Scalable Synthesis of Oxygen-Substituted Geranyl Flavanone for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking reliable sources for bioactive natural product analogs, particularly those with demonstrated therapeutic potential such as flavonoids. Patent CN107746399B, published in early 2020, introduces a robust and efficient synthetic methodology for producing oxygen-substituted geranyl flavanones, specifically targeting the synthesis of the natural product known as flavanone B. This compound, originally isolated from the rhizomes of Millettia usaramensis, belongs to a class of flavonoids renowned for their broad spectrum of biological activities, including antioxidant, antimalarial, antibacterial, and antitumor properties. While traditional methods rely on the extraction of these compounds from plant sources like Spatholobus suberectus, such approaches are plagued by low natural abundance, seasonal variability, and exorbitant costs associated with isolation and purification. The disclosed invention addresses these critical supply chain bottlenecks by establishing a concise six-step chemical synthesis that utilizes simple, commercially available starting materials.
The limitations of conventional extraction methods are profound when viewed through the lens of modern supply chain management and drug development. Natural products, while biologically potent, often exist in plant matrices at trace levels, necessitating the processing of massive quantities of biomass to obtain gram-scale amounts of the active ingredient. This not only drives up the cost of goods significantly but also introduces variability in the impurity profile due to environmental factors affecting plant growth. Furthermore, the extraction process typically involves extensive use of organic solvents and complex chromatographic separations to remove structurally similar congeners, resulting in substantial environmental waste and extended lead times. For a reliable pharmaceutical intermediate supplier, relying on such unpredictable natural sources poses a significant risk to production continuity and quality control standards required by regulatory bodies.
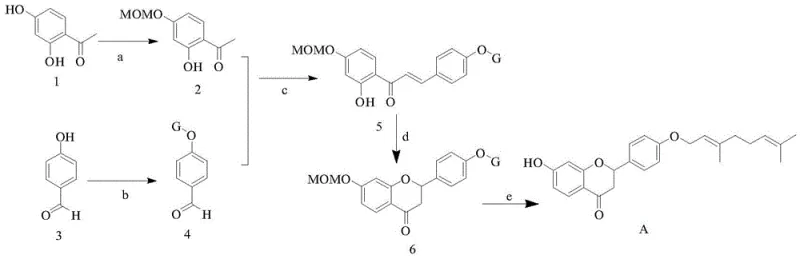
In stark contrast, the novel approach detailed in the patent leverages a rational design strategy that reconstructs the complex flavanone skeleton from basic aromatic building blocks. By employing 4-hydroxybenzaldehyde and 2,4-dihydroxyacetophenone as the foundational substrates, the synthesis bypasses the need for biological sourcing entirely. The route is designed with protecting group strategies, specifically utilizing the methoxymethyl (MOM) group, to ensure regioselectivity during the critical bond-forming steps. This methodological precision allows for the controlled introduction of the geranyl side chain, a lipophilic moiety essential for the compound's membrane permeability and biological activity. The result is a streamlined process that achieves a total yield of approximately 30%, a figure that represents a substantial improvement in material efficiency compared to the negligible recovery rates often seen in phytochemical extraction.
Mechanistic Insights into the Multi-Step Synthesis and Cyclization
The chemical logic underpinning this synthesis is rooted in classic heterocyclic chemistry, adapted for modern efficiency. The process initiates with the selective O-alkylation of 4-hydroxybenzaldehyde using geranyl bromide in the presence of potassium carbonate, effectively installing the terpenoid tail. Concurrently, the acetophenone partner undergoes protection of its phenolic hydroxyl groups using chloromethyl methyl ether (MOMCl) to prevent unwanted side reactions during the subsequent condensation. These two prepared fragments are then united via a Claisen-Schmidt condensation, catalyzed by potassium hydroxide in an ethanol-water system. This base-mediated aldol-type reaction forms the chalcone intermediate, establishing the carbon-carbon double bond that links the two aromatic rings. The reaction conditions are carefully controlled at 0°C initially to manage exothermicity before warming to room temperature, ensuring high conversion rates as evidenced by the reported step yields.
