Advanced Synthesis of Benzofuran Intermediates for High-Purity Antiarrhythmic Drug Manufacturing
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex active pharmaceutical ingredient (API) intermediates, particularly for cardiovascular therapies. Patent CN1306000A introduces a significant advancement in the preparation of 3-(1-hydroxy-pentylidene)-5-nitro-3H-benzofuran-2-one, a critical building block in the synthesis of Dronedarone, a potent antiarrhythmic agent. This patent details a novel chemical process that overcomes historical limitations associated with the acylation of benzofuran derivatives, offering a pathway that enhances both yield and purity profiles. By optimizing reaction conditions and introducing a strategic acidification workup, the disclosed method provides a reliable foundation for the commercial production of high-value pharmaceutical intermediates. For R&D directors and procurement specialists, understanding the nuances of this technology is essential for securing a stable supply chain for next-generation cardiac medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzofuran-2-ones has relied on methodologies described in earlier literature, such as the works by J.N. Chatterjea published in the mid-1950s. These conventional approaches often suffered from inefficient reaction stoichiometry, requiring large excesses of expensive acid anhydrides to drive the acylation to completion. Furthermore, the isolation of the target compound from the crude reaction mixture was frequently problematic, leading to significant product loss during purification and lower overall yields. The lack of a dedicated precipitation or crystallization step in these older protocols meant that impurities, including unreacted starting materials and side products, were difficult to remove, compromising the quality of the intermediate. For large-scale manufacturing, these inefficiencies translate into higher production costs, increased waste generation, and inconsistent batch-to-batch quality, posing substantial risks for supply chain reliability.
The Novel Approach
The methodology outlined in CN1306000A represents a distinct improvement over these legacy processes by introducing specific modifications to the reaction and workup phases. A key innovation is the controlled acidification of the reaction mixture post-synthesis, which facilitates the precise precipitation of the desired 3-(1-hydroxy-pentylidene)-5-nitro-3H-benzofuran-2-one. This step not only simplifies the isolation process but also significantly enhances the recovery rate of the product. Additionally, the new approach optimizes the molar ratios of reagents, specifically reducing the required amount of valeric anhydride while maintaining high conversion rates. By utilizing valerate salts and controlling the temperature profile effectively, the process achieves a cleaner reaction profile. This results in a crude product with higher purity, reducing the burden on downstream purification steps and ensuring a more consistent supply of the intermediate for subsequent drug synthesis.
Mechanistic Insights into Acylation and Tautomerization
The core of this synthetic strategy involves the acylation of 5-nitro-3H-benzofuran-2-one at the 3-position using valeric anhydride in the presence of a valerate salt base. The reaction proceeds through an enolate intermediate formed by the deprotonation of the benzofuran ring, which then attacks the carbonyl carbon of the anhydride. This nucleophilic attack leads to the formation of the 3-acyl derivative, which exists in equilibrium with its enol form, 3-(1-hydroxy-pentylidene)-5-nitro-3H-benzofuran-2-one. The presence of the nitro group on the benzene ring influences the electronic density of the system, stabilizing the intermediate and directing the regioselectivity of the acylation. Understanding this mechanistic pathway is crucial for R&D teams aiming to replicate the process, as slight deviations in base strength or temperature can shift the equilibrium or promote side reactions.
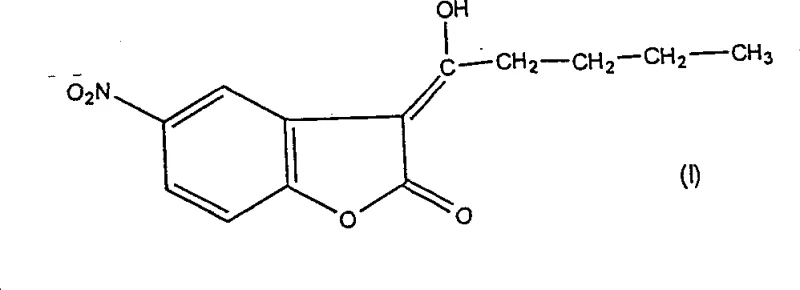
Furthermore, the patent highlights the existence of keto-enol tautomerism, where the hydroxy-pentylidene form (Formula I) can interconvert with the oxo-amyl form (Formula II). The stability of the enol form is favored under the specific reaction conditions employed, particularly due to intramolecular hydrogen bonding and conjugation with the lactone carbonyl. The final acidification step serves to protonate any remaining carboxylate salts and neutralize the reaction medium, locking the product into its stable solid form. This control over the tautomeric state is vital for ensuring the chemical integrity of the intermediate before it undergoes further transformation into 2-butyl-5-nitrobenzofuran. For quality control laboratories, monitoring this tautomeric ratio via NMR or HPLC ensures that the material meets the stringent specifications required for API synthesis.
