Optimizing Radiopharmaceutical Precursor Production via Direct Thymidine Functionalization
The landscape of radiopharmaceutical manufacturing is constantly evolving, driven by the need for high-purity precursors that can be produced efficiently and cost-effectively. A pivotal development in this sector is detailed in Chinese Patent CN101481399A, which discloses a novel method for synthesizing 5'-O-(4,4'-dimethoxytrityl)-2,3'-anhydrothymidine. This compound serves as a critical labeled precursor for the production of 3'-deoxy-3'-[18F]fluorothymidine ([18F]FLT), a widely used positron emission tomography (PET) imaging agent for detecting tumor proliferative activity. The patent outlines a strategic departure from traditional synthetic routes by utilizing thymidine directly as the starting material, rather than relying on expensive pre-protected derivatives. This technical breakthrough addresses long-standing inefficiencies in the supply chain of nuclear medicine intermediates, offering a pathway that combines operational simplicity with enhanced economic viability for global pharmaceutical manufacturers seeking reliable sources of complex nucleoside analogs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of anhydrothymidine derivatives was plagued by significant economic and operational bottlenecks. As illustrated in the background art, conventional pathways often relied on 5'-O-(4,4'-dimethoxytrityl)-thymidine as the starting substrate. This material is substantially more expensive than unprotected thymidine due to the additional processing required to manufacture it. Furthermore, established protocols, such as those cited in Documents [4] and [5], necessitated prolonged reaction times for the crucial ring-closure step, often requiring reflux conditions for up to three days. Such extended thermal exposure not only consumes excessive energy but also increases the risk of degradation for sensitive nucleoside structures. Other methods, like that in Document [3], attempted improvements but still suffered from complicated operational procedures and the persistent reliance on costly protected starting materials, creating a barrier to cost reduction in radiopharmaceutical intermediate manufacturing.
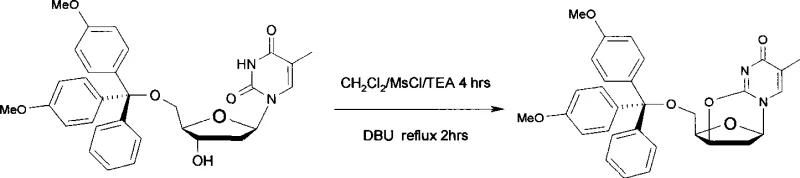
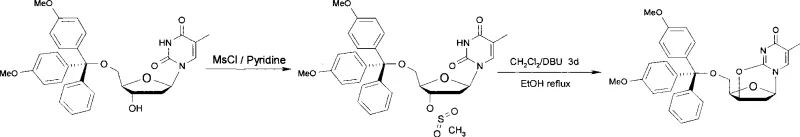
The Novel Approach
The methodology presented in CN101481399A fundamentally restructures the synthetic logic to overcome these historical limitations. By initiating the sequence with inexpensive thymidine, the process eliminates the markup associated with purchasing pre-protected intermediates. The innovation lies in a telescoped or semi-telescoped strategy where the 5'-hydroxyl group is protected with 4,4'-dimethoxytrityl chloride, followed immediately by activation of the 3'-hydroxyl group using methanesulfonyl chloride. Crucially, the subsequent cyclization is achieved under mild alkaline aqueous conditions rather than harsh anhydrous reflux. This shift allows the ring-closure reaction to proceed rapidly, typically within 10 to 60 minutes, drastically reducing the overall production cycle time. The result is a robust, high-yield process that delivers the target anhydrothymidine derivative with purity exceeding 98%, validating its suitability for the stringent quality requirements of the nuclear medicine industry.
Mechanistic Insights into Alkaline-Promoted Intramolecular Cyclization
The core chemical transformation in this patent involves a nucleophilic substitution reaction facilitated by a unique biphasic system. Initially, the 5'-hydroxyl of thymidine is selectively protected by the bulky 4,4'-dimethoxytrityl (DMTr) group in pyridine, leveraging the steric hindrance to favor the primary alcohol. Subsequently, the secondary 3'-hydroxyl group is activated by conversion into a mesylate ester using methanesulfonyl chloride. The mechanistic brilliance of this patent emerges in the final cyclization step. Unlike traditional methods that might employ strong anhydrous bases like DBU in organic solvents for extended periods, this method utilizes an alkaline aqueous solution (such as sodium hydroxide, potassium hydroxide, or sodium carbonate) in the presence of a water-immiscible organic solvent like dichloromethane or ethyl acetate. The hydroxide ions act as a base to deprotonate the N3 position or facilitate the direct attack of the 3'-oxygen (depending on the specific tautomeric state), promoting an intramolecular SN2 displacement of the mesylate group to form the 2,3'-anhydro bridge. This aqueous-organic interface likely helps in scavenging the acidic byproducts (methanesulfonic acid) efficiently, driving the equilibrium forward without the need for prolonged heating.
