Scalable Copper-Catalyzed Dimerization of Aromatic Olefins for Advanced Pharmaceutical Intermediates Manufacturing
Scalable Copper-Catalyzed Dimerization of Aromatic Olefins for Advanced Pharmaceutical Intermediates Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for sustainable, cost-effective, and scalable methodologies. A pivotal development in this arena is detailed in Chinese Patent CN115141073A, which discloses a novel preparation method for aromatic hydrocarbon dimer compounds. This technology represents a paradigm shift from traditional noble metal catalysis to a more economical copper-based system, specifically targeting the efficient construction of C-C bonds in diaryl skeleton structures. These structures are ubiquitous in high-value pharmaceutical intermediates and bioactive compounds, such as dimethindene, which possesses antipruritic and anti-inflammatory properties. By leveraging a composite catalyst system comprising copper salts and trifluoromethanesulfonic acid derivatives, this invention offers a streamlined, one-step pathway to produce aromatic olefin dimers with exceptional atom economy and operational simplicity.
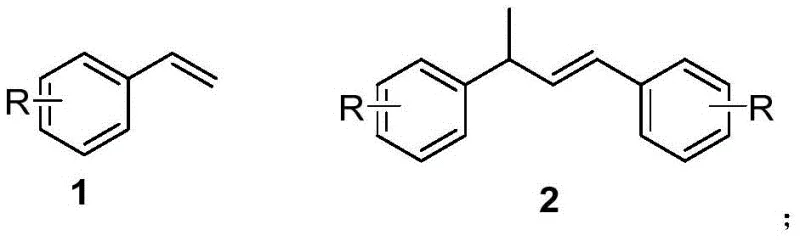
For R&D directors and process chemists, the implications of this patent extend beyond mere academic interest; it provides a tangible route to reduce the cost of goods sold (COGS) for complex intermediates. The ability to utilize readily available starting materials (Formula 1) and convert them into valuable dimeric structures (Formula 2) under mild heating conditions addresses critical bottlenecks in process development. Furthermore, the method's compatibility with various substituents (R = H, alkyl, halogen) ensures versatility across different synthetic targets, making it a robust candidate for integration into existing manufacturing pipelines for reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the catalytic dimerization of aryl alkenes has relied heavily on transition metal catalysis involving palladium, indium, ruthenium, or cobalt. While these methods are chemically efficient, they present substantial logistical and economic challenges for large-scale industrial application. The primary drawback lies in the exorbitant cost of the metal catalysts themselves, particularly palladium and ruthenium, which are subject to volatile market pricing and supply chain constraints. Additionally, these conventional protocols often necessitate the use of sophisticated and expensive phosphine ligands and specialized additives to stabilize the active catalytic species. This reliance on precious metals not only inflates production costs but also introduces significant environmental burdens due to the generation of heavy metal waste, which requires rigorous and costly removal processes to meet stringent pharmaceutical purity standards.
The Novel Approach
In stark contrast, the methodology described in CN115141073A utilizes a composite catalyst system based on abundant and inexpensive copper salts paired with trifluoromethanesulfonic acid derivatives. This approach effectively bypasses the need for noble metals and complex ligand systems, thereby drastically simplifying the reaction setup and post-processing requirements. The invention creatively combines the substrate, the composite catalyst, and an organic solvent in a single vessel, where heating facilitates a direct one-step conversion to the desired dimer. This simplification translates directly into operational efficiency, as it reduces the number of unit operations and minimizes the consumption of auxiliary reagents. Moreover, the use of copper, a base metal, aligns perfectly with green chemistry principles by reducing the environmental footprint and eliminating the need for extensive heavy metal scavenging steps typically required for Pd-catalyzed processes.
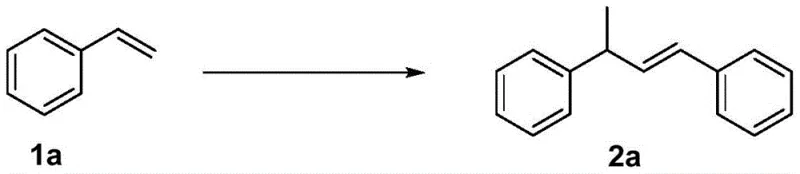
Mechanistic Insights into Cu/TfOH-Catalyzed Dimerization
The mechanistic pathway proposed for this transformation involves the formation of a unique composite metal catalyst species generated in situ from the interaction between the copper catalyst and the trifluoromethanesulfonic acid component. This synergistic combination creates a highly active electrophilic environment that facilitates the migration and insertion of one molecule of the starting aromatic olefin (Formula 1) into the catalyst complex, forming a key intermediate designated as Compound B. Subsequently, a second molecule of the starting material undergoes oxidative addition with Compound B to generate Compound C. The cycle concludes with a reductive elimination step that releases the final dimer product (Formula 2) and regenerates the active catalyst species. This clear mechanistic understanding allows for precise tuning of reaction parameters, such as the molar ratio of copper to acid (optimized at 1:10 to 1:30), ensuring maximum turnover frequency and minimizing side reactions.
