Advanced Copper-Catalyzed Synthesis of Ketone-Substituted Ionones for High-Value Fragrance Applications
Advanced Copper-Catalyzed Synthesis of Ketone-Substituted Ionones for High-Value Fragrance Applications
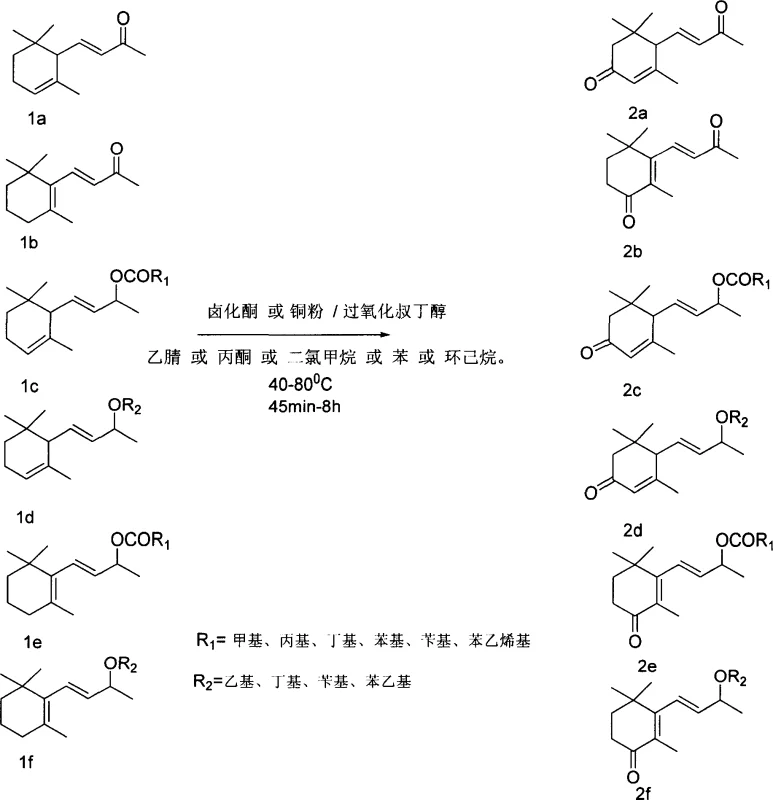
The global demand for high-purity fragrance intermediates continues to drive innovation in synthetic organic chemistry, particularly for complex terpenoid structures like ionones. Patent CN1817842A introduces a transformative one-step synthesis method for producing ketone-substituted alpha-ionone, ketone-substituted beta-ionone, and their respective ether and ester derivatives. This technology represents a significant paradigm shift from traditional stoichiometric oxidation methods, utilizing a catalytic system composed of copper halides or active copper powder alongside tert-butyl peroxide. By operating within a moderate temperature range of 40-80°C, this process achieves superior conversion rates while adhering to modern green chemistry principles. For R&D directors and technical leaders, this patent offers a robust pathway to access rare and unique aroma compounds that are critical for fruit, tobacco, and floral essence formulations. The elimination of heavy metal waste streams and the utilization of inexpensive, earth-abundant copper catalysts position this technology as a cornerstone for sustainable manufacturing in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the allylic oxidation of the cyclohexene ring in alpha-ionone and beta-ionone derivatives has been a challenging transformation plagued by low efficiency and severe environmental hazards. Traditional methodologies, such as those employing stoichiometric chromium reagents like chromium trioxide or tert-butyl chromate, have long been the standard despite their significant drawbacks. These legacy processes typically suffer from poor atom economy, resulting in yields that rarely exceed 20%, which drastically inflates the cost of goods sold due to material loss. Furthermore, the generation of toxic chromium-containing waste streams imposes a heavy burden on wastewater treatment facilities and requires specialized, costly disposal protocols to meet environmental regulations. The use of such hazardous oxidants also introduces safety risks related to handling and storage, complicating the scale-up process for commercial production. Additionally, older methods often lack regioselectivity, leading to complex mixtures of oxidation products that require extensive and yield-eroding purification steps to isolate the desired ketone functionality at the 3 or 4 positions of the ring.
