Advanced Synthesis of Bactericidal Pyridine Compounds for Scalable Agrochemical Applications
The chemical industry is constantly seeking more efficient and environmentally benign methods for producing active antimicrobial agents, and patent CN1875000A presents a significant breakthrough in this domain. This patent discloses a novel method for producing bactericidal pyridine compounds, specifically bis-quaternary ammonium salts, which exhibit superior antibacterial and fungicidal activity compared to conventional alternatives. The core innovation lies in the use of easily available pyridine compounds as starting materials, processed through a simplified synthetic route that drastically reduces production complexity. By leveraging a stepwise etherification followed by quaternization, the process achieves high yields while minimizing the formation of toxic biodegradation byproducts often associated with older technologies. This advancement is particularly critical for manufacturers aiming to enhance their portfolio with high-performance agrochemical intermediates that meet rigorous safety and efficacy standards. The technical details provided in the patent offer a robust framework for scaling these reactions from laboratory benchtops to industrial reactors, ensuring consistency and reliability in supply.
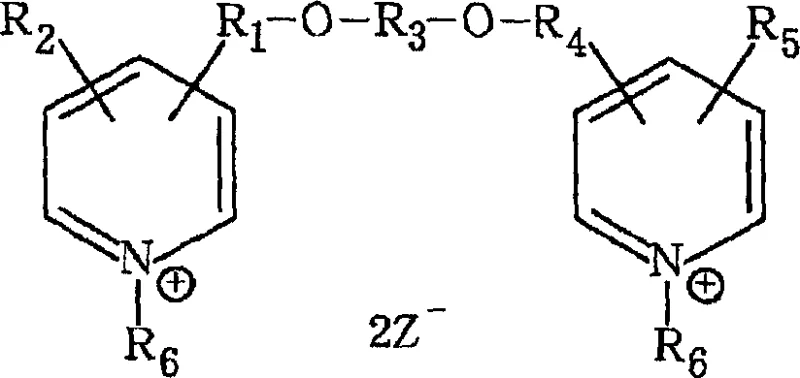
The limitations of conventional methods for synthesizing bis-quaternary ammonium compounds are well-documented, primarily revolving around environmental safety and process efficiency. Traditional routes often rely on starting materials that are difficult to source or require harsh reaction conditions that generate significant waste streams. Furthermore, the biodegradation products of many existing antibacterial agents possess high residual toxicity, posing risks to aquatic ecosystems and limiting their application scope in sensitive environments. Conventional processes also struggle with selectivity, often resulting in complex mixtures that require energy-intensive purification steps to achieve the necessary purity levels for commercial use. These factors collectively drive up manufacturing costs and extend lead times, creating bottlenecks for procurement teams seeking reliable sources of high-quality intermediates. The inability of older methods to effectively handle scale-up without compromising on safety or cost efficiency remains a persistent challenge in the fine chemical sector.
In contrast, the novel approach detailed in the patent overcomes these hurdles through a strategically designed multi-step synthesis that prioritizes atom economy and operational simplicity. The process begins with the nucleophilic substitution of a pyridine derivative with a diol, facilitated by a strong base in a polar aprotic solvent, which ensures high conversion rates under mild temperature conditions. This initial etherification step is carefully controlled to prevent over-reaction, setting the stage for the subsequent formation of the bis-ether intermediate. The final quaternization step utilizes readily available alkyl halides or sulfonates, reacting them with the bis-ether precursor to form the target cationic structure. This modular approach allows for flexibility in tuning the alkyl chain lengths to optimize biological activity without altering the core synthetic workflow. By eliminating the need for exotic reagents and complex catalytic systems, the new method significantly lowers the barrier to entry for commercial production while enhancing the overall sustainability profile of the manufacturing process.
Mechanistic Insights into Stepwise Etherification and Quaternization
The mechanistic pathway of this synthesis is rooted in classic nucleophilic substitution chemistry, optimized for high selectivity and yield. In the first stage, the hydroxyl group of the diol is activated by a strong base, such as potassium tert-butoxide, to form an alkoxide species which then attacks the electrophilic carbon of the pyridine methyl halide. This reaction is conducted in dimethylformamide (DMF), a solvent chosen for its ability to dissolve both organic salts and polar intermediates, thereby facilitating efficient molecular collisions. The temperature is maintained between -20°C and 10°C during this phase to suppress side reactions and ensure the formation of the mono-ether intermediate with minimal byproduct generation. The precise control of stoichiometry and addition rates is crucial here, as it dictates the purity of the intermediate which directly impacts the efficiency of the subsequent steps. This level of mechanistic control is essential for R&D directors focused on impurity profiling and process robustness.
