Advanced Acetylation Strategy for High-Purity 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-pentenyl Acetate Production
The chemical landscape for acyclic monoterpenoid enol esters has long been dominated by oxidative strategies that often suffer from stereoselectivity limitations and harsh processing conditions. Patent CN103833691A introduces a transformative preparation method for 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-pentenyl acetate, a compound of significant value in both the fragrance and pharmaceutical sectors. This innovation pivots away from the traditional reliance on geraniol acetate epoxidation, instead utilizing a direct acetylation of the corresponding epoxy-alcohol precursor. By leveraging sodium acetate as a mild base catalyst in conjunction with acetic anhydride, the process achieves a unique 1:1 mass ratio of cis and trans isomers, a profile unattainable through legacy oxidative pathways. This technical breakthrough not only simplifies the synthetic route but also inherently enhances the safety profile by eliminating the need for hazardous organic peracids or urea-hydrogen peroxide complexes typically required in prior art. For industrial stakeholders, this represents a shift towards more predictable, scalable, and economically viable manufacturing protocols that align with modern green chemistry principles while delivering superior stereochemical diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-pentenyl acetate has been constrained by the inherent limitations of olefin epoxidation strategies applied to geraniol acetate. As documented in earlier literature, such as the 1968 work by Govindan VN, the use of organic peroxyacids at low temperatures (0-10°C) yielded exclusively the trans-isomer with moderate efficiency around 68.75%. Subsequent improvements by researchers like Harry H in 1993 utilized urea hydrogen peroxide systems, pushing yields to approximately 82%, yet the stereochemical outcome remained stubbornly fixed on the (2E)-trans configuration. These oxidative methods introduce significant process complexities, including the handling of unstable oxidizing agents, strict temperature controls to prevent runaway exotherms, and the generation of stoichiometric amounts of acidic or nitrogenous waste byproducts. Furthermore, the inability to access the cis-isomer limits the utility of the final product in applications where specific olfactory notes or biological activities associated with the (2Z)-configuration are desired, forcing formulators to rely on incomplete chemical profiles or expensive separation techniques.
The Novel Approach
In stark contrast, the methodology disclosed in CN103833691A circumvents these oxidative hurdles entirely by adopting a nucleophilic acyl substitution strategy on a pre-formed epoxide alcohol. By reacting 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-penten-1-ol directly with acetic anhydride in the presence of sodium acetate, the process operates under remarkably mild thermal conditions of 30-40°C. This approach preserves the delicate epoxide ring structure that is often vulnerable to acid-catalyzed ring-opening under harsher oxidative conditions. Crucially, because the stereochemistry is established in the precursor alcohol rather than during the reaction itself, the method faithfully transfers the geometric configuration of the starting material, resulting in a final product containing both cis and trans isomers in a precise 1:1 mass ratio. This dual-isomer output expands the functional utility of the intermediate for complex fragrance accords or pharmaceutical syntheses, while the avoidance of strong oxidants drastically reduces safety risks and waste treatment costs associated with peroxide disposal.
Mechanistic Insights into Sodium Acetate-Catalyzed Acetylation
The core of this synthetic advancement lies in the elegant simplicity of the base-catalyzed acetylation mechanism, which proceeds through a well-defined nucleophilic pathway. Initially, the sodium acetate acts as a weak base to deprotonate the hydroxyl group of the 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-penten-1-ol substrate, generating a reactive epoxy-alkoxide intermediate in situ. This alkoxide species is a potent nucleophile that rapidly attacks the electrophilic carbonyl carbon of the acetic anhydride. The subsequent collapse of the tetrahedral intermediate expels an acetate ion, forming the desired ester bond and regenerating the catalytic base species. This cycle ensures high atom economy regarding the catalyst, as the sodium acetate is not consumed but rather facilitates the proton transfer necessary for activation. The mild basicity of the acetate ion is particularly critical; it is sufficiently strong to activate the alcohol but too weak to induce elimination reactions or epoxide ring-opening, thereby maintaining the structural integrity of the 3,3-dimethyloxirane moiety throughout the 5-hour reaction duration.
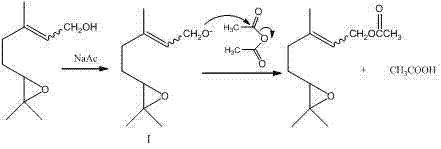
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed esterification or oxidative routes. The absence of strong acids prevents the isomerization of the double bond or the hydrolysis of the epoxide ring, which are common degradation pathways in terpene chemistry. Furthermore, the use of acetic anhydride as the acylating agent drives the reaction to completion by irreversibly forming acetic acid as a byproduct, which is easily removed during the aqueous workup. The reaction kinetics are optimized by maintaining the temperature between 30-40°C; lower temperatures would slow the nucleophilic attack unnecessarily, while higher temperatures could risk thermal degradation of the sensitive terpene backbone. This precise control over the reaction environment ensures that side products are minimized, leading to a crude product purity that is exceptionally high even before chromatographic purification, thus streamlining the downstream processing requirements for industrial scale-up.
