Advanced Green Nitration Technology for High-Purity 1,8-Dinitro-3,6-Naphthalenedisulfonic Acid Production
The global demand for high-purity naphthalene derivatives continues to surge, driven by the expanding applications in advanced dye synthesis and specialty agrochemical formulations. A pivotal breakthrough in this sector is documented in Chinese patent CN102936213A, which introduces a revolutionary clean preparation method for 1,8-dinitro-3,6-naphthalenedisulfonic acid. This intermediate is critical for the production of H-acid, a cornerstone molecule in the azo dye industry. Traditionally, the synthesis of such nitrated naphthalene sulfonic acids has been plagued by severe environmental challenges, particularly the generation of massive quantities of high-concentration spent acid that are difficult and costly to treat. The innovation presented in this patent fundamentally alters the economic and ecological landscape of this manufacturing process by implementing a closed-loop system for sulfuric acid recovery.
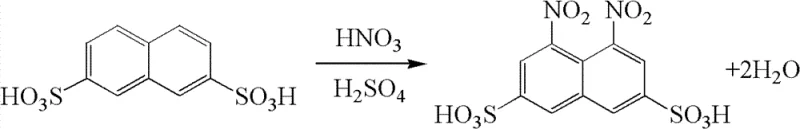
By integrating a specialized flocculation treatment step prior to acid concentration, the technology effectively removes organic impurities that typically degrade recycled solvents. This allows the nitration mother liquor to be processed and reused indefinitely, achieving a near-zero wastewater discharge status for the nitration stage. For R&D directors and process engineers, this represents a significant leap forward in sustainable chemistry, offering a robust pathway to produce high-purity dye intermediates while drastically reducing the environmental footprint associated with traditional nitration workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of H-acid and its precursors relied heavily on the alkali fusion of 1-naphthylamine-3,6,8-trisulfonic acid, a process notorious for its harsh reaction conditions and poor atom economy. Even newer nitration-based routes, such as those described in earlier Japanese patents, introduced significant improvements in yield but failed to address the critical bottleneck of waste management. In conventional nitration processes using sulfuric acid as a solvent, the reaction generates a large volume of spent acid contaminated with oxidized organic by-products and residual nitro compounds. Disposing of this high-COD (Chemical Oxygen Demand) waste acid is not only environmentally hazardous but also incurs substantial operational costs due to the need for neutralization and specialized treatment facilities. Furthermore, the inability to recycle the sulfuric acid efficiently leads to a continuous consumption of fresh reagents, inflating the raw material costs and complicating the supply chain logistics for bulk chemical manufacturers.
The Novel Approach
The methodology outlined in patent CN102936213A disrupts this status quo by introducing a sophisticated post-reaction purification strategy that enables the circular economy of reagents. Instead of discarding the nitration mother liquor, the process employs a targeted flocculation step using polyacrylamide to aggregate and remove dissolved organic impurities. This crucial intervention lowers the COD value of the mother liquor from approximately 40,000-50,000 mg/L to below 4,000 mg/L, rendering the acid suitable for recovery. Following filtration, the clarified acid is concentrated under reduced pressure to restore its strength to ≥93%, making it fully viable for reuse in subsequent nitration batches. This approach not only eliminates the discharge of nitration wastewater but also stabilizes the reaction environment, ensuring consistent product quality and yield over multiple cycles without the accumulation of deleterious side products.
Mechanistic Insights into Polyacrylamide-Assisted Nitration and Acid Recovery
The core chemical transformation involves the electrophilic aromatic nitration of 2,7-naphthalenedisulfonic acid using 98% fuming nitric acid in a sulfuric acid medium. The reaction proceeds through the formation of the nitronium ion (NO2+), which attacks the electron-rich positions on the naphthalene ring, specifically targeting the 1 and 8 positions due to the directing effects of the existing sulfonic acid groups. Maintaining the temperature between 0°C and 70°C is critical to control the reaction kinetics and prevent over-oxidation or sulfonation side reactions. The use of fuming nitric acid ensures a high concentration of the nitrating agent, driving the reaction to completion with high conversion rates. However, the true mechanistic innovation lies not in the nitration itself, but in the subsequent handling of the reaction matrix, where the stability of the sulfonic acid groups under strong acidic conditions is leveraged to facilitate separation.
The separation mechanism relies on the unique behavior of polyacrylamide flocculants in strong acidic environments, a condition where most conventional flocculants fail. The patent specifies the use of non-ionic or cationic polyacrylamide with a molecular weight between 8 million and 10 million. These long-chain polymers function by bridging suspended organic particles and colloidal impurities formed during the nitration and air-blowing denitration steps. Upon addition to the mother liquor at controlled temperatures (10-40°C) and stirring speeds, the flocculant induces the aggregation of these impurities into larger settleable flocs. This physical separation is essential because it prevents the buildup of tarry substances in the recycled acid, which would otherwise catalyze decomposition reactions or discolor the final product. By effectively scrubbing the acid of organic contaminants, the process maintains the integrity of the solvent for repeated use.
