Advanced Synthesis of N,5-Dimethyl-2-Oxazolamine for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the production of critical heterocyclic intermediates. Patent CN102702127A introduces a significant technological advancement in the synthesis of N,5-dimethyl-2-oxazolamine, a valuable building block in medicinal chemistry. This innovation addresses long-standing inefficiencies in conventional manufacturing by replacing hazardous alkylation reagents with a safer, direct condensation strategy using 1-methylurea. For R&D directors and procurement specialists, this patent represents a pivotal shift towards greener chemistry that does not compromise on yield or scalability. By integrating this novel approach, manufacturers can achieve a more robust supply chain for high-purity pharmaceutical intermediates while simultaneously mitigating regulatory risks associated with toxic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,5-dimethyl-2-oxazolamine has relied on a multi-step sequence that begins with the bromination of propanal to form 2-bromopropionaldehyde, followed by condensation with urea to yield 2-amino-5-methyloxazole. The critical bottleneck in this traditional route is the final methylation step, which necessitates the use of methyl iodide. Methyl iodide is a potent alkylating agent known for its high toxicity and carcinogenic potential, imposing severe handling restrictions and waste disposal costs on manufacturing facilities. Furthermore, the separation of the mono-methylated product from di-methylated byproducts and unreacted starting materials often requires complex purification protocols, leading to reduced overall yields and increased production timelines. These factors collectively inflate the cost of goods sold and introduce significant supply chain vulnerabilities due to the stringent regulatory controls on methyl iodide procurement and usage.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes 1-methylurea as a sophisticated bifunctional reagent that serves as both the nitrogen source and the methyl donor in a single operational step. This strategic substitution allows for the direct conversion of 2-bromopropionaldehyde into the target N,5-dimethyl-2-oxazolamine without the need for a separate alkylation stage. By collapsing the synthetic sequence, the process inherently reduces the number of unit operations, solvent exchanges, and isolation steps required. This streamlining not only accelerates the manufacturing cycle but also drastically minimizes the generation of chemical waste. The elimination of methyl iodide removes a major safety hazard from the plant floor, aligning the production process with modern green chemistry principles and reducing the environmental footprint of the facility.
Mechanistic Insights into 1-Methylurea Mediated Cyclization
The core of this technological breakthrough lies in the unique reactivity of 1-methylurea under thermal conditions. In the first stage, propanal undergoes alpha-bromination with liquid bromine at controlled low temperatures ranging from -20 to 10°C to generate 2-bromopropionaldehyde. Rather than isolating this unstable intermediate, the novel process proceeds directly to the cyclization phase. The 1-methylurea molecule attacks the electrophilic carbonyl carbon of the aldehyde, initiating a condensation reaction. Subsequently, the nitrogen atom adjacent to the methyl group participates in an intramolecular nucleophilic substitution, displacing the bromide ion to close the oxazole ring. This mechanism effectively installs the N-methyl group during the ring formation itself, bypassing the need for external methylating agents.
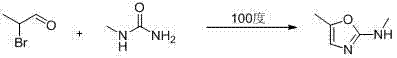
From an impurity control perspective, this mechanism offers distinct advantages. Traditional alkylation routes often suffer from over-alkylation, where the nitrogen atom reacts twice with methyl iodide to form quaternary ammonium salts or N,N-dimethyl byproducts that are difficult to separate. The 1-methylurea route inherently limits methylation to a single position because the methyl group is pre-installed on the urea scaffold. This structural constraint significantly simplifies the impurity profile of the crude reaction mixture. Consequently, downstream purification becomes more efficient, requiring less solvent and fewer chromatographic passes to achieve the stringent purity specifications demanded by pharmaceutical customers. This level of control is essential for maintaining batch-to-batch consistency in commercial scale-up.
How to Synthesize N,5-Dimethyl-2-Oxazolamine Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the careful addition of liquid bromine to propanal in a suitable solvent system, ensuring the temperature remains within the specified sub-zero to ambient range to prevent poly-bromination. Once the bromination is complete, indicated by the formation of a transparent solution, the reaction mixture is treated directly with 1-methylurea under a nitrogen atmosphere. The subsequent heating phase at 100°C for approximately 16 hours is critical for driving the cyclization to completion. Detailed standard operating procedures regarding stoichiometry, agitation rates, and workup protocols are essential for successful technology transfer.
