Advanced Synthesis of Alpha-Chlorinated Carbonates for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Alpha-Chlorinated Carbonates for High-Purity Pharmaceutical Intermediates
The landscape of peptide synthesis and pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity, reduced racemization, and more cost-effective protecting group strategies. A pivotal advancement in this domain is detailed in patent CN85101227A, which introduces a novel class of alpha-chlorinated carbonates. These compounds represent a significant departure from traditional protecting groups, offering a robust solution for the temporary protection of amino groups in amino acids. For R&D directors and process chemists, understanding the mechanistic nuances of this technology is crucial, as it provides a pathway to synthesize complex peptides with superior control over stereochemistry and impurity profiles. The patent outlines a versatile synthetic route that not only simplifies the introduction of protecting groups but also ensures their facile removal, thereby addressing long-standing challenges in the production of high-value API intermediates.
Traditionally, the protection of amino acids has relied heavily on established groups such as benzyloxycarbonyl (Z), tert-butoxycarbonyl (BOC), and fluorenylmethyloxycarbonyl (FMOC). While effective, these conventional methods often suffer from significant limitations that impact both the efficiency and the economic viability of large-scale manufacturing. For instance, the synthesis of certain chloroformates required for these groups can be hazardous or involve unstable intermediates that are difficult to handle safely. Furthermore, the removal of these protecting groups sometimes necessitates harsh acidic or basic conditions, or even hydrogenolysis, which can lead to the degradation of sensitive peptide bonds or the racemization of chiral centers. In contrast, the novel approach described in the patent utilizes alpha-chlorinated carbonates that are remarkably stable yet reactive enough to form carbamates under mild conditions. This stability allows for easier handling and storage, while their specific reactivity profile ensures that the deprotection step proceeds cleanly without compromising the integrity of the final pharmaceutical product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on traditional protecting groups has created bottlenecks in the supply chain of complex pharmaceutical intermediates. Methods involving azides, for example, pose severe safety risks due to their potential for explosive decomposition, requiring specialized infrastructure and rigorous safety protocols that drive up operational costs. Similarly, the use of fluorides or mixed carbonates often involves expensive raw materials that are not readily available on the global market, leading to supply chain vulnerabilities and price volatility. Moreover, the byproducts generated during the deprotection of conventional groups can be difficult to remove, necessitating additional purification steps such as chromatography or recrystallization, which significantly reduce overall yield and increase solvent waste. These inefficiencies accumulate, resulting in a manufacturing process that is not only costly but also environmentally burdensome, failing to meet the increasingly stringent sustainability standards of the modern chemical industry.
The Novel Approach
The innovative methodology presented in the patent circumvents these issues by employing alpha-chlorinated carbonates that are synthesized through a straightforward reaction between hydroxy compounds and alpha-chloro chloroformates. This reaction is conducted in the presence of an acid scavenger, such as pyridine or triethylamine, under mild temperature conditions ranging from -20°C to +50°C. The beauty of this approach lies in its simplicity and versatility; it accommodates a wide range of R1 groups, including aliphatic, alicyclic, and aromatic substituents, allowing chemists to tailor the protecting group to the specific needs of the synthesis. Furthermore, the deprotection mechanism is uniquely advantageous, as it releases volatile aldehydes (CX3CHO) alongside the free amine, facilitating easy separation and driving the reaction to completion without the need for complex workup procedures. This streamlined process translates directly into higher throughput and reduced operational complexity for manufacturing facilities.
Mechanistic Insights into Alpha-Chlorinated Carbonate Formation and Application
At the heart of this technology is a sophisticated yet elegant chemical mechanism that ensures high fidelity in peptide bond formation. The synthesis of the alpha-chlorinated carbonate itself involves the nucleophilic attack of the hydroxyl group of an alcohol on the carbonyl carbon of the alpha-chloro chloroformate. This step is critical, as the presence of the electron-withdrawing halogen atoms (fluorine, chlorine, or bromine) on the alpha-carbon enhances the electrophilicity of the carbonyl group, facilitating the reaction even under mild conditions. The role of the acid scavenger is paramount here; it neutralizes the hydrochloric acid generated during the reaction, preventing the protonation of the alcohol or the decomposition of the sensitive chloroformate intermediate. By carefully controlling the addition of the base and maintaining the temperature within the optimal window, the process minimizes side reactions, ensuring that the resulting carbonate is obtained in high purity and yield, ready for immediate use in downstream applications.
When applied to amino acid protection, the mechanism demonstrates exceptional selectivity. The alpha-chlorinated carbonate reacts with the amino group of the amino acid to form a stable carbamate linkage. Crucially, unlike other alpha-chloro compounds that might alkylate the nitrogen or cause unwanted chlorination of the carbon skeleton, this specific reagent architecture directs the reaction exclusively towards carbamate formation. The subsequent deprotection is equally fascinating; under acidic conditions, the carbamate cleaves to release the free amine, carbon dioxide, and a trichloroacetaldehyde derivative. This specific cleavage pathway avoids the formation of stable urea byproducts that often plague other methods, thereby simplifying the purification of the final peptide. For R&D teams, this means a cleaner reaction profile, easier analytical characterization, and a final product that meets the rigorous purity specifications required for clinical-grade APIs.