How to Synthesize 1,8-Dinitro-3,6-Naphthalenedisulfonic Acid Efficiently
The synthesis protocol described in the patent offers a streamlined workflow that balances reaction efficiency with environmental stewardship. The process begins with the dissolution of the starting material, 2,7-naphthalenedisulfonic acid, in sulfuric acid, followed by the controlled addition of the nitrating agent. Critical parameters such as the molar ratio of sulfuric acid to substrate (5-20 times) and the dropping time of nitric acid (1-2 hours) must be strictly adhered to ensure optimal heat dissipation and reaction homogeneity. After the reaction reaches completion, indicated by the disappearance of starting materials via liquid chromatography, the product is precipitated by dilution with water. The detailed standardized synthesis steps, including specific dosages for flocculation and vacuum concentration parameters, are provided in the guide below to assist technical teams in replicating this high-efficiency route.
- Dissolve 2,7-naphthalenedisulfonic acid in sulfuric acid and add 98% fuming nitric acid dropwise at 0-70°C.
- Separate the product by water dilution and filter the precipitate.
- Treat the remaining mother liquor with polyacrylamide flocculant, filter impurities, and concentrate under reduced pressure to recover sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this clean synthesis technology translates into tangible strategic advantages beyond mere regulatory compliance. The ability to recycle sulfuric acid internally creates a closed-loop system that significantly insulates the manufacturing process from fluctuations in raw material pricing and availability. By eliminating the need for external waste acid treatment services, the facility reduces its dependency on third-party disposal vendors, thereby mitigating supply chain risks associated with environmental logistics. Furthermore, the simplified workflow reduces the overall complexity of the production line, allowing for more predictable scheduling and faster turnaround times for large-scale orders of this critical dye intermediate.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic reduction in sulfuric acid consumption. Since the solvent is recovered and reused with high efficiency, the recurring cost of purchasing fresh concentrated acid is minimized. Additionally, the elimination of wastewater treatment fees and the associated energy costs for neutralization lead to substantial operational savings. The high yield of over 90% ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final active pharmaceutical or dye intermediate. This economic efficiency makes the process highly competitive in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The robustness of the flocculation and recovery system ensures consistent production capacity without the interruptions often caused by waste storage limits or treatment bottlenecks. Because the process generates negligible liquid waste, the facility is less vulnerable to environmental shutdowns or regulatory penalties that can disrupt supply continuity. The use of commercially available polyacrylamide flocculants ensures that the auxiliary materials are easily sourced, preventing supply chain fragility. This reliability is crucial for downstream customers who require steady volumes of high-purity intermediates for their own continuous manufacturing operations.
- Scalability and Environmental Compliance: The technology is inherently scalable, as demonstrated by the patent's examples which cover significant batch sizes suitable for industrial translation. The clear reduction in COD levels and the absence of wastewater discharge align perfectly with increasingly stringent global environmental regulations, such as REACH in Europe and various EPA standards. This compliance future-proofs the manufacturing asset, allowing for seamless expansion into new markets without the need for costly retrofits to waste treatment infrastructure. The clean nature of the process also enhances the corporate sustainability profile, appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean nitration technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production of naphthalene-based intermediates.
Q: How does this process solve the waste acid problem in traditional nitration?
A: The process utilizes polyacrylamide flocculation to remove organic impurities from the nitration mother liquor, allowing the sulfuric acid to be concentrated and reused without generating wastewater.
Q: What is the purity and yield of the final product?
A: The method consistently achieves a product purity of over 98% and a yield exceeding 90%, meeting the stringent requirements for subsequent hydrogenation to H-acid.
Q: Can the recovered sulfuric acid be reused directly?
A: Yes, after flocculation and vacuum concentration, the recovered sulfuric acid reaches a concentration of ≥93% and is suitable for use as a solvent in the next batch of nitration reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,8-Dinitro-3,6-Naphthalenedisulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of sustainable and efficient manufacturing pathways for complex organic intermediates like 1,8-dinitro-3,6-naphthalenedisulfonic acid. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102936213A are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process control systems to maintain stringent purity specifications, consistently delivering products with purity levels exceeding 98% to meet the exacting demands of the global dye and agrochemical industries.
We invite potential partners to engage with our technical procurement team to discuss how this innovative clean synthesis route can be tailored to your specific supply chain requirements. By leveraging our expertise in process optimization and waste minimization, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your organization. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your sourcing strategy is built on a foundation of technical excellence and commercial viability.
