Advanced Synthesis of Substituted Biphenyl HMG-CoA Reductase Inhibitor Intermediates for Commercial Scale-Up
Introduction to Novel Substituted Biphenyl Therapeutics
The landscape of cardiovascular medicine has been profoundly shaped by the discovery of HMG-CoA reductase inhibitors, commonly known as statins. Patent CN1045769A introduces a groundbreaking class of substituted biphenyl compounds that exhibit superior inhibitory activity against this critical enzyme. Unlike earlier iterations derived from fungal cultures or complex heterocyclic systems, these novel structures offer a robust synthetic pathway that is highly amenable to industrial optimization. The core innovation lies in the versatile substitution pattern allowed on the biphenyl scaffold, where groups A, B, D, and E can be independently modified to fine-tune pharmacokinetic properties without compromising biological efficacy. This flexibility is crucial for developing next-generation therapeutics with improved metabolic stability and reduced side effect profiles.
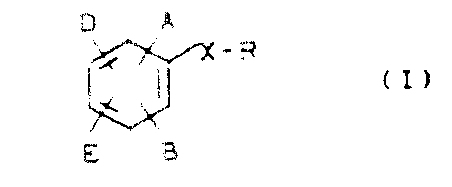
For pharmaceutical developers, the significance of this patent extends beyond mere molecular novelty; it represents a strategic opportunity to secure supply chains for high-purity active pharmaceutical ingredients (APIs). The described compounds are not limited to a single rigid structure but encompass a broad genus of molecules defined by specific stereochemical configurations, particularly the erythro-isomers and trans-lactones which demonstrate the highest potency. By leveraging the synthetic methodologies outlined in this intellectual property, manufacturers can produce reliable pharmaceutical intermediates that meet the stringent quality standards required for global regulatory submission. The ability to access these molecules through a scalable, non-fermentative route marks a significant departure from traditional production methods, offering greater control over impurity profiles and batch-to-batch consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of HMG-CoA reductase inhibitors relied heavily on fermentation processes or semi-synthetic modifications of natural products like mevinolin. While effective, these biological routes are inherently constrained by the metabolic capabilities of the host organisms, often resulting in complex mixtures of analogs that are difficult and costly to separate. Furthermore, fermentation processes are susceptible to contamination and yield fluctuations, creating significant supply chain vulnerabilities for procurement managers. Chemical syntheses that did exist often involved harsh conditions, multiple protection-deprotection steps, and the use of expensive chiral auxiliaries that drove up the cost of goods significantly. The reliance on transition metal catalysts in earlier synthetic routes also introduced the risk of heavy metal contamination, necessitating additional purification steps that further eroded overall process efficiency and increased waste generation.
The Novel Approach
The methodology presented in CN1045769A circumvents these bottlenecks by employing a convergent synthetic strategy centered on the construction of the biphenyl core followed by precise side-chain elaboration. Instead of relying on unpredictable biological systems, this approach utilizes well-defined organic transformations such as palladium-catalyzed coupling and stereoselective hydride reductions. The process allows for the introduction of diverse functional groups—such as halogens, alkyl chains, and alkoxy moieties—at specific positions on the aromatic rings, enabling medicinal chemists to optimize the molecule's interaction with the enzyme active site. This chemical precision ensures that the final product is obtained with high regioselectivity and stereochemical purity, drastically reducing the burden on downstream purification teams. By shifting from extraction-based methods to total synthesis, manufacturers gain full autonomy over production schedules and capacity, ensuring a stable supply of high-purity pharmaceutical intermediates regardless of external biological factors.
Mechanistic Insights into Stereoselective Reduction and Lactonization
At the heart of this synthetic innovation is a highly controlled reduction step that dictates the stereochemical outcome of the molecule. The patent details the conversion of a beta-keto ester intermediate into the corresponding dihydroxy ester, a transformation that creates two new chiral centers. The preferred method utilizes sodium borohydride in the presence of triethylboron within an inert solvent system such as tetrahydrofuran or diethyl ether. This specific reagent combination is critical because it facilitates a chelation-controlled reduction mechanism that favors the formation of the erythro-diastereomer over the threo-isomer. The reaction is typically conducted at low temperatures, ranging from -80°C to +30°C, with an optimal window between -78°C and 0°C to maximize stereoselectivity. This level of control is essential because the biological activity of the final drug is heavily dependent on the spatial arrangement of the hydroxyl groups, with the 3R,5S configuration often exhibiting superior potency.
