue in anhydrous methanol, and allow natural volatilization to precipitate brown-red crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cobalt-based catalytic system offers multifaceted benefits that resonate deeply with procurement managers and supply chain heads focused on resilience and margin protection. The shift towards base metal catalysis is not merely a scientific preference but a strategic economic imperative that decouples production costs from the volatile pricing of precious metals like palladium or rhodium. By utilizing a catalyst derived from abundant cobalt and inexpensive amino alcohols, manufacturers can insulate their supply chains from geopolitical fluctuations in metal markets. Furthermore, the simplified synthesis workflow reduces the consumption of utilities and solvents, contributing to a lower overall cost of goods sold (COGS) without compromising on performance. This alignment of technical efficiency with economic prudence makes the technology highly attractive for long-term contracting and capacity planning.
- Cost Reduction in Manufacturing: The elimination of expensive chiral ligands and precious metal salts results in a substantial decrease in raw material expenditures. Since the catalyst synthesis is a one-step process performed in common solvents like ethanol and methanol, the capital expenditure required for specialized reactor setups is significantly reduced. Additionally, the mild reaction conditions for the catalytic application (room temperature) imply lower energy consumption for heating or cooling, further driving down operational costs. These cumulative savings allow for more competitive pricing strategies in the bidding for large-scale API contracts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as cobalt acetate and phenylglycinol ensures a robust and continuous supply of catalyst precursors. Unlike bespoke ligands that may have single-source suppliers and long lead times, the inputs for this complex are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of the isolated brown-red crystals allows for batch production and inventory stocking, enabling just-in-time delivery models that enhance responsiveness to customer demand spikes.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, as the reflux conditions and filtration steps are unit operations that translate seamlessly from liter-scale flasks to cubic-meter reactors. The use of ethanol and methanol, which are greener solvents compared to chlorinated alternatives, aligns with increasingly strict environmental regulations and corporate sustainability goals. The high atom economy of the coordination reaction and the potential for catalyst recycling or low-loading applications contribute to a reduced environmental footprint, simplifying waste treatment protocols and minimizing disposal costs associated with heavy metal contaminants.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and performance of this catalytic technology, we have compiled a set of answers based on the empirical data and structural insights provided in the patent literature. These responses are intended to clarify the operational parameters and potential applications for partners evaluating this technology for their specific synthetic routes. Understanding these nuances is essential for conducting accurate feasibility studies and risk assessments prior to process adoption.
Q: What is the catalytic efficiency of this cobalt complex in Henry reactions?
A: According to patent data, the complex demonstrates significant catalytic activity in the Henry reaction between benzaldehyde and nitromethane, achieving a conversion rate of 61.4% under mild room temperature conditions.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis utilizes a straightforward one-step reflux method in common solvents like ethanol and methanol, avoiding exotic reagents or extreme pressures, which facilitates easy scale-up from laboratory to commercial tonnage.
Q: What are the structural characteristics of this catalyst?
A: The catalyst features a unique trinuclear cobalt core coordinated by chiral L-phenylglycinol ligands, creating a rigid chiral environment essential for inducing stereoselectivity in asymmetric synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cobalt Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced coordination chemistry in modern drug synthesis. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like this chiral cobalt complex can be successfully translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and potency. Whether you require custom synthesis of the catalyst itself or contract manufacturing of intermediates utilizing this technology, our infrastructure is designed to support your growth with reliability and precision.
We invite you to engage with our technical procurement team to discuss how this catalytic solution can be integrated into your specific manufacturing challenges. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic impact of switching to this cobalt-based system. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless transition to more efficient and cost-effective production methodologies.