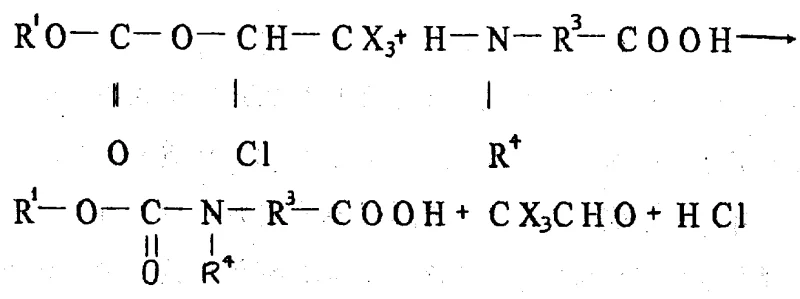
How to Synthesize Alpha-Chlorinated Carbonates Efficiently
Implementing this novel synthesis route requires a precise understanding of the reaction parameters to maximize yield and safety. The process begins with the selection of appropriate starting materials, specifically the hydroxy compound and the alpha-chloro chloroformate, which must be of high purity to prevent the introduction of impurities early in the sequence. The reaction is typically carried out in an inert solvent such as dichloromethane, dioxane, or a mixture thereof, which provides a homogeneous medium for the reactants to interact effectively. The addition of the acid scavenger must be controlled carefully, often added dropwise to manage the exotherm and ensure complete neutralization of the evolved acid. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility and are outlined in the comprehensive guide below.
- React a hydroxy compound (ROH) with an alpha-chloro chloroformate in an inert solvent medium such as dichloromethane or dioxane.
- Maintain the reaction temperature between -20°C and +50°C, preferably between -5°C and +5°C, while gradually adding an acid scavenger like pyridine or triethylamine.
- After completion, separate the organic phase, wash with water, dry over magnesium sulfate, and evaporate the solvent to isolate the pure alpha-chlorinated carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this alpha-chlorinated carbonate technology offers compelling strategic advantages that extend beyond mere technical performance. The primary benefit lies in the drastic simplification of the raw material supply chain. Unlike traditional methods that rely on scarce or hazardous reagents, this process utilizes readily available alcohols and chloroformates that are produced on a massive industrial scale, ensuring a stable and continuous supply. This reliability mitigates the risk of production stoppages due to material shortages, a critical factor in maintaining the continuity of API manufacturing. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure reactors or cryogenic equipment, allowing the process to be run in standard glass-lined or stainless steel vessels found in most multipurpose chemical plants. This compatibility with existing infrastructure significantly lowers the barrier to entry for scale-up, reducing capital expenditure requirements.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the elimination of expensive and hazardous reagents. By avoiding the use of azides or complex fluorides, manufacturers can significantly reduce raw material costs and the associated expenses for safety management and waste disposal. The high yields reported in the patent examples, often exceeding 90%, mean that less starting material is wasted, directly improving the cost of goods sold (COGS). Additionally, the simplified workup procedure, which often involves simple extraction and crystallization rather than column chromatography, reduces solvent consumption and labor hours. These cumulative efficiencies result in a substantially lower production cost per kilogram of the protected amino acid, providing a competitive edge in the pricing of final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route translates directly into supply chain resilience. Because the reaction tolerates a wide range of functional groups and conditions, it is less prone to batch-to-batch variability, ensuring consistent quality output. The ability to produce a variety of protecting groups (such as t-butyl, fluorenylmethyl, or trichloroethyl derivatives) using the same core methodology allows for a flexible manufacturing strategy. This flexibility enables suppliers to respond quickly to changing market demands for different peptide sequences without needing to retool entire production lines. Consequently, lead times for high-purity pharmaceutical intermediates can be reduced, allowing downstream drug developers to accelerate their own clinical timelines and time-to-market.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles. The avoidance of heavy metals and explosive reagents reduces the toxicity of the waste stream, simplifying effluent treatment and lowering compliance costs. The generation of volatile aldehydes as byproducts during deprotection facilitates their removal, minimizing the burden on wastewater treatment facilities. Furthermore, the scalability of the reaction is proven by its operation at atmospheric pressure and moderate temperatures, which are inherently safer and easier to control on a multi-ton scale. This ease of scale-up ensures that the transition from laboratory benchtop to commercial production is smooth and predictable, minimizing the risks associated with technology transfer and validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of alpha-chlorinated carbonate technology in pharmaceutical synthesis. These insights are derived directly from the experimental data and theoretical framework provided in the patent literature, offering a clear perspective on its practical utility. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the advantages of using alpha-chlorinated carbonates over traditional protecting groups like BOC or Z?
A: Unlike traditional groups that may require harsh conditions for removal or complex multi-step synthesis, these novel alpha-chlorinated carbonates offer mild reaction conditions, high yields, and a unique cleavage mechanism that generates volatile aldehydes, simplifying purification.
Q: Can this method be used for amino acids with sensitive side chains?
A: Yes, the process operates under mild temperatures (0-30°C) and neutral to slightly basic conditions, making it highly suitable for protecting amino acids with sensitive functional groups like hydroxyls (Serine, Tyrosine) or sulfhydryls without causing racemization.
Q: Is this synthesis scalable for industrial production?
A: Absolutely. The reaction uses readily available raw materials, common solvents, and atmospheric pressure, eliminating the need for specialized high-pressure equipment or exotic catalysts, which facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Chlorinated Carbonates Supplier
The potential of alpha-chlorinated carbonates to revolutionize peptide synthesis is immense, offering a blend of efficiency, safety, and cost-effectiveness that is rare in fine chemical manufacturing. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory techniques into robust industrial processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot scale to full commercialization is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation, allowing us to meet stringent purity specifications for even the most complex API intermediates. We are committed to delivering high-quality chemical solutions that empower your drug development pipeline.
We invite you to explore how our expertise in advanced organic synthesis can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating exactly how this technology can benefit your bottom line. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. Let us collaborate to bring your next-generation pharmaceutical products to market faster and more efficiently.
