Scalable Electrochemical Halogenation: A Green Route for Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize complex building blocks, particularly halogenated heterocycles which serve as critical precursors for cross-coupling reactions. Patent CN114672829A introduces a groundbreaking electrochemical methodology for synthesizing unsaturated ring halides, addressing long-standing challenges regarding toxicity and waste generation in traditional halogenation processes. This innovation utilizes a diaphragm-free electrolytic cell where electrons act as the primary redox reagent, enabling the direct conversion of substrates like pyrazoles, indoles, and thiophenes into their corresponding halogenated derivatives. By leveraging anodic oxidation strategies, this technique generates active halogen intermediates in situ, bypassing the need for hazardous elemental halogens or high-valence oxidants. The versatility of this approach is evident in its broad substrate scope, successfully accommodating electron-rich systems that are often sensitive to harsh chemical conditions.
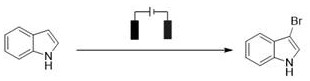
Furthermore, the patent highlights the operational simplicity of the method, which proceeds under mild conditions such as room temperature and atmospheric pressure. The use of inexpensive graphite electrodes and common halide salts like sodium bromide drastically lowers the barrier to entry for implementation. For R&D teams focused on process intensification, the ability to tune the degree of halogenation—from mono- to poly-substituted products—simply by adjusting the charge passed (Faradays) and reaction time offers unprecedented control over the impurity profile. This level of precision is crucial for producing high-purity pharmaceutical intermediates where strict regulatory standards must be met. The integration of flow chemistry capabilities, as demonstrated in the larger-scale examples within the patent, further underscores the potential for this technology to transition seamlessly from laboratory discovery to commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of unsaturated ring halides has relied heavily on the use of elemental halogens such as bromine or chlorine, which pose severe safety risks due to their high toxicity, corrosivity, and volatility. Handling these reagents requires specialized equipment and rigorous safety protocols, increasing both capital expenditure and operational complexity. Alternatively, chemists have often turned to N-halosuccinimides (like NBS) or high-valence metal oxidants to achieve similar transformations. However, these stoichiometric reagents generate substantial amounts of organic waste, such as succinimide byproducts, which complicate downstream purification and increase the environmental footprint of the process. Additionally, many traditional methods require transition metal catalysts that can leave trace metal residues in the final product, necessitating expensive and time-consuming purification steps to meet pharmaceutical purity specifications. The reliance on these conventional reagents often results in poor atom economy and higher overall production costs.
The Novel Approach
The electrochemical method described in CN114672829A represents a paradigm shift by replacing chemical oxidants with electrical energy. This approach fundamentally alters the reaction landscape by generating the active halogenating species directly at the anode surface through the oxidation of benign halide ions. This in situ generation ensures that the concentration of the reactive species is kept low and controlled, minimizing side reactions such as over-halogenation or polymerization that are common in batch chemical processes. The absence of external oxidants and metal catalysts means that the reaction mixture is significantly cleaner, simplifying the workup procedure to basic extraction and crystallization. Moreover, the compatibility of this method with aqueous solvent systems, such as acetonitrile and water mixtures, aligns perfectly with modern green chemistry principles, reducing the reliance on volatile organic compounds. The visual contrast between the traditional NBS method and this electrochemical route is stark, particularly when considering the yield improvements and waste reduction achieved.
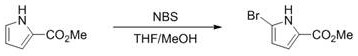
Mechanistic Insights into Anodic Oxidation Halogenation
The core mechanism driving this transformation involves the anodic oxidation of halide ions (X⁻) to generate highly reactive halogen radicals or cationic species (X• or X⁺) at the electrode interface. When a constant current is applied across the graphite electrodes, the halide ions present in the solution migrate to the anode where they lose electrons. These electro-generated active species then act as electrophiles, attacking the electron-rich positions of the unsaturated ring substrate, such as the C3 position of indoles or the C4 position of pyrazoles. This electrophilic aromatic substitution proceeds with high regioselectivity, dictated by the electronic density of the substrate and the specific potential applied. Simultaneously, at the cathode, protons from the solvent or added proton sources (like acetic acid) are reduced to hydrogen gas, maintaining charge balance without introducing foreign chemical reductants. This dual-electrode process ensures that the only net reagents consumed are the substrate and the halide salt, with electricity serving as the traceless reagent.
Controlling the impurity profile in such electrochemical reactions is achieved through precise management of the current density and the total charge passed (measured in Faradays). The patent specifies a preferred current density range of 50 to 150 mA/cm², which optimizes the rate of active species generation to match the kinetics of the substitution reaction, thereby preventing the accumulation of reactive intermediates that could lead to decomposition. Furthermore, the addition of a proton source helps to facilitate the cathodic reaction and can enhance the solubility of the substrate, ensuring a homogeneous reaction environment. The use of a diaphragm-free cell simplifies the reactor design but relies on the rapid consumption of the generated halogen species to prevent back-reaction or diffusion to the cathode. This mechanistic elegance allows for the synthesis of not just monohalogenated products but also di- and tri-halogenated derivatives by simply extending the electrolysis time and increasing the molar ratio of the halide salt, offering a versatile platform for diverse synthetic needs.