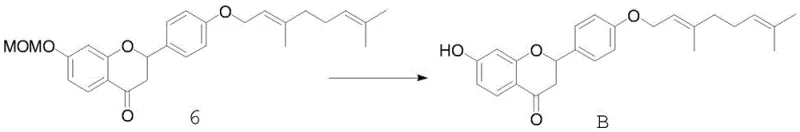
The transformation of the linear chalcone into the cyclic flavanone core represents the pivotal mechanistic step of the entire sequence. This intramolecular cyclization is achieved through a conjugate addition of the phenolic oxygen to the alpha,beta-unsaturated ketone system. In the patented procedure, this is facilitated by heating the chalcone in ethanol with sodium acetate, which acts as a mild base to promote the nucleophilic attack without causing degradation of the sensitive geranyl chain. Following the ring closure, the final step involves the removal of the MOM protecting groups under acidic conditions using hydrochloric acid in methanol. This deprotection reveals the free phenolic hydroxyl groups essential for the compound's antioxidant activity, yielding the final target molecule, flavanone B. The ability to execute these transformations with high fidelity minimizes the formation of regioisomers and byproducts, thereby simplifying downstream purification.
How to Synthesize Oxygen-Substituted Geranyl Flavanone Efficiently
Executing this synthesis requires strict adherence to the reaction parameters outlined in the patent to maximize yield and purity. The process is divided into distinct operational units: precursor preparation, coupling, cyclization, and final deprotection. Each stage utilizes standard laboratory equipment such as reflux condensers and rotary evaporators, making the transition from benchtop to pilot plant straightforward. The use of common solvents like acetone, ethanol, and ethyl acetate further enhances the practicality of the method. For detailed operational specifics regarding stoichiometry, temperature profiles, and workup procedures, please refer to the standardized guide below which encapsulates the critical process parameters.
- Preparation of geranyl ether intermediate via alkylation of 4-hydroxybenzaldehyde with geranyl bromide.
- Protection of 2,4-dihydroxyacetophenone followed by Claisen-Schmidt condensation to form the chalcone intermediate.
- Intramolecular cyclization to form the flavanone core, followed by acid-catalyzed deprotection to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from extraction to total synthesis offers transformative benefits in terms of cost stability and sourcing reliability. The reliance on commodity chemicals such as hydroxybenzaldehyde and acetophenone derivatives decouples production from agricultural cycles and geopolitical risks associated with raw plant material sourcing. This synthetic route ensures a consistent supply of high-purity material, which is critical for maintaining the quality standards required in cost reduction in API manufacturing. By eliminating the variability inherent in natural products, manufacturers can better forecast production schedules and reduce inventory buffers, leading to a leaner and more responsive supply chain.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the low cost of starting materials and the avoidance of expensive transition metal catalysts. Traditional cross-coupling methods often require palladium or other precious metals which necessitate costly removal steps to meet residual metal specifications. This metal-free synthetic route significantly lowers the cost of goods sold (COGS) by utilizing inexpensive inorganic bases and organic acids. Furthermore, the high step-wise yields reported in the patent minimize material loss, ensuring that the theoretical cost savings are realized in actual production runs.
- Enhanced Supply Chain Reliability: Sourcing natural extracts often involves long lead times due to harvesting seasons and complex logistics. In contrast, the chemical precursors for this synthesis are available from multiple global suppliers, creating a competitive market that drives down prices and ensures availability. This redundancy in the supply base mitigates the risk of shortages and allows for reducing lead time for high-purity intermediates. The robustness of the chemistry means that production can be ramped up quickly in response to market demand without the multi-year lead time required to cultivate new crop sources.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable to large-scale reactors. The absence of hazardous reagents and the use of relatively green solvents like ethanol and acetone simplify waste treatment and disposal. This aligns with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing sites. The ability to perform commercial scale-up of complex natural product analogs using such environmentally benign protocols positions this technology as a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of flavanone B. These answers are derived directly from the technical specifications and data provided in patent CN107746399B, offering clarity on the feasibility and advantages of this synthetic approach for potential partners and licensees.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: The synthetic method described in patent CN107746399B offers a total yield of approximately 30%, which is significantly more reliable than natural extraction where content is extremely limited. It eliminates the dependency on seasonal plant availability and complex isolation procedures.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard organic reactions such as reflux, extraction, and column chromatography which are well-established in industrial settings. The use of cheap, commodity raw materials like substituted acetophenone facilitates easy scale-up.
Q: What is the purity profile of the synthesized flavanone B?
A: The patent describes rigorous purification via silica gel column chromatography at multiple stages, ensuring high purity. The step-wise yields (ranging from 65% to 94%) indicate a robust process capable of producing high-quality intermediates suitable for further pharmaceutical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flavanone B Supplier
The synthetic methodology described in patent CN107746399B represents a significant advancement in the production of bioactive flavonoids, offering a viable path to high-purity flavanone derivatives that was previously inaccessible through extraction. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent literature into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your supply chain goals with efficiency and reliability.