- Perform controlled bromination of propanal with liquid bromine at low temperatures (-20 to 10°C) to generate 2-bromopropionaldehyde in situ.
- Without isolation, react the crude 2-bromopropionaldehyde solution with 1-methylurea in 1,4-dioxane solvent under nitrogen protection.
- Maintain the reaction mixture at 100°C for approximately 16 hours to facilitate cyclization, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible operational improvements and risk mitigation. The primary value driver is the complete removal of methyl iodide from the bill of materials. Methyl iodide is not only expensive due to its hazardous nature but also subject to volatile market availability and strict transportation regulations. By substituting this with 1-methylurea, a stable and widely available commodity chemical, the supply chain becomes significantly more resilient and less prone to disruption. This change also alleviates the burden on EHS departments, as the facility no longer needs to maintain specialized containment systems for highly toxic alkylating agents.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the reduction in synthetic steps and the elimination of costly reagents. By combining the amidation and methylation into a single thermal step, the process saves on labor, energy, and solvent consumption associated with intermediate isolation. Furthermore, the avoidance of methyl iodide removes the substantial costs linked to the disposal of halogenated hazardous waste and the remediation of equipment contaminated with toxic alkylating agents. These cumulative savings contribute to a lower cost of goods sold, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this new route enhances stability by relying on robust, non-hazardous raw materials. Propanal, bromine, and 1-methylurea are bulk chemicals with established global supply networks, ensuring consistent availability even during market fluctuations. The simplified process flow also reduces the likelihood of batch failures caused by complex multi-step manipulations. With fewer unit operations, there are fewer opportunities for human error or equipment malfunction, leading to higher on-time delivery rates and greater confidence for downstream API manufacturers who depend on timely intermediate supply.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology is inherently designed for large-scale production. The absence of highly toxic methyl iodide simplifies the permitting process for new manufacturing lines and reduces the regulatory scrutiny associated with emissions and effluent treatment. The reaction conditions, while requiring heat, do not demand extreme pressures or cryogenic temperatures beyond the initial bromination, making them compatible with standard stainless steel reactor infrastructure. This compatibility facilitates rapid scale-up from pilot plant to commercial tonnage, ensuring that supply can meet growing market demand without requiring prohibitive capital investment in specialized hazardous processing units.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the practical application and benefits of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear picture of why this method represents a superior alternative to legacy processes.
Q: Why is the 1-methylurea route superior to the traditional methyl iodide method?
A: The traditional method requires a separate methylation step using toxic methyl iodide, which poses significant environmental and safety hazards. The novel route utilizes 1-methylurea as a dual nitrogen and methyl source, eliminating the need for hazardous alkylating agents and shortening the synthetic sequence by one full step.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization between 2-bromopropionaldehyde and 1-methylurea requires elevated temperatures, specifically around 100°C, typically in a solvent like 1,4-dioxane. Maintaining an inert nitrogen atmosphere is crucial to prevent oxidation of the aldehyde intermediate and ensure high yield.
Q: How does this process impact impurity profiles for API manufacturing?
A: By avoiding the use of methyl iodide, the process eliminates specific genotoxic impurity risks associated with alkyl halides. Furthermore, the direct condensation reduces the formation of side products often seen in multi-step alkylation sequences, resulting in a cleaner crude profile that simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,5-Dimethyl-2-Oxazolamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to drive efficiency in the pharmaceutical sector. Our technical team has thoroughly analyzed the potential of this 1-methylurea mediated route and is prepared to leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We possess the rigorous QC labs and stringent purity specifications necessary to ensure that every batch of N,5-dimethyl-2-oxazolamine meets the highest international standards. Our commitment to continuous improvement allows us to offer clients a supply partner that is not only capable of delivering volume but also dedicated to optimizing the underlying chemistry for maximum value.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your production scale. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
