Advanced One-Pot Synthesis of 2,9-Diamide-1,10-Phenanthroline Derivatives for Commercial Scale-Up
The chemical landscape for synthesizing complex heterocyclic ligands is undergoing a significant transformation, driven by the urgent need for greener, more efficient manufacturing protocols. Patent CN111116581A introduces a groundbreaking methodology for the production of 2,9-diamide-1,10-phenanthroline compounds, which are critical intermediates in the fields of radiochemistry and pharmaceutical analysis. This innovation specifically addresses the longstanding challenges associated with the functionalization of the 1,10-phenanthroline skeleton, traditionally a cumbersome process plagued by toxic reagents and low efficiency. By shifting the synthetic starting point to 2,9-dicarboxylic acid-1,10-phenanthroline, the disclosed method bypasses the hazardous oxidation steps previously required, establishing a new benchmark for sustainability in fine chemical synthesis.
For R&D directors and process chemists, the implications of this technology are profound, offering a robust pathway to high-purity materials essential for lanthanide and actinide separation processes. The structural versatility of these compounds allows for precise tuning of extraction dynamics, making them invaluable for nuclear waste treatment applications where separation factors must exceed 100. Furthermore, the simplified workflow reduces the operational burden on laboratory personnel, minimizing exposure to corrosive acids and volatile organic compounds. As we delve into the technical specifics, it becomes clear that this patent represents not just a incremental improvement, but a fundamental rethinking of how these valuable chelating agents are constructed.
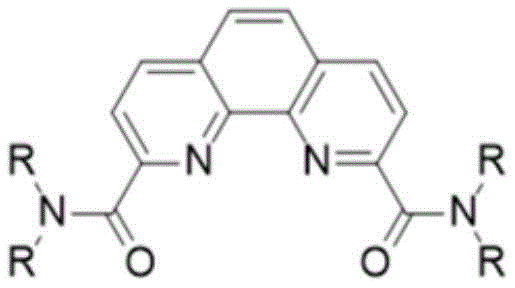
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,9-diamide-1,10-phenanthroline derivatives has been hindered by a reliance on 2,9-dimethyl-1,10-phenanthroline as the primary starting material. This conventional route necessitates a perilous four-step sequence, beginning with a two-stage oxidation process to convert methyl groups into carboxylic acids. This oxidation phase typically employs selenium dioxide, a highly toxic oxidant, and concentrated nitric acid, a strong corrosive agent, creating severe environmental and safety liabilities for manufacturing facilities. The cumulative yield of this traditional pathway is often dismally low, frequently falling below 30%, which renders the process economically unviable for large-scale commercial production. Additionally, the post-reaction processing is notoriously difficult, requiring extensive extraction procedures and column chromatography using chloroform, a regulated solvent that complicates waste disposal and increases operational costs significantly.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes 2,9-dicarboxylic acid-1,10-phenanthroline as the direct feedstock, effectively truncating the synthetic route to just two critical steps performed in a single vessel. This one-pot strategy involves an initial acyl chlorination followed immediately by amidation, eliminating the need to isolate the reactive acyl chloride intermediate. By removing the oxidation steps entirely, the process eradicates the use of selenium dioxide and nitric acid, aligning perfectly with modern green chemistry principles. The operational simplicity is further enhanced by the avoidance of liquid-liquid extraction, which often leads to product loss due to the water solubility of polar derivatives. Instead, the method employs a streamlined purification protocol combining flash column chromatography with recrystallization, resulting in superior product recovery and significantly reduced solvent consumption.
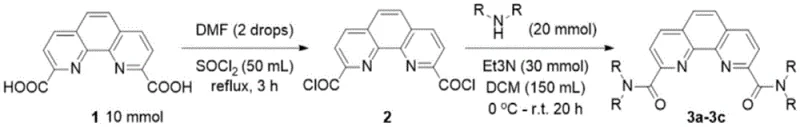
Mechanistic Insights into One-Pot Acyl Chlorination and Amidation
The core of this technological advancement lies in the seamless integration of acyl chlorination and nucleophilic substitution within a unified reaction system. The mechanism initiates with the activation of the carboxylic acid groups on the phenanthroline scaffold using thionyl chloride (SOCl2), catalyzed by trace amounts of N,N-dimethylformamide (DMF). This step generates the highly reactive 2,9-diacid chloride intermediate in situ. Crucially, rather than isolating this moisture-sensitive species, the reaction mixture is simply concentrated to remove excess thionyl chloride, and the crude residue is immediately subjected to amidation. This continuity prevents the degradation of the intermediate and maintains high reaction momentum. The subsequent addition of secondary amines in the presence of an acid scavenger like triethylamine facilitates a rapid nucleophilic attack on the carbonyl carbons, forming the stable amide bonds that define the final product structure.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise synthesis. In traditional methods, the isolation of the acid chloride often leads to hydrolysis upon exposure to atmospheric moisture, generating carboxylic acid impurities that are difficult to separate from the final amide. By maintaining an anhydrous environment throughout the one-pot sequence, the formation of these hydrolysis byproducts is minimized. Furthermore, the use of flash column chromatography with a sand core funnel allows for efficient separation of unreacted amines and minor side products without the need for large volumes of methanol/chloroform eluents. The final recrystallization from ethanol serves as a polishing step, ensuring that the resulting white solid meets stringent purity specifications required for analytical and extractant applications, thereby guaranteeing consistent performance in downstream processes.
