Advanced Multi-Enzyme Cascade Technology for Commercial Scale p-Coumaric Acid Manufacturing
The global demand for high-purity p-coumaric acid, a critical precursor for flavonoids, resveratrol, and various pharmaceutical intermediates, has necessitated a shift from traditional extraction methods to advanced biosynthetic pathways. Patent CN113265431A introduces a groundbreaking multi-enzyme cascade catalysis method that fundamentally alters the production landscape for this valuable fine chemical. By utilizing p-hydroxyphenylpyruvic acid as a starting substrate, this technology leverages a sophisticated coupling of carbonyl reductase and dehydratase to achieve superior conversion rates. Unlike conventional biosynthesis which often struggles with low titers and expensive cofactor requirements, this innovation integrates an in-situ coenzyme regeneration system that ensures sustained catalytic activity. For R&D directors and procurement specialists seeking a reliable p-coumaric acid supplier, this patent represents a pivotal advancement in process chemistry, offering a route that combines the specificity of enzymatic reactions with the robustness required for industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of p-coumaric acid has been plagued by significant inefficiencies inherent to plant extraction and chemical synthesis. Plant extraction, while natural, is constrained by long growth cycles, large collection radii, and notoriously low extraction efficiencies that cannot meet the surging demands of the nutraceutical and cosmetic industries. Chemical synthesis, on the other hand, often involves harsh reaction conditions, high energy consumption, and the generation of substantial environmental pollutants, making it increasingly untenable under modern regulatory frameworks. Even earlier biosynthetic attempts using whole-cell fermentation or single-enzyme systems have faced hurdles such as low substrate tolerance, with some reports indicating yields as low as 0.4 g/L or 1.9 g/L, which are commercially unviable. Furthermore, the reliance on expensive external cofactors like NAD(P)H in previous enzymatic methods created a prohibitive cost barrier, preventing the widespread adoption of biocatalysis for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The novel approach detailed in the patent data overcomes these historical bottlenecks through a meticulously engineered multi-enzyme cascade system that operates with remarkable efficiency. By employing a dual-reactor setup coupled with membrane separation, the process facilitates continuous production rather than being limited to batch constraints. The core innovation lies in the strategic pairing of carbonyl reductase from Candida parapsilosis with a dehydratase from Bacillus licheniformis, creating a seamless metabolic channel that minimizes intermediate accumulation. Crucially, the system addresses the cofactor cost issue by integrating glucose dehydrogenase to regenerate NAD(P)H in situ, effectively turning a stoichiometric expense into a catalytic cycle. This architectural shift not only enhances the overall yield but also simplifies the downstream processing, as the membrane units allow for the simultaneous recovery of enzymes and purification of the product stream, thereby streamlining the commercial scale-up of complex fine chemicals.
Mechanistic Insights into Multi-Enzyme Cascade Catalysis
The biochemical foundation of this process relies on a precise two-step transformation that maximizes atomic economy and stereo-selectivity. In the first stage, p-hydroxyphenylpyruvic acid undergoes a stereospecific reduction catalyzed by carbonyl reductase to form p-hydroxyphenyllactic acid. This step is energetically driven by the oxidation of NAD(P)H to NAD(P)+, which would typically halt the reaction if not for the concurrent action of glucose dehydrogenase. The glucose dehydrogenase oxidizes glucose to gluconic acid, simultaneously reducing NAD(P)+ back to NAD(P)H, thus maintaining a steady state of cofactor availability without external addition. Following this reduction, the intermediate flows into the second stage where dehydratase catalyzes the elimination of water to form the conjugated double bond characteristic of p-coumaric acid. This cascade design ensures that the reaction equilibrium is constantly pushed forward, preventing the accumulation of the hydroxy-acid intermediate and driving the conversion rate to exceed 80 percent.
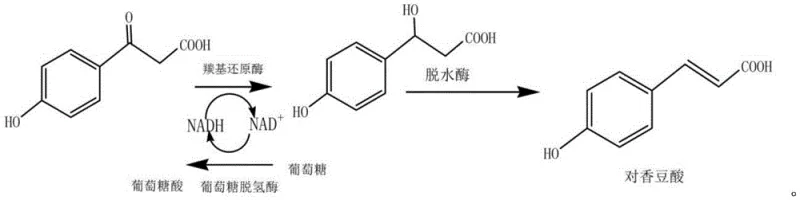
Beyond mere conversion, the mechanism provides exceptional control over the impurity profile, a critical parameter for R&D directors evaluating new supply chains. Enzymatic catalysis is inherently specific, meaning side reactions common in chemical synthesis, such as over-reduction or non-specific oxidation, are virtually eliminated. The use of ultrafiltration membranes with specific molecular weight cutoffs (5-20 kD for the first stage and 30-50 kD for the second) acts as a physical barrier that retains the high-molecular-weight enzymes while allowing the low-molecular-weight product and substrates to pass. This selective permeability ensures that the enzyme concentration within the reactor remains high, sustaining reaction velocity, while the product is continuously removed, minimizing potential degradation or reverse reactions. The result is a product purity that consistently reaches above 99%, meeting the stringent specifications required for high-purity p-coumaric acid applications in sensitive pharmaceutical and food formulations.
