Scalable Nickel-Catalyzed Synthesis of Biphenyl Sulfide Compounds for High-Efficiency Acaricides
Scalable Nickel-Catalyzed Synthesis of Biphenyl Sulfide Compounds for High-Efficiency Acaricides
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance high purity with economic viability, particularly for complex sulfur-containing scaffolds. A pivotal advancement in this domain is detailed in patent CN111100048B, which discloses a novel preparation method for diphenyl sulfide compounds represented by general formula (I). These compounds serve as potent acaricides, exhibiting excellent activity against pests such as Tetranychus cinnabarinus. The core innovation lies in replacing traditional precious metal catalysis with a cost-effective nickel-based system, fundamentally altering the economic landscape of producing these high-value intermediates. This technical breakthrough not only addresses the scarcity and cost issues associated with palladium but also streamlines the supply chain by utilizing more abundant halogenated benzene starting materials. For R&D directors and procurement managers alike, understanding this shift is critical for optimizing future manufacturing strategies.
The strategic implementation of this technology offers a reliable agrochemical intermediate supplier pathway to secure long-term production stability. By leveraging the specific reaction conditions outlined in the patent, manufacturers can achieve consistent quality while mitigating the volatility of precious metal markets. The following analysis delves into the mechanistic advantages and commercial implications of this proprietary synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenyl thioether compounds relied heavily on palladium-catalyzed cross-coupling reactions, often utilizing aryl iodides and diboron acid pinacol esters as key substrates. As illustrated in prior art references like CN105541682A, these conventional methods impose significant financial and logistical burdens on the supply chain. The reliance on palladium, a precious metal with fluctuating global prices, introduces substantial cost variability that is difficult to hedge against in long-term contracts. Furthermore, the requirement for aryl iodides, which are generally more expensive and less stable than their chloro- or bromo- counterparts, exacerbates the raw material expenditure. From a process safety and environmental perspective, the removal of trace palladium residues from the final API or intermediate often necessitates additional purification steps, such as specialized scavenging resins or extensive recrystallization, which lowers overall throughput and increases waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN111100048B introduces a paradigm shift by employing a nickel compound catalytic system combined with metal zinc and organic phosphine ligands. This approach allows for the use of significantly cheaper chloro- or bromo-benzene derivatives as starting materials, effectively decoupling production costs from the volatile iodine and palladium markets. The reaction conditions are optimized to proceed efficiently at moderate temperatures, typically ranging from 20°C to 100°C, ensuring energy efficiency without compromising reaction kinetics. Moreover, the subsequent steps involving sulfonylation, reduction, and etherification are designed for high yield and operational simplicity. This holistic process redesign results in a streamlined workflow that minimizes unit operations and maximizes atom economy, positioning it as a superior choice for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
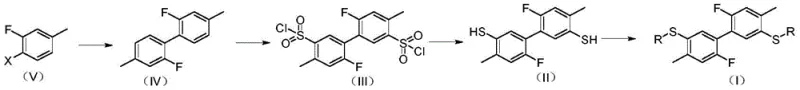
The heart of this synthetic innovation is the nickel-catalyzed reductive coupling of halogenated benzenes. In this mechanism, a nickel species, such as nickel chloride or bis(triphenylphosphine)nickel chloride, acts as the central catalyst, facilitated by triphenylphosphine ligands that stabilize the active metal center. The presence of metal zinc serves as a stoichiometric reductant, regenerating the active low-valent nickel species necessary for the oxidative addition into the carbon-halogen bond. Crucially, the addition of activating additives like sodium bromide or potassium iodide enhances the reaction rate and conversion efficiency, likely by facilitating halogen exchange or stabilizing transition states. This catalytic cycle is remarkably robust, tolerating various functional groups and delivering the biphenyl core (Formula IV) with exceptional purity, often exceeding 99% HPLC content as demonstrated in the patent examples. Such high fidelity in the early stages of synthesis is vital for preventing the propagation of impurities downstream.
Impurity control is further reinforced in the subsequent transformation steps. The conversion of the biphenyl intermediate to the disulfonyl chloride (Formula III) via chlorosulfonic acid is tightly controlled by temperature and molar ratios, preventing over-sulfonation or decomposition. The reduction of the sulfonyl chloride to the thiophenol (Formula II) using red phosphorus is a classic yet highly effective transformation that avoids the use of hazardous hydrogen gas or sensitive hydride reagents. Finally, the alkylation or etherification step to form the thioether bond is conducted under mild basic conditions, ensuring that the sensitive fluorine substituents on the aromatic ring remain intact. This meticulous attention to chemoselectivity ensures that the final high-purity biphenyl sulfide products meet the stringent specifications required for biological efficacy.