From an impurity control perspective, the mild reaction conditions (50°C to 70°C) play a crucial role in maintaining high product purity. Unlike harsher conditions that might promote polymerization or uncontrolled oligomerization, this thermal window is sufficient to drive the dimerization while suppressing the formation of higher molecular weight byproducts. The inclusion of quinone compounds, such as 1,4-benzoquinone or 2-hydroxy-1,4-naphthoquinone, further refines the reaction profile by acting as oxidants or co-catalysts that stabilize the copper oxidation states involved in the cycle. This results in a cleaner crude reaction mixture, which significantly eases the burden on downstream purification processes like flash chromatography or crystallization, ultimately delivering high-purity aromatic hydrocarbon dimers suitable for sensitive pharmaceutical applications.
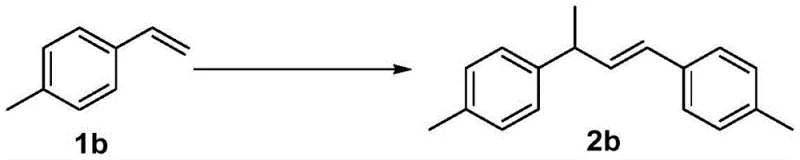
How to Synthesize (E)-1,3-Diphenyl-1-butene Efficiently
The synthesis of high-value dimeric intermediates using this patented protocol is designed for straightforward execution in standard laboratory or pilot plant settings. The procedure involves dissolving the aromatic olefin substrate in a polar aprotic solvent such as 1,4-dioxane or acetonitrile, followed by the sequential addition of the copper source (e.g., cuprous chloride) and the triflate promoter. The addition of a catalytic amount of a quinone additive is a critical step to unlock the full potential of the catalyst system, pushing yields towards quantitative levels. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the aromatic olefin substrate (Formula 1), a copper catalyst (e.g., CuCl), and a trifluoromethanesulfonic acid catalyst in an organic solvent such as 1,4-dioxane.
- Add a quinone compound additive, such as 2-hydroxy-1,4-naphthoquinone, to the mixture to enhance catalytic efficiency and promote higher yields.
- Heat the reaction mixture to a temperature between 50°C and 70°C and stir for 8 to 12 hours, followed by standard aqueous workup and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed dimerization technology offers compelling strategic advantages centered around cost stability and supply security. By transitioning away from palladium and other precious metals, manufacturers can insulate their production costs from the volatility of the noble metal market. The raw materials required for this process—copper salts, triflic acid derivatives, and simple styrenes—are commodity chemicals with robust global supply chains, ensuring consistent availability and preventing production stoppages due to material shortages. This shift not only lowers the direct material costs but also reduces the overhead associated with the storage and handling of hazardous or highly regulated precious metal catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive phosphine ligands and noble metal catalysts results in a substantial reduction in raw material expenditure. Since copper is orders of magnitude cheaper than palladium or ruthenium, the catalyst cost per kilogram of product is drastically minimized. Furthermore, the simplified workup procedure, which avoids complex metal scavenging resins, reduces the consumption of auxiliary processing materials and lowers waste disposal costs, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Relying on base metal catalysis mitigates the risk of supply chain disruptions often associated with geographically concentrated sources of precious metals. The reagents used in this protocol are widely produced by multiple chemical suppliers globally, fostering a competitive sourcing environment that drives down prices and ensures continuity of supply. This reliability is critical for maintaining uninterrupted production schedules for key pharmaceutical intermediates, thereby strengthening relationships with downstream clients who demand consistent delivery performance.
- Scalability and Environmental Compliance: The reaction operates under mild thermal conditions and uses common organic solvents, making it inherently safer and easier to scale from kilogram to multi-ton production volumes. The reduced generation of heavy metal waste simplifies compliance with increasingly stringent environmental regulations regarding effluent discharge. This environmental friendliness not only reduces regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in securing contracts with major multinational pharmaceutical companies focused on green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this dimerization technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection and reaction scope. Understanding these nuances is essential for process engineers aiming to adapt this methodology to specific proprietary substrates.
Q: What is the primary advantage of the Cu/TfOH catalyst system over traditional palladium catalysis?
A: The copper/trifluoromethanesulfonic acid system eliminates the need for expensive noble metals like palladium or ruthenium and avoids costly phosphine ligands, significantly reducing raw material costs while maintaining high atom economy.
Q: Can this dimerization method tolerate electron-withdrawing groups on the aromatic ring?
A: Yes, the method demonstrates robust substrate scope, successfully dimerizing substrates with halogen substituents such as 4-chlorostyrene, although yields may vary slightly compared to electron-donating groups.
Q: Is the addition of quinone compounds necessary for the reaction to proceed?
A: While the reaction can initiate without quinones, experimental data indicates that additives like 2-hydroxy-1,4-naphthoquinone are critical for achieving optimal yields (up to 99%) and ensuring complete conversion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Hydrocarbon Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed dimerization technology described in CN115141073A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aromatic hydrocarbon dimer we deliver meets the exacting standards required for drug substance manufacturing.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this cost-effective synthesis route into your supply chain. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how our commitment to innovation and quality can drive value for your organization.