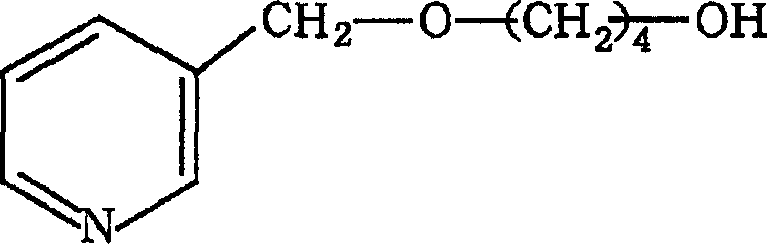
Following the formation of the mono-ether, the second etherification step involves reacting this intermediate with another equivalent of the pyridine derivative under similar basic conditions. This step completes the construction of the bis-pyridine ether backbone, which serves as the scaffold for the final quaternary ammonium salt. The reaction mechanism here mirrors the first step but requires careful monitoring to ensure complete conversion of the remaining hydroxyl group. Once the bis-ether is secured, the final transformation involves quaternization of the pyridine nitrogen atoms using long-chain alkyl halides. This step proceeds at elevated temperatures, typically between 40°C and 80°C, driving the reaction to completion and forming the stable cationic centers responsible for the compound's bactericidal properties. The resulting bis-quaternary ammonium salt exhibits a unique balance of lipophilicity and charge density, which enhances its ability to penetrate microbial cell membranes and disrupt vital biological functions.
How to Synthesize Bactericidal Pyridine Compound Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and purity while ensuring safety. The process is divided into distinct stages, beginning with the preparation of the alkoxide solution and the controlled addition of the pyridine starting material. Detailed standard operating procedures guide the handling of strong bases and the management of exothermic events during the addition phases. The workup procedures involve straightforward solid-liquid separations and solvent exchanges, which are amenable to standard industrial equipment. For a comprehensive guide on the exact molar ratios, addition times, and crystallization conditions, please refer to the standardized synthesis steps provided below.
- React a substituted pyridine compound with a diol in the presence of a strong base like potassium tert-butoxide in DMF to form a mono-ether intermediate.
- Further react the mono-ether intermediate with another equivalent of pyridine compound under basic conditions to form the bis-ether pyridine precursor.
- Quaternize the bis-ether precursor with an alkyl halide or sulfonate at elevated temperatures to yield the final bactericidal bis-quaternary ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
The commercial implications of adopting this synthesis route are profound, offering tangible benefits for procurement managers and supply chain heads focused on cost efficiency and reliability. By utilizing easily available starting materials such as chloromethylpyridine and common diols, the process decouples production from volatile supply chains dependent on specialized precursors. This shift significantly enhances supply chain reliability, ensuring that manufacturing schedules are not disrupted by raw material shortages. Furthermore, the simplified reaction sequence reduces the number of unit operations required, leading to substantial cost savings in terms of energy consumption, labor, and equipment utilization. The ability to recover and reuse excess alkylating agents further contributes to cost reduction in agrochemical intermediate manufacturing, making the final product highly competitive in the global market. These efficiencies translate directly into improved margins and a more resilient supply network for downstream formulators.
- Cost Reduction in Manufacturing: The elimination of complex catalytic systems and the use of cost-effective solvents like DMF drive down the overall production cost significantly. The process avoids expensive transition metals and intricate purification columns, relying instead on crystallization and extraction techniques that are cheaper to operate at scale. This streamlined approach reduces the capital expenditure required for plant setup and lowers the operational expenditure associated with waste treatment and solvent recovery. Consequently, manufacturers can offer high-purity bactericidal pyridine compounds at a more attractive price point without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as the key reagents are commodity chemicals produced by multiple suppliers globally. This diversity in the supply base mitigates the risk of single-source dependency and allows for flexible procurement strategies. The robustness of the reaction conditions also means that production can be maintained consistently across different facilities, reducing lead time for high-purity bactericidal pyridine compounds. This reliability is crucial for maintaining continuous production lines in the agrochemical and personal care sectors where interruptions can be costly.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plants to full-scale commercial reactors. The absence of hazardous heavy metals and the generation of less toxic biodegradation products align with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and associated costs for waste disposal, facilitating smoother market entry and long-term sustainability. The commercial scale-up of complex quaternary ammonium salts via this route is therefore not only technically feasible but also environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bactericidal pyridine compounds. The answers are derived directly from the patent data to ensure accuracy and relevance for industry stakeholders. Understanding these aspects helps in making informed decisions about integrating this technology into existing product lines or supply chains.
Q: What are the advantages of this pyridine compound synthesis over conventional methods?
A: This method uses easily available starting materials and avoids complex purification steps, leading to significant cost reduction and simplified supply chain management compared to traditional bis-quaternary ammonium salt production.
Q: How does this process ensure high purity for agrochemical applications?
A: The process utilizes specific reaction conditions and solvent systems like DMF that facilitate high conversion rates and allow for effective crystallization or extraction, ensuring stringent purity specifications are met.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the reaction conditions operate within standard temperature ranges and use common industrial solvents, making the commercial scale-up of complex quaternary ammonium salts feasible and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bactericidal Pyridine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a partner who can translate complex patent chemistry into reliable commercial supply. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless. We are committed to meeting stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify the identity and quality of every batch. Our expertise in handling quaternization reactions and etherification processes allows us to optimize yields and minimize impurities, delivering a product that consistently meets the high standards required by global agrochemical and fine chemical manufacturers.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. We encourage you to reach out to request specific COA data and route feasibility assessments to understand how we can support your supply chain optimization goals. By partnering with us, you gain access to a reliable source of high-performance intermediates that can enhance the efficacy and sustainability of your final products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