How to Synthesize 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-pentenyl Acetate Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and thermal parameters defined in the patent to ensure reproducibility and optimal yield. The process begins with the preparation of a homogeneous mixture containing the epoxy-alcohol substrate, acetic anhydride, and sodium acetate, which is then subjected to controlled heating and agitation. The subsequent workup involves a careful neutralization sequence to remove residual acids and bases without compromising the product stability, followed by extraction and concentration. Finally, high-resolution purification is achieved through silica gel chromatography using a specific solvent gradient to separate the target esters from any minor impurities.
- Mix 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-penten-1-ol with acetic anhydride and sodium acetate, reacting at 30-40°C for 5 hours.
- Wash the reaction mixture sequentially with 10% Na2CO3 and 5% HCl solutions until neutral pH is achieved, then extract with ether.
- Purify the crude product via silica gel column chromatography using a petroleum ether and ethyl acetate solvent system to isolate the final ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this acetylation-based methodology offers substantial strategic benefits regarding cost structure and operational reliability. The elimination of expensive and hazardous oxidizing agents, such as organic peracids or urea-hydrogen peroxide complexes, directly reduces the raw material expenditure and lowers the regulatory burden associated with storing and transporting dangerous goods. Additionally, the mild reaction conditions of 30-40°C significantly decrease energy consumption compared to processes requiring cryogenic cooling or high-temperature reflux, contributing to a lower overall carbon footprint and reduced utility costs per kilogram of produced intermediate. The robustness of the reaction also implies fewer batch failures due to thermal runaways or sensitivity to moisture, thereby enhancing the predictability of production schedules and ensuring consistent availability for downstream customers.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of commodity chemicals like acetic anhydride and sodium acetate, which are widely available and priced significantly lower than specialized oxidants. By avoiding the need for complex waste treatment systems required for peroxide residues, the facility can achieve significant operational expenditure savings. Furthermore, the high crude yield ranging from 83.04% to 91.60% minimizes material loss, ensuring that a greater proportion of the input feedstock is converted into saleable product, which directly improves the gross margin profile for large-scale manufacturing campaigns.
- Enhanced Supply Chain Reliability: The starting material, 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-penten-1-ol, is noted for its ready availability and low cost, reducing the risk of supply bottlenecks that often plague specialty chemical supply chains. The simplicity of the one-pot reaction design reduces the number of unit operations required, shortening the overall cycle time from raw material intake to finished goods. This streamlined workflow allows for faster turnaround times on customer orders and provides the flexibility to scale production up or down in response to market demand fluctuations without the need for extensive retooling or specialized equipment modifications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial volumes is facilitated by the non-exothermic nature of the acetylation step, which removes the need for complex heat exchange systems typically required for safe epoxidation reactions. The waste stream primarily consists of acetic acid and salt solutions, which are far easier to treat and neutralize than the nitrogenous or chlorinated waste streams associated with alternative oxidative methods. This alignment with environmental compliance standards reduces the risk of regulatory penalties and supports corporate sustainability goals, making the supply chain more resilient to evolving environmental legislation.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and quality attributes of this synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this intermediate into their own formulation or synthesis pipelines.
Q: What is the primary advantage of this synthesis method over traditional epoxidation routes?
A: Unlike conventional methods using geraniol acetate which exclusively yield the trans-isomer, this patented acetylation route successfully produces a 1:1 mass ratio mixture of both cis and trans isomers, offering broader application potential in fragrance profiling.
Q: What are the critical reaction conditions for maintaining epoxide stability?
A: The process strictly maintains a temperature range of 30-40°C during the 5-hour reaction window. This mild thermal profile prevents the acid-catalyzed ring-opening of the sensitive 3,3-dimethyloxirane moiety, ensuring high structural integrity.
Q: What purity levels can be achieved after purification?
A: Following silica gel column chromatography with a optimized petroleum ether to ethyl acetate ratio, the final product achieves a purity exceeding 98.00%, suitable for high-end flavor and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(3,3-dimethyloxirane-2-yl)-3-methyl-2-pentenyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex fine chemicals requires more than just a patent; it demands rigorous process engineering and unwavering quality control. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering this high-value intermediate with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the >98.00% purity standard required for sensitive fragrance and pharmaceutical applications. Our infrastructure is designed to handle the specific handling requirements of epoxy-containing compounds, guaranteeing product stability and consistency upon delivery.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain efficiency and product performance. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this acetylation method for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