The Novel Approach
In stark contrast to these obsolete techniques, the novel approach detailed in the patent utilizes a catalytic cycle driven by copper species and tert-butyl peroxide to achieve efficient allylic oxidation. This method operates under much milder conditions, typically between 40°C and 80°C, and utilizes a controlled addition strategy where the oxidant is introduced in three distinct portions to maintain reaction stability and maximize selectivity. The versatility of this system is demonstrated by its ability to tolerate various functional groups, successfully converting not only the parent ionones but also their ether and ester derivatives into valuable ketone products with yields ranging significantly higher than historical benchmarks. By replacing toxic chromium with catalytic copper, the process inherently reduces the environmental footprint, eliminating the generation of heavy metal sludge and simplifying the workup procedure. The reaction can be conducted in a variety of common organic solvents such as acetonitrile, acetone, or dichloromethane, providing process engineers with the flexibility to optimize for cost or solubility without compromising reaction performance.
Mechanistic Insights into Copper-Catalyzed Allylic Oxidation
The core of this technological advancement lies in the radical-mediated mechanism facilitated by the copper catalyst. The reaction initiates with the interaction between the copper species (either halide or active powder) and tert-butyl peroxide, generating reactive radical intermediates capable of abstracting an allylic hydrogen atom from the ionone substrate. This hydrogen abstraction creates a resonance-stabilized allylic radical on the cyclohexene ring, which is the key determinant for the regioselectivity observed in the formation of 3-oxo or 4-oxo derivatives. The subsequent rebound of the oxygen species or further oxidation steps lead to the installation of the carbonyl group, effectively transforming the inert C-H bond into a highly reactive ketone functionality. This mechanistic pathway is highly advantageous because it avoids the harsh acidic or basic conditions often required by other oxidation methods, thereby preserving sensitive functional groups present in the ether or ester side chains of the starting materials. The controlled addition of the peroxide oxidant in three stages is critical to managing the concentration of radical species, preventing over-oxidation or polymerization side reactions that could degrade the delicate terpene skeleton.
From an impurity control perspective, this catalytic system offers a cleaner reaction profile compared to stoichiometric oxidants. The primary byproducts of the reaction are tert-butanol and water, both of which are relatively benign and easily removed during the workup phase via distillation or extraction. The absence of chromium residues means that the final product does not require aggressive chelation or washing steps to meet stringent heavy metal specifications, which is crucial for applications in flavors and fragrances where purity is paramount. The selectivity of the copper catalyst ensures that the double bond in the side chain remains intact, preserving the structural integrity required for the specific olfactory characteristics of the target molecule. Furthermore, the ability to tune the reaction by adjusting the ratio of copper catalyst to substrate allows chemists to balance reaction rate against selectivity, minimizing the formation of over-oxidized byproducts or ring-opened degradation products that often plague radical oxidation chemistries.
How to Synthesize Ketone-Substituted Ionones Efficiently
The practical implementation of this synthesis route is designed for scalability and operational simplicity, making it highly attractive for industrial adoption. The general procedure involves charging a reactor with the specific ionone derivative substrate, a suitable organic solvent, and the copper catalyst source. The reaction mixture is then heated to the target temperature, typically around 55°C for optimal balance between rate and selectivity, while the oxidant is added in a controlled manner. The specific protocol dictates adding one-third of the tert-butyl peroxide initially, followed by subsequent additions at defined time intervals to sustain the catalytic cycle without overwhelming the system. After the final addition and a holding period to ensure complete conversion, the reaction mixture is worked up by removing volatiles under reduced pressure and quenching any residual peroxides with a sodium sulfite solution. The crude product is then extracted and purified, typically using silica gel chromatography, to yield the high-purity ketone-substituted ionone ready for downstream formulation or further derivatization.
- Prepare the reaction mixture by combining the ionone substrate (alpha or beta derivatives), organic solvent (acetonitrile, acetone, or DCM), and copper catalyst (halide or powder) in a reactor equipped with stirring and condensation.
- Maintain the reaction temperature between 40°C and 80°C while adding tert-butyl peroxide (TBHP) in three distinct portions over a period ranging from 45 minutes to 8 hours to control exothermicity and selectivity.