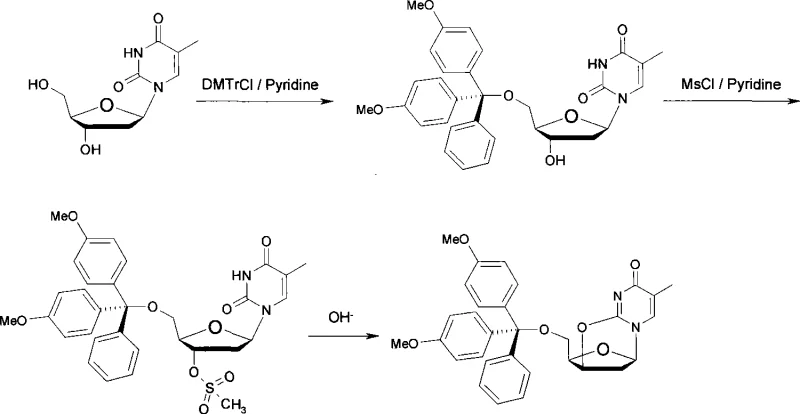
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction temperature (0-35°C) during the activation and cyclization phases minimizes the formation of thermal degradation products that are common in refluxing conditions. Furthermore, the use of aqueous workup allows for the effective removal of inorganic salts and polar byproducts, simplifying the downstream purification process. The patent data indicates that silica gel column chromatography yields a white solid with high purity, suggesting that the reaction profile is clean with minimal side reactions such as hydrolysis of the DMTr group or opening of the anhydro ring. This level of control is essential for a reliable radiopharmaceutical intermediate supplier, as impurities in the precursor can significantly affect the radiochemical yield and specific activity of the final [18F]FLT imaging agent.
How to Synthesize 5'-O-(4,4'-dimethoxytrityl)-2,3'-anhydrothymidine Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins by dissolving thymidine in anhydrous pyridine and reacting it with 4,4'-dimethoxytrityl chloride at room temperature. Without isolating the intermediate, methanesulfonyl chloride is added to the mixture to effect the 3'-mesylation. Following concentration, the residue is subjected to the critical cyclization step using an alkaline solution. For a comprehensive understanding of the specific molar ratios, solvent volumes, and precise workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Protect thymidine with 4,4'-dimethoxytrityl chloride in pyridine at 10-35°C for 1-4 hours.
- React the intermediate with methanesulfonyl chloride at 0-35°C for 1-4 hours to form the mesylate.
- Perform ring-closure by stirring the residue in an organic solvent with alkaline aqueous solution for 10-60 minutes.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits beyond mere technical feasibility. The shift from expensive protected starting materials to commodity-grade thymidine represents a fundamental restructuring of the cost of goods sold (COGS). By eliminating the need to source or synthesize 5'-O-DMTr-thymidine externally, manufacturers can capture the margin associated with that protection step internally. Additionally, the drastic reduction in reaction time—from days to less than an hour for the cyclization—means that reactor occupancy time is minimized, allowing for higher throughput in existing facilities without the need for capital expenditure on new equipment. This efficiency gain is critical for maintaining supply continuity in the fast-paced radiopharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the raw material selection. Thymidine is a widely available, bulk commodity chemical, whereas its DMTr-protected counterpart is a specialized fine chemical with a significantly higher price point. By performing the protection in-house as the first step of the sequence, the overall material cost is drastically simplified and reduced. Furthermore, the elimination of long reflux periods reduces utility costs associated with heating and cooling, contributing to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: Relying on complex, multi-day synthesis routes introduces significant risk of batch failure and delays. The robustness of this new method, characterized by mild conditions and short reaction times, enhances the predictability of production schedules. Since the starting material thymidine is sourced from a broad global supply base, the risk of raw material shortage is mitigated compared to relying on niche protected intermediates. This reliability ensures that downstream customers producing [18F]FLT can maintain their clinical and commercial supply chains without interruption, a key factor for any reliable radiopharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple operational parameters. The use of common solvents like dichloromethane and ethyl acetate, along with standard inorganic bases, facilitates easy technology transfer from the lab to commercial scale. Moreover, the shorter reaction times and higher yields mean less waste generation per kilogram of product. The ability to achieve high purity (>98%) through standard chromatographic techniques implies that the process generates a cleaner crude profile, reducing the burden on waste treatment systems and aligning with modern environmental compliance standards for the commercial scale-up of complex nucleoside derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on how this method compares to legacy processes in terms of yield, purity, and operational complexity.
Q: What is the primary cost advantage of this synthesis method?
A: The method utilizes thymidine directly as the starting material, bypassing the need for expensive pre-protected 5'-O-(4,4'-dimethoxytrityl)-thymidine, significantly reducing raw material costs.
Q: How does the reaction time compare to conventional cyclization methods?
A: The novel alkaline aqueous cyclization completes in 10 to 60 minutes, whereas traditional methods described in prior art often require refluxing for up to three days.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process features simple operation, mild reaction temperatures (0-35°C), and high yields (>60%), making it highly scalable for commercial production of radiopharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5'-O-(4,4'-dimethoxytrityl)-2,3'-anhydrothymidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality precursors play in the success of radiopharmaceutical development. Our technical team has extensively analyzed the methodology described in CN101481399A and possesses the expertise to implement this efficient route at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 5'-O-(4,4'-dimethoxytrityl)-2,3'-anhydrothymidine meets the exacting standards required for PET imaging agent synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of nuclear medicine.