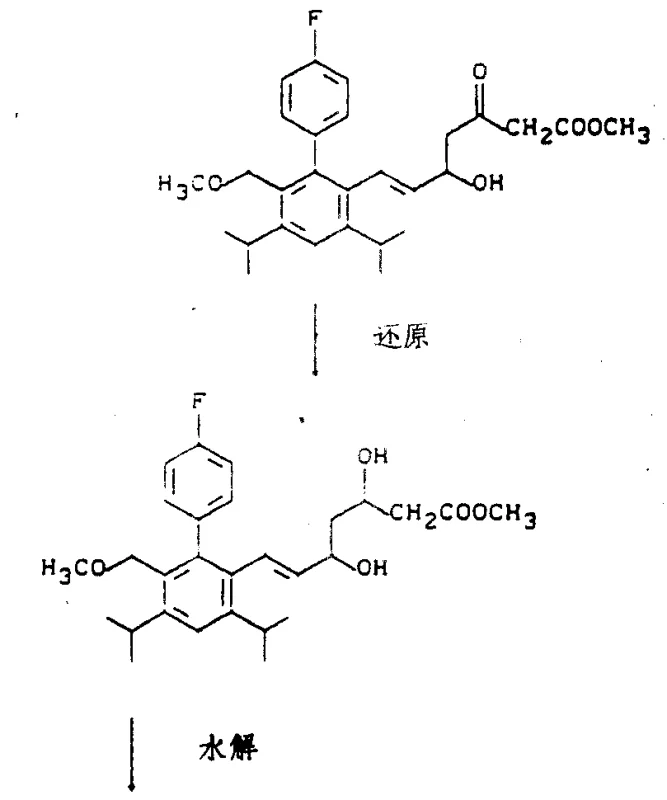
Following the reduction, the pathway proceeds through hydrolysis and cyclization to form the pharmacologically active lactone ring. The hydrolysis step converts the ester functionality into a carboxylic acid salt using common inorganic bases like sodium hydroxide or potassium hydroxide in alcoholic solvents. Subsequent acidification yields the free acid, which then undergoes intramolecular esterification to close the lactone ring. This cyclization can be driven thermally in hydrocarbon solvents like toluene, often in the presence of molecular sieves to remove water and shift the equilibrium towards product formation. Alternatively, dehydrating agents such as carbodiimides can be employed to facilitate ring closure under milder conditions. The entire sequence is designed to preserve the delicate stereochemistry established in the earlier reduction step, ensuring that the final trans-lactone product retains the necessary 4R,6S configuration required for effective enzyme inhibition. This mechanistic robustness makes the process highly suitable for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Erythro-Substituted Biphenyl Intermediates Efficiently
The practical execution of this synthesis begins with the preparation of the key aldehyde precursor via palladium-catalyzed coupling, followed by condensation with an acetoacetate derivative. 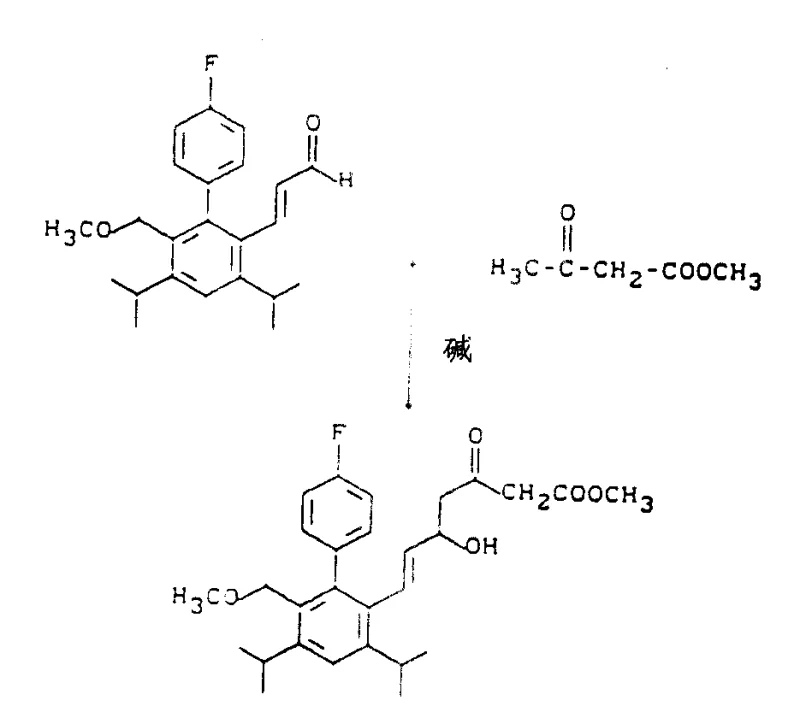
- Condensation of substituted biphenyl aldehydes with acetoacetates using strong bases like n-butyllithium or sodium hydride to form beta-keto esters.
