Advanced Iron-Catalyzed Synthesis of Alpha-Alkyl Amides for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies to construct nitrogen-containing scaffolds, particularly alpha-alkyl substituted amines and amides, which serve as critical building blocks in drug discovery. Patent CN108276233B introduces a groundbreaking approach to synthesizing these valuable motifs through an iron-catalyzed three-component coupling reaction. This technology addresses a long-standing challenge in organic synthesis: the direct amino-alkylation of alkenes using alkyl electrophiles. By leveraging inexpensive iron salts and peroxide initiators, this method provides a sustainable alternative to traditional noble metal-catalyzed processes, offering significant implications for the supply chain reliability of high-purity pharmaceutical intermediates.
The core innovation lies in the ability to transform simple alkenes, peroxides, and nitriles into complex amide structures under mild conditions. This represents a paradigm shift from conventional strategies that often rely on expensive catalysts or harsh reaction environments. For R&D directors focused on impurity profiles and process robustness, this iron-mediated pathway offers a cleaner reaction profile with manageable byproducts, facilitating easier purification and higher overall quality of the final active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-N and C-C bonds simultaneously via alkene functionalization has been dominated by transition metals such as copper, rhodium, and ruthenium. While these methods have achieved success in amino-arylation or amino-trifluoromethylation, they face severe limitations when attempting amino-alkylation. The primary bottleneck is the lack of effective alkyl electrophiles that can participate in these catalytic cycles without undergoing rapid decomposition or side reactions. Furthermore, ordinary alkyl carboxylic acids, which are ideal cheap feedstocks, form unstable radical species during decarboxylation, making them poor candidates for traditional cross-coupling. These constraints often force manufacturers to rely on multi-step syntheses involving pre-functionalized substrates, driving up costs and extending lead times for critical drug intermediates.
The Novel Approach
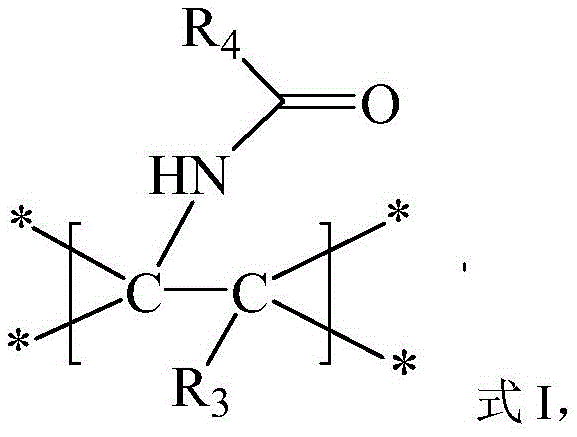
The methodology disclosed in CN108276233B circumvents these hurdles by utilizing peroxides derived from carboxylic acids as stable alkyl radical precursors. In this novel system, the iron catalyst activates the peroxide bond to generate alkyl radicals, which then add across the carbon-carbon double bond of the alkene. This is followed by trapping with a nitrile solvent and subsequent hydrolysis to yield the target amide. This strategy effectively bridges the gap in amino-carbonization chemistry, allowing for the direct use of abundant alkenes and carboxylic acid derivatives. The result is a streamlined synthetic route that reduces step count and eliminates the need for precious metal catalysts, thereby enhancing the economic viability of producing complex amine derivatives.
Mechanistic Insights into Fe-Catalyzed Radical Amino-Alkylation
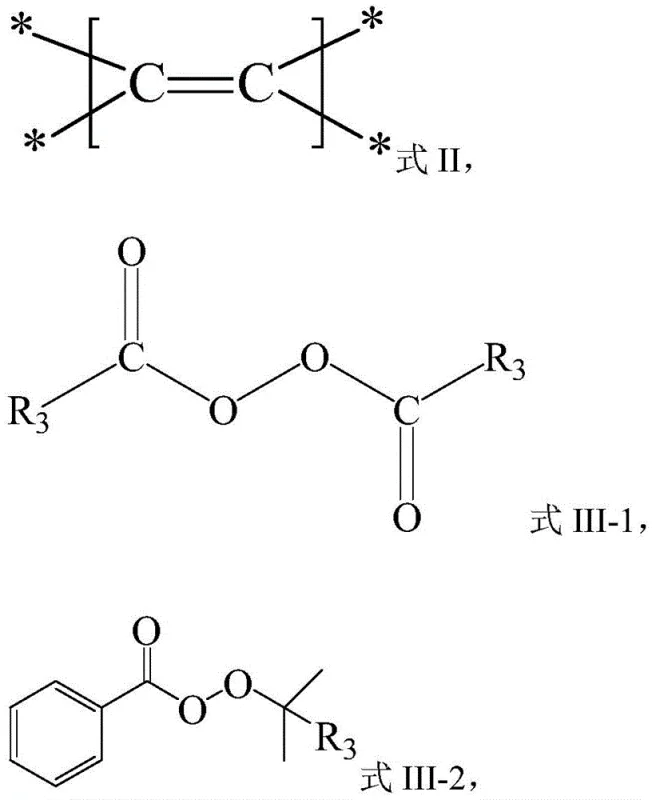
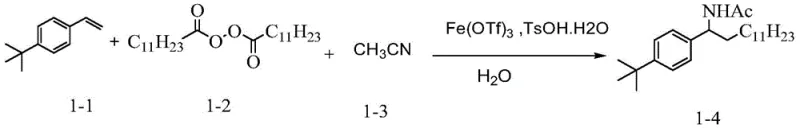
The reaction mechanism proceeds through a radical cascade initiated by the iron catalyst. Initially, the iron species, such as Fe(OTf)3 or FeCl2, interacts with the peroxide reagent (Compound III), facilitating homolytic cleavage of the weak O-O bond. This generates a reactive alkyl radical species along with an iron-oxo intermediate. The alkyl radical then undergoes regioselective addition to the electron-rich double bond of the alkene substrate (Compound II), forming a new carbon-centered radical at the benzylic or stabilized position. This intermediate radical is subsequently trapped by the nitrile group from the solvent or added reagent (Compound IV), forming an imidoyl radical. Finally, in the presence of water and acid additives like TsOH, the imidoyl species undergoes hydrolysis to furnish the stable amide product (Compound I). This mechanistic pathway ensures high atom economy and minimizes the formation of oligomeric byproducts common in free-radical polymerizations.