How to Synthesize p-Coumaric Acid Efficiently
Implementing this synthesis route requires a disciplined approach to reactor configuration and enzyme management to fully realize the theoretical benefits described in the patent literature. The process is designed to be flexible, accommodating both intermittent batch reactors for smaller campaigns and tubular reactor-membrane separation coupling devices for continuous large-scale operations. Operators must carefully monitor the pH and temperature gradients across the two stages, maintaining the first stage between 20-40°C and pH 6-9, and the second stage between 15-35°C and pH 7-10 to optimize enzyme stability. The detailed standardized synthesis steps, including specific enzyme loading rates, membrane flux parameters, and crystallization protocols, are essential for replicating the reported production efficiency of up to 50 g/L.h.
- Conduct first-stage tubular reaction with p-hydroxyphenylpyruvic acid, glucose, carbonyl reductase, and glucose dehydrogenase at 20-40°C and pH 6-9.
- Perform ultrafiltration using 5-20 kD membrane to recover carbonyl reductase and glucose dehydrogenase for recycling.
- Pass permeate to second-stage reactor, add dehydratase at 15-35°C and pH 7-10 to convert intermediate to p-coumaric acid.
- Execute second-stage ultrafiltration with 30-50 kD membrane to recover dehydratase, then crystallize product from permeate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic cascade technology offers profound strategic advantages that extend beyond simple unit cost calculations. The primary value driver is the drastic simplification of the supply chain for raw materials; by replacing expensive stoichiometric reagents and transition metal catalysts with recyclable biocatalysts and glucose, the process insulates manufacturers from volatile petrochemical pricing. The ability to recycle enzymes more than 5 times means that the effective cost of the biocatalyst per kilogram of product is significantly reduced, contributing to substantial cost savings over the lifecycle of the production campaign. Furthermore, the mild reaction conditions eliminate the need for high-pressure or high-temperature equipment, reducing capital expenditure on specialized reactors and lowering energy consumption profiles, which aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of costly external cofactors and the high reusability of the enzyme preparations. By regenerating NAD(P)H in situ using inexpensive glucose, the process removes a major cost center associated with traditional biocatalysis. Additionally, the membrane-based enzyme recovery system allows the same batch of carbonyl reductase, glucose dehydrogenase, and dehydratase to be utilized repeatedly, which drastically lowers the variable cost of goods sold. This efficiency translates into a more competitive pricing structure for the final API intermediate, allowing downstream partners to improve their margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the continuous production mode enabled by the tubular reactor design. Unlike plant extraction, which is subject to seasonal harvest cycles and agricultural variability, this biosynthetic method can operate year-round with consistent output levels. The use of recombinant enzymes produced in E. coli ensures a stable and scalable source of biocatalysts, removing the dependency on rare natural sources. This reliability is crucial for reducing lead time for high-purity nutritional ingredients, ensuring that pharmaceutical and cosmetic manufacturers can maintain their own production schedules without interruption due to raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with clear parameters for moving from laboratory benchtop to 100 MT annual commercial production. The aqueous-based reaction systems and the absence of heavy metal catalysts simplify wastewater treatment and reduce the environmental footprint of the manufacturing facility. The membrane separation units minimize solvent usage compared to traditional extraction methods, and the high selectivity of the enzymes reduces the formation of hazardous byproducts. This alignment with green chemistry principles facilitates easier regulatory approval and enhances the brand value of the final product as sustainably sourced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this multi-enzyme cascade technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this synthetic route into their existing supply chains.
Q: How does the multi-enzyme cascade improve yield compared to traditional extraction?
A: Traditional plant extraction suffers from low efficiency and seasonal limitations. This patented cascade method achieves conversion rates over 80% with production efficiency reaching up to 50 g/L.h, significantly outperforming natural extraction yields.
Q: What mechanisms ensure cost reduction in this enzymatic process?
A: Cost is minimized through in-situ regeneration of the expensive NAD(P)H coenzyme using glucose dehydrogenase. Additionally, the ultrafiltration system allows enzymes to be recycled more than 5 times, drastically reducing biocatalyst consumption.
Q: Is this process suitable for large-scale continuous manufacturing?
A: Yes, the technology is designed for both batch and continuous production using tubular reactor-membrane separation coupling devices. This architecture supports scalable operations from pilot studies to 100 MT annual commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Coumaric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced biosynthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of patent CN113265431A are realized in practical, large-volume outputs. Our facilities are equipped with state-of-the-art bioreactors and membrane separation units capable of handling the specific requirements of multi-enzyme cascades, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical and food-grade intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this enzymatic route can optimize your bill of materials. Please contact us to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your upcoming projects, ensuring a secure and efficient supply of high-quality p-coumaric acid for your global operations.