- Stereoselective reduction of the ketone moiety using sodium borohydride in the presence of triethylboron at low temperatures (-78°C to 0°C) to achieve erythro configuration.
- Hydrolysis of the ester group followed by acid-catalyzed cyclization to form the desired trans-lactone ring system.
The subsequent reduction of the ketone group is the pivotal step where stereochemical fidelity is established. Detailed operational parameters, including specific reagent stoichiometry and temperature gradients, are critical for achieving the desired erythro-selectivity. For a comprehensive guide on executing these transformations with maximum yield and purity, please refer to the standardized protocol below.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial advantages in terms of cost structure and supply reliability. By eliminating the dependency on fermentation, manufacturers can decouple production from biological variables, leading to more predictable lead times and consistent output volumes. The use of commodity chemicals such as sodium borohydride, simple aromatic aldehydes, and acetoacetates means that raw material costs are significantly lower compared to processes requiring specialized chiral pool starting materials. Furthermore, the avoidance of precious metal catalysts in the final reduction and cyclization steps removes the need for expensive metal scavenging operations, thereby simplifying the purification workflow and reducing waste disposal costs. These factors combine to create a manufacturing process that is not only economically efficient but also environmentally sustainable, aligning with modern green chemistry principles.
- Cost Reduction in Manufacturing: The streamlined synthetic sequence minimizes the number of unit operations required to reach the final intermediate. By utilizing robust reagents like triethylboron-modified borohydrides, the process achieves high conversion rates without the need for exotic catalysts. This directly translates to lower operational expenditures and a reduced cost of goods sold, allowing for more competitive pricing in the global market. Additionally, the ability to recycle solvents such as toluene and tetrahydrofuran further enhances the economic viability of the process.
- Enhanced Supply Chain Reliability: Because the starting materials are widely available petrochemical derivatives, the risk of supply disruption is minimal. Unlike natural product extraction which is subject to seasonal and agricultural variations, this totally synthetic route can be operated year-round in standard chemical reactors. This reliability is crucial for maintaining continuous API production schedules and meeting the demands of large-scale clinical trials or commercial launches without interruption.
- Scalability and Environmental Compliance: The reaction conditions described, such as atmospheric pressure and moderate temperatures, are easily transferable from laboratory to pilot and production scales. The process generates manageable waste streams that can be treated using standard effluent protocols, ensuring compliance with increasingly strict environmental regulations. The high atom economy of the coupling and reduction steps also contributes to a lower overall environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding these nuances is vital for process engineers and quality assurance teams tasked with technology transfer.
Q: What is the primary therapeutic application of these substituted biphenyl compounds?
A: These compounds act as potent inhibitors of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase, making them effective candidates for treating hyperlipoproteinemia, atherosclerosis, and lowering blood cholesterol levels.
Q: How is stereochemical control achieved during the synthesis?
A: High stereoselectivity for the erythro-configuration is achieved by performing the ketone reduction with sodium borohydride in the presence of triethylboron at controlled low temperatures, typically between -78°C and 0°C.
Q: Are the starting materials for this synthesis commercially viable?
A: Yes, the process utilizes readily available substituted benzene derivatives and standard reagents like acetoacetates and metal hydrides, avoiding the need for rare or prohibitively expensive transition metal catalysts in the final steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of substituted biphenyl intermediate meets the highest industry standards. Our commitment to excellence extends beyond mere manufacturing; we act as a strategic partner, offering technical support to optimize your specific process requirements.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume. Whether you require specific COA data for regulatory filings or detailed route feasibility assessments for process optimization, our experts are ready to provide the data-driven insights you need. Partner with us to secure a stable, cost-effective supply of these critical HMG-CoA inhibitor precursors and accelerate your path to market.