How to Synthesize Unsaturated Ring Halides Efficiently
To implement this synthesis effectively, one must carefully prepare the reaction mixture by dissolving the chosen unsaturated substrate and the appropriate halide salt in a solvent system, typically a mixture of acetonitrile and water. The addition of a proton source like acetic acid is often beneficial to enhance conductivity and reaction rates. Once the solution is prepared, it is subjected to constant current electrolysis in a cell equipped with graphite electrodes. The detailed standardized synthesis steps, including specific molar ratios, current densities, and workup procedures for various substrates, are outlined in the guide below.
- Mix the unsaturated ring substrate (e.g., indole, pyrazole) with a cheap halide salt (e.g., NaBr) and a solvent system like acetonitrile/water in a reaction vessel.
- Apply a constant current (preferably 50-150 mA/cm²) using graphite electrodes in a diaphragm-free electrolytic cell at room temperature to generate active halogen species in situ.
- Upon completion, perform standard workup procedures including extraction with ethyl acetate, drying, and column chromatography to isolate the high-purity halogenated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material supply chain. By replacing expensive, hazard-classified reagents like elemental bromine or NBS with commodity chemicals like sodium bromide, companies can significantly reduce raw material costs and mitigate supply risks associated with regulated substances. The elimination of transition metal catalysts further removes the need for costly scavenging resins or complex filtration steps, streamlining the manufacturing process and reducing the consumption of auxiliary materials. This simplification translates directly into lower operating expenses and a more robust, resilient supply chain capable of withstanding market fluctuations in reagent pricing.
- Cost Reduction in Manufacturing: The electrochemical process fundamentally changes the cost structure of halogenation by utilizing electricity as the primary driver of the reaction. Since electrons are generally cheaper and more abundant than chemical oxidants, the variable cost per kilogram of product is substantially lowered. Furthermore, the avoidance of stoichiometric oxidants means that there is less waste to treat and dispose of, leading to significant savings in waste management fees. The use of durable graphite electrodes, which are inexpensive and long-lasting compared to precious metal electrodes like platinum, ensures that capital depreciation costs remain low over the lifecycle of the equipment. These factors combine to create a highly cost-competitive manufacturing route that enhances profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: Relying on widely available inorganic salts like sodium bromide or potassium bromide ensures a stable and secure supply of key reagents, unlike specialized organic oxidants which may have limited suppliers or long lead times. The ability to perform the reaction in a flow cell setup, as demonstrated in the patent's 100g-scale example, allows for continuous manufacturing operations that can be easily ramped up to meet sudden increases in demand. This scalability reduces the lead time for delivering high-purity intermediates to customers, providing a strategic advantage in fast-paced pharmaceutical development timelines. Additionally, the reduced hazard profile of the reagents simplifies logistics and storage requirements, allowing for more flexible inventory management.
- Scalability and Environmental Compliance: The green nature of this electrochemical method aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. By generating hydrogen gas as the only byproduct at the cathode and avoiding the creation of heavy metal waste or organic oxidant residues, the process minimizes the environmental footprint of the manufacturing site. The patent explicitly demonstrates the feasibility of scaling this reaction from milligram scales in the lab to 100g scales in a flow reactor, indicating a clear path to multi-kilogram and ton-scale production. This scalability ensures that the technology can support the commercialization of new drug candidates without requiring a complete process redesign, thereby securing long-term supply continuity for critical API intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical halogenation technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN114672829A, ensuring accuracy and relevance for process development teams. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: How does this electrochemical method improve safety compared to traditional halogenation?
A: Traditional methods often require handling highly toxic elemental halogens (like bromine gas) or explosive high-valence oxidants. This electrochemical approach generates the active halogenating species in situ from safe, inexpensive salts like sodium bromide, eliminating the need to store or transport hazardous reagents.
Q: Can this process be scaled up for industrial manufacturing?
A: Yes, the patent explicitly demonstrates scalability using a flow cell apparatus. Example 16 details a 100g-scale preparation in a flow system with high yield, proving that the technology is suitable for continuous manufacturing and large-scale commercial production.
Q: What are the cost advantages of using this electrochemical route?
A: The process eliminates the need for expensive transition metal catalysts and stoichiometric chemical oxidants. By using electricity as the primary reagent and cheap halide salts, the raw material costs are significantly reduced, and the downstream purification is simplified by avoiding metal residue removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Unsaturated Ring Halide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114672829A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-quality unsaturated ring halides that empower your drug discovery and development programs.
We invite you to collaborate with us to explore how this green halogenation technology can optimize your specific supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how switching to electrochemical methods can reduce your overall cost of goods. Please contact us today to request specific COA data for our catalog of halogenated intermediates or to discuss route feasibility assessments for your custom synthesis projects. Together, we can build a more sustainable and efficient future for chemical manufacturing.