How to Synthesize 3-(1-Hydroxy-Pentylidene)-5-Nitro-3H-Benzofuran-2-One Efficiently
The practical execution of this synthesis requires careful attention to reagent addition and temperature control to maximize yield and safety. The process begins with the combination of 5-nitro-3H-benzofuran-2-one, valeric anhydride, and a valerate salt in a suitable reactor, followed by heating to promote the acylation. Detailed operational parameters, including specific molar ratios and cooling rates, are critical for reproducibility. The following guide outlines the standardized steps derived from the patent examples to assist process chemists in implementing this route effectively.
- React 5-nitro-3H-benzofuran-2-one with valeric anhydride and a valerate salt (such as sodium valerate) at temperatures exceeding 30°C, preferably around 80°C, to form the crude intermediate.
- Cool the reaction mixture and perform a critical acidification step using dilute sulfuric acid to precipitate the desired product and improve separation efficiency.
- Isolate the solid product via filtration, wash with water and heptane, and purify through recrystallization in acetic acid to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis offers tangible benefits for procurement managers and supply chain heads focused on cost efficiency and continuity. The optimization of reagent usage directly impacts the bill of materials, while the improved isolation technique reduces processing time and solvent consumption. These factors collectively contribute to a more sustainable and economically viable manufacturing process, making the supplier of this intermediate a strategic partner for pharmaceutical companies.
- Cost Reduction in Manufacturing: The significant reduction in the amount of valeric anhydride required for the reaction translates directly into lower raw material costs. Unlike conventional methods that rely on vast excesses of anhydride to drive equilibrium, this process operates efficiently with a stoichiometric ratio of 1 to 5 moles, and optimally even lower. Furthermore, the elimination of complex purification steps due to the high purity of the precipitated crude product reduces the consumption of solvents and energy. This streamlined approach minimizes waste disposal costs and enhances the overall economic feasibility of producing this high-value intermediate on a commercial scale.
- Enhanced Supply Chain Reliability: The robustness of the synthesis, characterized by its tolerance to standard industrial conditions and the use of readily available reagents like valeric acid and sodium carbonate, ensures a stable supply chain. The simplified workup procedure, which avoids intricate extraction or chromatography steps, reduces the risk of batch failures and delays. For supply chain planners, this means shorter lead times and a more predictable delivery schedule for the intermediate. The ability to produce the compound with consistent quality reduces the need for extensive re-testing or re-processing, further securing the supply line for downstream API manufacturing.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram to kilogram scales within the patent examples. The use of aqueous acidification and simple filtration aligns well with green chemistry principles by reducing the reliance on hazardous organic solvents for extraction. The potential for recycling solvents like heptane and acetic acid further enhances the environmental profile of the manufacturing process. For facilities operating under strict environmental regulations, this method offers a compliant pathway to scale up production without necessitating major infrastructure upgrades or incurring excessive waste treatment costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this benzofuran derivative. These answers are derived directly from the technical disclosures in patent CN1306000A, providing clarity on the process capabilities and product specifications.
Q: What is the primary advantage of the acidification step in this synthesis?
A: The acidification step significantly improves the separation and recovery of the desired product from the reaction mixture, addressing purification challenges found in older methods.
Q: How does this method reduce raw material costs compared to conventional techniques?
A: The process optimizes the stoichiometry of valeric anhydride, requiring significantly less excess reagent (1 to 5 moles) compared to traditional methods, thereby lowering material costs.
Q: Can this intermediate be directly converted into Dronedarone precursors?
A: Yes, the compound can be efficiently converted into 2-butyl-5-nitrobenzofuran, a direct precursor for the antiarrhythmic drug Dronedarone, via hydrolysis and decarboxylation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(1-Hydroxy-Pentylidene)-5-Nitro-3H-Benzofuran-2-One Supplier
As the global demand for cardiovascular therapeutics continues to rise, securing a dependable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of 3-(1-hydroxy-pentylidene)-5-nitro-3H-benzofuran-2-one meets stringent purity specifications, fully compliant with international regulatory standards. We understand the critical nature of this intermediate in the Dronedarone supply chain and are committed to delivering consistency and excellence.
We invite you to collaborate with our technical team to explore how this optimized synthesis can benefit your specific project requirements. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify the suitability of our material for your process. Let us be your trusted partner in advancing the next generation of antiarrhythmic medications through superior chemical manufacturing.