- Upon completion, remove solvents and excess oxidant under reduced pressure, quench residual peroxides with sodium sulfite solution, extract the product with ether, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology offers substantial strategic benefits beyond mere technical performance. The shift away from chromium-based reagents immediately mitigates regulatory risks associated with hazardous substance handling and disposal, ensuring long-term compliance with increasingly strict environmental laws globally. This transition also decouples production costs from the volatility of specialized oxidant markets, as copper salts and tert-butyl peroxide are commodity chemicals available from multiple global suppliers, enhancing supply chain resilience. The simplified workup procedure, which eliminates the need for complex heavy metal scavenging steps, translates directly into reduced processing time and lower utility consumption per kilogram of product. Consequently, manufacturers can achieve a more predictable and stable cost structure, insulating the business from the price fluctuations often seen with proprietary or scarce reagents. The high yield profile of this method further amplifies these economic advantages by maximizing the output from each batch of raw materials, effectively lowering the unit cost of production without requiring capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chromium reagents removes a significant cost center associated with both the purchase of expensive oxidants and the disposal of toxic waste. By utilizing catalytic amounts of inexpensive copper salts, the direct material cost of the reaction is drastically reduced. Furthermore, the simplified purification process reduces the consumption of solvents and stationary phases required for chromatography, leading to additional operational savings. The higher overall yield means that less raw material is wasted, improving the mass balance efficiency of the entire production line. These factors combine to create a leaner manufacturing process that delivers significant cost advantages over legacy methods, allowing for more competitive pricing in the marketplace while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like copper halides and common organic solvents ensures that the supply chain is not vulnerable to single-source bottlenecks. Unlike specialized chromium reagents which may have limited suppliers or face shipping restrictions due to hazard classifications, the inputs for this process are widely available globally. This abundance allows for flexible sourcing strategies and bulk purchasing opportunities that can further drive down costs. Additionally, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by sensitivity to minor variations in reagent quality or environmental conditions. This reliability is critical for meeting the just-in-time delivery expectations of major fragrance and flavor houses, ensuring consistent availability of these high-value intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory scale to potential multi-ton production without fundamental changes to the chemistry. The absence of toxic heavy metal waste simplifies the environmental permitting process for new production lines and reduces the liability associated with long-term waste storage. This "green" credential is increasingly valuable in B2B negotiations, as end-users in the consumer goods sector demand sustainable sourcing for their ingredients. The ability to market these intermediates as produced via an environmentally friendly process adds intangible value to the supply chain, aligning with corporate sustainability goals and enhancing the brand reputation of all partners involved in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed oxidation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is essential for R&D teams planning pilot runs and for procurement teams assessing the risk profile of adopting this new supply source. The answers highlight the versatility of the method across different substrate classes and its alignment with modern manufacturing standards.
Q: What are the primary advantages of the copper-catalyzed method over traditional chromium oxidation?
A: The copper-catalyzed method described in patent CN1817842A eliminates the use of stoichiometric toxic chromium reagents, thereby removing the need for hazardous waste disposal and significantly improving the environmental profile of the manufacturing process while achieving yields of 40-80% compared to historical yields of roughly 20%.
Q: Which solvents are compatible with this allylic oxidation process?
A: The process demonstrates robust compatibility with a variety of common organic solvents including acetonitrile, acetone, dichloromethane, benzene, and cyclohexane, allowing flexibility in solvent selection based on cost, boiling point requirements, and downstream processing needs.
Q: Can this method be applied to ether and ester derivatives of ionones?
A: Yes, the methodology is highly versatile and successfully applies to alpha and beta-ionone ethers (such as ethyl, butyl, and benzyl ethers) and esters (such as acetate, butyrate, and benzoate), enabling the production of a diverse library of fragrance intermediates with unique olfactory profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketone-Substituted Ionone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality fragrance intermediates through sustainable and efficient manufacturing routes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative copper-catalyzed synthesis described in patent CN1817842A can be seamlessly transferred to an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ketone-substituted ionone meets the exacting standards required by the global flavors and fragrances industry. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your own production stability and product quality.
We invite you to engage with our technical team to explore how this advanced synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient process for your supply chain. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Contact our technical procurement team today to discuss how we can collaborate to bring these high-value aroma compounds to your market faster and more sustainably.