How to Synthesize 2,9-Diamide-1,10-Phenanthroline Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the careful addition of thionyl chloride to the dicarboxylic acid starting material in batches to manage exothermic heat release, followed by refluxing at 80°C for approximately 3 hours. Once the acyl chloride is formed, the system is cooled, and the amidation is conducted at 0°C to room temperature over a period of 10 to 20 hours. The detailed standardized operating procedures, including specific molar ratios, solvent volumes, and purification techniques such as the specific use of G3 sand core funnels for filtration, are critical for reproducibility. For a comprehensive guide on implementing this protocol in your facility, please refer to the structured steps below.
- Acyl Chlorination: Reflux 2,9-dicarboxylic acid-1,10-phenanthroline with thionyl chloride and catalytic DMF at 80°C for 3 hours, then remove excess thionyl chloride.
- Amidation: Dissolve the crude acyl chloride in dichloromethane, cool to 0°C, and add secondary amine with triethylamine, stirring until completion.
- Purification: Perform flash column chromatography followed by recrystallization in ethanol to obtain high-purity white solid products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere technical elegance. The elimination of hazardous oxidants like selenium dioxide removes a significant regulatory burden and reduces the costs associated with hazardous waste disposal and worker safety compliance. By shortening the synthetic route from four steps to two, the overall throughput of the manufacturing process is drastically increased, allowing for faster response times to market demand. The reduction in solvent usage, particularly the avoidance of chloroform in favor of more manageable systems like dichloromethane and ethanol, further drives down the variable costs of production. These efficiencies collectively contribute to a more resilient supply chain capable of delivering high-quality intermediates with greater reliability.
- Cost Reduction in Manufacturing: The streamlined one-pot process significantly lowers manufacturing costs by reducing the number of unit operations and eliminating the need for expensive and toxic reagents. By avoiding the isolation of intermediates, labor hours and equipment utilization time are minimized, leading to substantial operational savings. Furthermore, the improved overall yield means that less raw material is required to produce the same amount of final product, directly enhancing the cost-efficiency of the supply chain. The simplified purification process also reduces the consumption of high-purity solvents, which are often a major cost driver in fine chemical synthesis.
- Enhanced Supply Chain Reliability: Relying on a shorter, more robust synthetic route mitigates the risk of production delays caused by complex multi-step sequences. The use of readily available starting materials like 2,9-dicarboxylic acid-1,10-phenanthroline ensures a stable supply of feedstock, reducing vulnerability to raw material shortages. The ability to perform the reaction in a single vessel simplifies equipment requirements, allowing for greater flexibility in manufacturing scheduling and capacity allocation. This operational agility enables suppliers to maintain consistent delivery schedules even in the face of fluctuating market demands or logistical challenges.
- Scalability and Environmental Compliance: The green chemistry attributes of this method facilitate easier scale-up from laboratory to commercial production without encountering the safety bottlenecks associated with toxic oxidants. The reduced generation of hazardous waste simplifies environmental compliance and lowers the overhead costs related to waste treatment and disposal. The use of recrystallization as a final purification step is inherently scalable and produces high-purity solids suitable for industrial applications. This alignment with environmental standards not only future-proofs the manufacturing process but also enhances the corporate sustainability profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these phenanthroline derivatives. The answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing accurate guidance for potential users and buyers. Understanding these nuances is essential for evaluating the feasibility of integrating these compounds into your specific research or production workflows. We encourage you to review these insights to fully appreciate the value proposition of this advanced synthetic methodology.
Q: What are the advantages of using 2,9-dicarboxylic acid over 2,9-dimethyl-1,10-phenanthroline?
A: Using 2,9-dicarboxylic acid eliminates the need for a hazardous two-step oxidation process involving toxic selenium dioxide and corrosive nitric acid, significantly shortening the synthetic route and improving environmental safety.
Q: How does the one-pot synthesis method improve product yield?
A: By avoiding the isolation and transfer of the unstable acyl chloride intermediate, the one-pot method minimizes product loss during workup and prevents hydrolysis, leading to higher overall yields compared to traditional multi-step protocols.
Q: What purification method is recommended for these phenanthroline derivatives?
A: The patent recommends a combination of flash column chromatography using a sand core funnel followed by recrystallization in ethanol, which effectively removes impurities without requiring large volumes of chlorinated solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,9-Diamide-1,10-Phenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in advancing radiochemical separation and pharmaceutical analysis. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We are committed to delivering materials that adhere to stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Whether you require custom derivatives or standard catalog items, our infrastructure is designed to support your most demanding projects with unwavering reliability.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your operations. Together, we can build a more sustainable and productive future for the fine chemical industry.