How to Synthesize Biphenyl Sulfide Compounds Efficiently
The synthesis protocol described in the patent provides a clear, step-by-step roadmap for replicating this high-yield process in a pilot or production setting. The procedure begins with the assembly of the catalytic system under inert atmosphere, followed by the controlled addition of substrates to manage exotherms. Detailed operational parameters, including specific solvent choices like DMF or dichloroethane and precise temperature ramps, are critical for success. For R&D teams looking to implement this technology, adhering to the specified molar ratios—such as the 1:0.01-0.2 ratio of substrate to nickel catalyst—is essential to balance cost and performance. The following guide outlines the standardized synthesis steps derived directly from the patent disclosure to facilitate technology transfer.
- Perform a nickel-catalyzed coupling reaction on halogenated benzene (Formula V) using zinc and a phosphine ligand to form the biphenyl core (Formula IV).
- Conduct sulfonylation of the biphenyl intermediate with chlorosulfonic acid to generate the disulfonyl chloride derivative (Formula III).
- Reduce and hydrolyze the sulfonyl chloride using red phosphorus and alkali to obtain the dimercapto-biphenyl thiophenol (Formula II).
- React the thiophenol with a halogenating agent or sulfonate in the presence of a base to finalize the biphenyl sulfide compound (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed route translates into tangible strategic benefits that extend beyond simple unit price reductions. The primary advantage lies in the stabilization of the raw material basket; by shifting from iodine and palladium to chlorine/bromine and nickel, the manufacturing process becomes immune to the geopolitical and market volatilities that frequently plague precious metal supply chains. This resilience ensures a more predictable cost structure, allowing for more accurate long-term budgeting and pricing strategies for downstream customers. Additionally, the use of commodity chemicals like zinc powder and chlorosulfonic acid simplifies logistics, as these materials are widely available from multiple global vendors, reducing the risk of single-source supply disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the substitution of costly aryl iodides with cheaper chloro- or bromo-benzene derivatives result in a drastic reduction in direct material costs. Furthermore, the simplified purification processes, which avoid the need for specialized heavy metal scavengers, lower the operational expenditure associated with waste treatment and consumables. This cumulative effect significantly enhances the gross margin potential for manufacturers producing these acaricide intermediates at scale.
- Enhanced Supply Chain Reliability: The reliance on abundant base metals and common organic solvents ensures that production schedules are not held hostage by the limited availability of niche reagents. The robustness of the nickel catalytic system also implies a wider operating window, reducing the likelihood of batch failures due to minor process deviations. This reliability is crucial for maintaining continuous supply to agrochemical formulators who depend on timely deliveries to meet seasonal agricultural demands.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex agrochemical intermediates, utilizing standard reactor configurations and workup techniques like filtration and liquid-liquid extraction. The avoidance of pyrophoric reagents and the use of recyclable solvents align with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility. This compliance advantage is increasingly important in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of answers based on the specific data and claims within patent CN111100048B. These responses clarify the operational boundaries and performance metrics of the new method, providing stakeholders with the confidence needed to evaluate its integration into their existing portfolios. Understanding these nuances is key to leveraging the full potential of this innovative chemical pathway.
Q: Why is the nickel-catalyzed method superior to the traditional palladium route?
A: The nickel-catalyzed method replaces expensive palladium catalysts and costly iodo-benzene raw materials with cheaper nickel systems and chloro- or bromo-benzene derivatives, significantly reducing raw material costs while maintaining high yields suitable for industrial scale.
Q: What are the key purity specifications achievable with this process?
A: The patent examples demonstrate HPLC normalization content reaching up to 99% for intermediate biphenyls and over 98% for the final thioether products, indicating a robust impurity profile suitable for high-efficiency acaricide manufacturing.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process utilizes standard industrial reagents like zinc powder, chlorosulfonic acid, and red phosphorus, with straightforward workup procedures involving filtration and extraction, making it highly adaptable for large-scale commercial manufacturing from 100 kgs to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the nickel-catalyzed synthesis route for biphenyl sulfide compounds. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust, industrial-scale reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite global partners to collaborate with us to optimize their supply chains for high-efficiency acaricides. By leveraging our process engineering capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a competitive edge in the agrochemical market through superior chemistry and reliable supply.