From an impurity control perspective, the use of iron salts and acidic additives creates a controlled environment that suppresses uncontrolled radical chain reactions. The specific choice of additives, such as p-toluenesulfonic acid monohydrate, plays a crucial role in protonating intermediates and facilitating the final hydrolysis step without degrading sensitive functional groups on the substrate. This level of control is essential for maintaining the stringent purity specifications required for pharmaceutical applications. The compatibility of this system with a wide range of substituents, including halogens, trifluoromethyl groups, and heterocycles, further underscores its versatility in generating diverse chemical libraries for medicinal chemistry campaigns.
How to Synthesize Alpha-Alkyl Amides Efficiently
The practical execution of this synthesis involves mixing the alkene, peroxide, and nitrile components in a suitable solvent system, typically acetonitrile or a mixture with dioxane. The reaction is promoted by catalytic amounts of iron salts and stoichiometric amounts of acid and water. The process is operationally simple, requiring standard heating equipment and atmospheric pressure, which makes it highly attractive for both laboratory-scale optimization and pilot plant operations. Detailed standardized synthesis steps follow below.
- Prepare the reaction mixture by combining the alkene substrate (Compound II), peroxide reagent (Compound III), and nitrile solvent/reactant (Compound IV) in an organic solvent such as acetonitrile.
- Add the iron catalyst, such as Iron(III) triflate (Fe(OTf)3) or Ferrous Chloride (FeCl2), along with an acidic additive like p-toluenesulfonic acid monohydrate (TsOH·H2O) and water.
- Heat the reaction mixture to a temperature between 60°C and 80°C under stirring for 4 to 6 hours, then cool, remove solvent, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology presents compelling strategic advantages. The shift from noble metals to iron drastically reduces raw material costs, as iron salts are orders of magnitude cheaper than rhodium or ruthenium complexes. Additionally, the removal of heavy metal residues from the final product becomes significantly easier, reducing the burden on quality control labs and minimizing the risk of batch rejection due to metal contamination. This translates directly into improved margin protection and more predictable production schedules for high-volume API intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like peroxides and nitriles lead to substantial cost savings in raw material procurement. Furthermore, the mild reaction conditions (60°C to 80°C) reduce energy consumption compared to high-temperature or high-pressure alternatives. The simplified workup procedure, often involving simple distillation and chromatography, lowers operational expenditures related to solvent recovery and waste disposal, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: Iron catalysts and peroxide reagents are widely available from multiple global suppliers, mitigating the risk of supply disruptions often associated with specialized noble metals. The robustness of the reaction against moisture and air, facilitated by the specific additive system, ensures consistent batch-to-batch reproducibility. This reliability is critical for maintaining continuous production lines and meeting tight delivery windows for downstream pharmaceutical customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its exothermic nature being manageable under standard cooling conditions and the absence of hazardous gaseous reagents. From an environmental standpoint, replacing toxic heavy metals with benign iron aligns with green chemistry principles and increasingly strict regulatory standards regarding heavy metal discharge. This facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing facility, supporting long-term sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility and process parameters.
Q: What are the primary advantages of using iron catalysts over noble metals for amide synthesis?
A: Iron catalysts offer significant cost reductions compared to traditional copper, rhodium, or ruthenium systems. They are also less toxic, more abundant, and operate effectively under mild conditions, simplifying downstream purification and waste treatment processes.
Q: Can this synthetic method accommodate complex alkene substrates?
A: Yes, the patent demonstrates high functional group tolerance. The method successfully processes various alkenes including styrenes with electron-donating or withdrawing groups, heterocyclic alkenes, and even complex steroid-derived alkenes, yielding the corresponding alpha-alkyl amides efficiently.
Q: Is the process scalable for industrial manufacturing of API intermediates?
A: The process utilizes commercially available raw materials and standard reaction conditions (60-80°C, atmospheric pressure). The absence of sensitive transition metals and the use of simple workup procedures like distillation and chromatography indicate strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Alkyl Amides Supplier
The technological advancements detailed in CN108276233B highlight the evolving landscape of amide synthesis, offering new opportunities for efficiency and cost optimization. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of alpha-alkyl amides delivered meets the highest industry standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to explore how this iron-catalyzed technology can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timelines and secure a competitive advantage in the market.
